Effect of Physical Variables on Laminar Burning Velocity of Liquid Fuels and Gases: A Review
Hayder Khudhair Obayes1*, Oras Khudhayer Obayes2
1Ministry of Education, Babylon, Iraq
2Department of Mechanical Engineering, Technical Institute of Babylon, Al-Furat Al-Awsat Technical University (ATU), Babylon, Iraq
*Correspondence to: Hayder Khudhair Obayes, Ministry of Education, 10001, Bab Al-Sharqi, Babylon, Iraq; Email: hayder.physics1@gmail.com
Abstract
Laminar burning velocity (LBV) is one of the most important properties of fuel combustion. Although the role of LBV on how combustion systems are designed and performed have been widely reported, the purpose of this study was to review which physical variables (initial pressure, initial temperature, flame temperature, thermal diffusivity impact, specific heat, and ignition energy) highlight this key combustion trait.
Keywords: burning velocity, bunsen burner, internal combustion engines
1 INTRODUCTION
Laminar burning velocity (LBV) is a one of the most central elements of a responsive premixed mixture, and reliable data on it is needed on a regular basis for combustion applications. Various approaches for measuring one-dimensional LBV have been used to date from the perspective of temperatures, pressures, and fuels. Some of these methods include stagnation flow with flat or curved flames, spreading spherical flames in the combustion vessel, and flat flames stabilized on the burner. When employing any of these methods of measurement, removing the influence of flame stretch merits attention, either during the experiment or after data process.
According to the most widely accepted definition of flames, visible chemical component undergoing extremely exothermic chemical reaction occurs in a restricted zone with the evolution of heat. The flame front's measured rate of expansion in a combustion reaction is called the flame speed. Taylor et al.[1] argued that premixed or non-premixed reactants can produce flames in two different ways.
Prior to reaching the flame front in a premixed flame, the oxidizer has been mingled with the fuel. The reaction produces a thin flame front due to the availability of all reactants. Premixed flammability limits range from 0.6 to 3 for most hydrocarbon fuels. W. C. Strahle revealed that the reactants are initially separated (non-premixed) in a diffusion flame, and the reaction occurs only at the interface between the fuel and the oxidizer, when mixing and reaction occur simultaneously[2]. A flame in which the oxidizer diffuses into the fuel is another way to describe it. As a result, flame speed is limited by the rate of diffusion. Diffusion flames burn slower and produce more soot than premixed flames due to the insufficient oxidizer to complete the process. Taylor et al.[1] and Jerzembeck et al.[3] measured laminar burning velocities and Markstein lengths in spherical flames of n-heptane, iso-octane, and gasoline/air blends at engine-relevant conditions using the constant volume bomb method. At 373K, data was gathered with equivalence ratios ranging from 0.7 to 1.2 and starting pressures ranging from 10 to 25 bars. Scientists employed a dark field He-Ne laser Schlieren measurement technique and digital image processing to follow the flame front in the tank. A linear extrapolation to zero stretch was used to achieve the laminar burning velocities. Simultaneously, theoretical projections were compared to experimentally determined Markstein numbers. Saeed and Stone[4] investigated the variation of burning velocity as a function of equivalency ratio and pressure, and concluded that pressure rises, burning velocity decreases serve as the variation. S.P. Marshall et al.[5] observed LBV measurements of liquid fuels at high pressures and temperatures with combustion residuals. The burning velocity was calculated using pressure and schlieren data via a constant volume vessel (rated at 3.4MPa.) and a multi-zone model. n-heptane, iso-octane, toluene, ethylbenzene, and ethanol were tested at various starting pressures (50, 100, 200, and 400KPa), temperatures (310, 380, and 450K), and equivalency ratios (0.7-1.4), as well as studies with combustion residuals as low as 0.3.
When employing ignition system energies of (25, 45, and 65mJ) to ignite natural gas samples, Liao et al.[7] found that burning velocity increased with spark energy at a flame radius less than 6mm.
2 RESULTS AND DISCUSSIONS
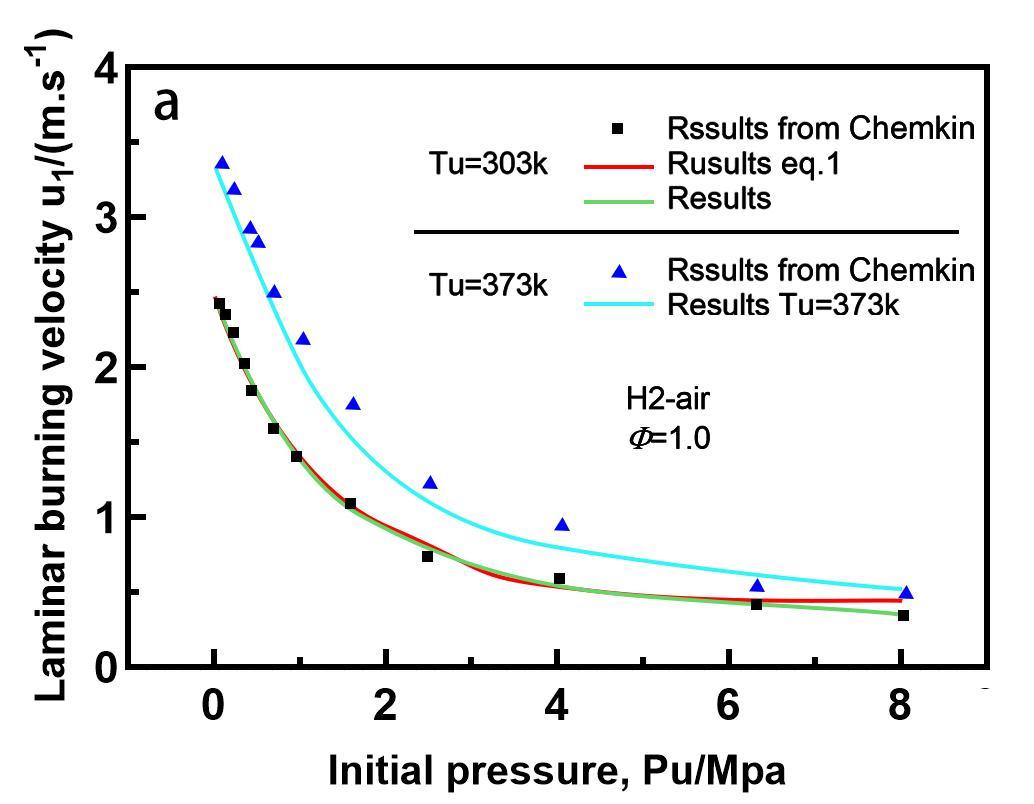
The LBV of hydrogen-air flames was demonstrated in Figure 1 at varied starting pressures and temperatures. The LBV falls exponentially when the starting pressure is raised. Hu et al.[8] investigated the LBV of hydrogen-air flames at different temperatures and pressures. As shown in the diagram, as the starting pressure rises, the LBV drops Exponentially 1. The following formulas can be used to fit a curve to pressure:
|
|
Figure 1. LBV of H2/air mixture as a function of initial pressure[8].
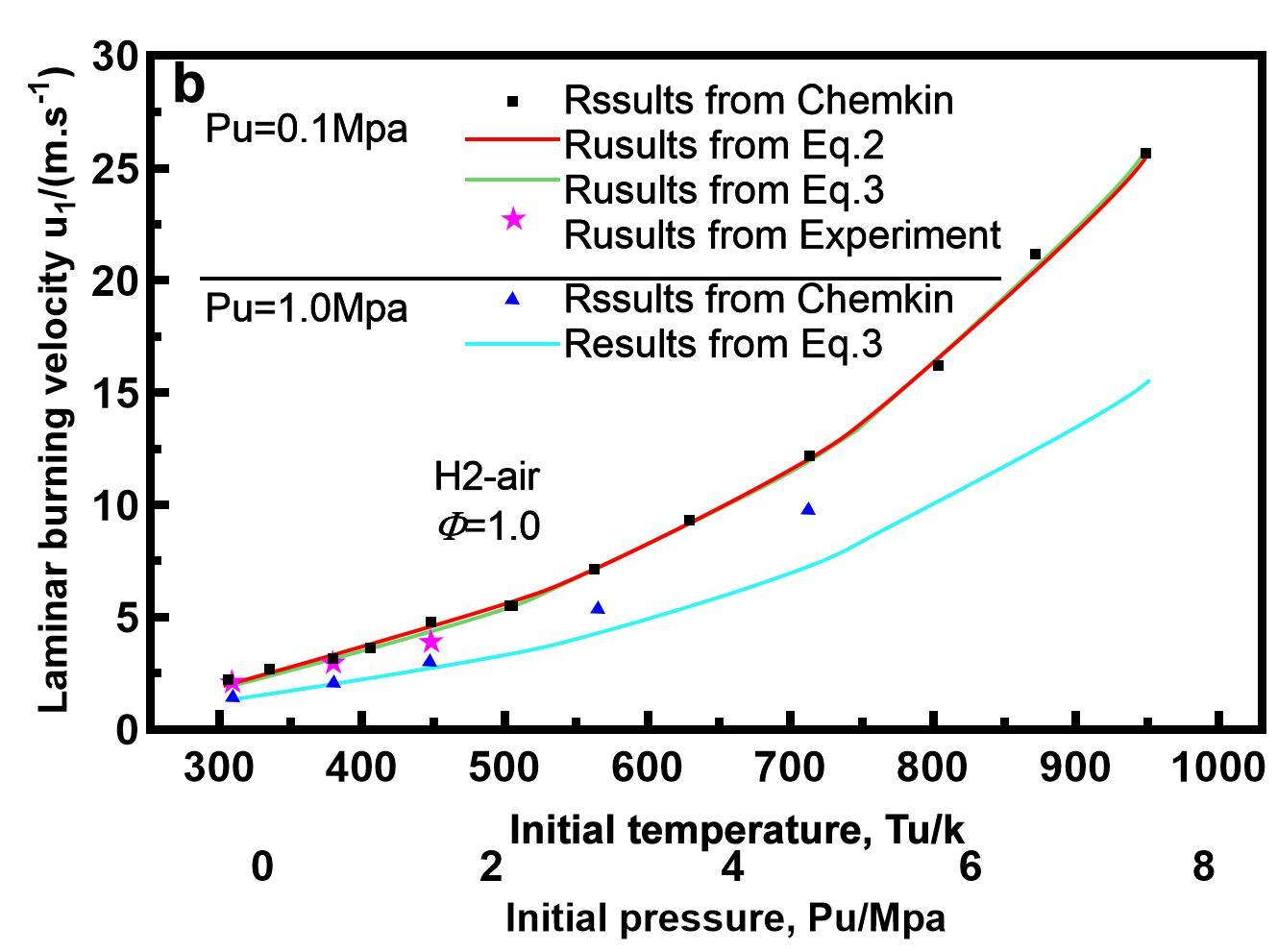
At various starting temperatures, the LBV of hydrogen-air flames was demonstrated in Figure 2. As the initial temperature rises, the LBV grows exponentially. Hu et al.[8] investigated the LBV of hydrogen-air flames at different temperatures and pressures at the beginning of their research. As seen in Figure 1, as the starting temperature rises, the LBV rises Exponentially 2. To match the curve, they presented the equation below:
|
The following empirical expression can be used to link the effects of temperature and pressure on LBV:
|
Subscript 0 is used to signify the reference condition of Tu0=303K and Pu0=0.1MPa.
The LBV for the stoichiometric mixture was discovered to be roughly 2.406m/s at the reference condition, ul0. A linear formula was suggested for the temperature and pressure exponents:
|
|
|
Figure 2. Impact of of praimary temperature on LBV of H2/air mixture[8].
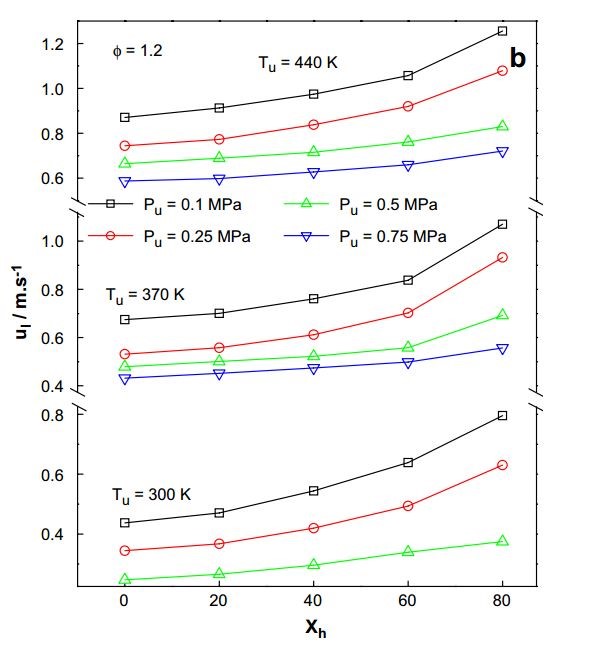
Figure 1 depicted the burning velocity of propane at different hydrogen percentages. According to the data, the LBV remains high when the beginning temperature rises. Tang et al.[9] investigated propane combustion velocity at different hydrogen concentrations and equivalency ratios. As the initial temperature rises, so does the LBV, as shown in Figure 1.
Burning velocity rises as Xh rises, and the upward trend becomes more obvious. When hydrogen is added to the activation reaction region, the amount of radicals created increases. While hydrogen has a lower volumetric heating value than propane, at stoichiometric circumstances, its fuel-air ratio is ten times that of propane. As a result, increasing the hydrogen fraction reduces the amount of air in which nitrogen plays a significant role in determining the mixture's specific heat, resulting in an increase in adiabatic temperature and reaction rate (Figure 3).
|
Figure 3. LBV of C3H8/H2/air mixture variations with changing initial temperature and pressure[9].
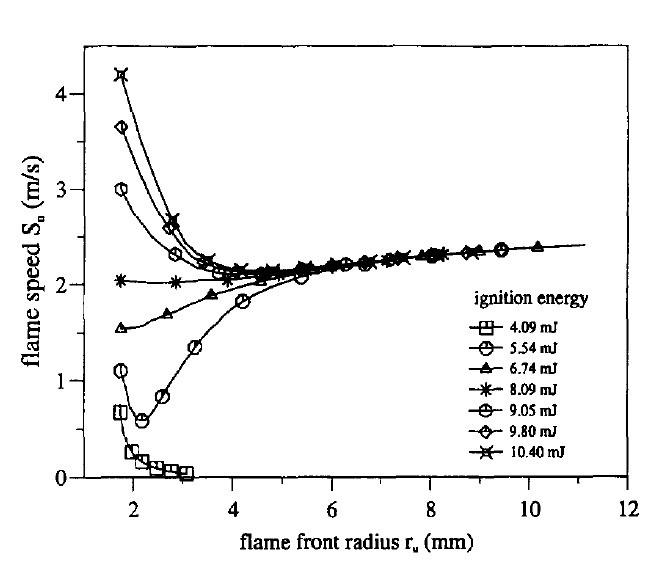
At normal pressure and temperature, the flame characteristics of natural gas-hydrogen-air in a constant volume bomb were presented in Figure 4. The spark discharge generates a shock wave, followed by a slower thermal wave. Bradley is a character in the film Bradley (nine). Laminar burning velocities were studied at different hydrogen-to-natural-gas ratios (volume fractions ranging from 0 to 100%) and equivalency ratios ranging from 0.6 to 1.4. The flame speeds become independent of the ignition energy after the flame radius exceeds a specific threshold (6mm). This energy can cause the mixture's temperature to rise, resulting in a quicker flame. For ignition energies below the minimum ignition energy, the flame speed quickly slowed to a halt.
|
Figure 4. Computations of flame speed VS. Radius for different ignition energies[10].
3 CONCLUSION
As the starting temperature rises, the rate of laminar burning increases, while as the initial pressure rises, the rate of laminar burning drops. With rising initial pressure, the onset of cellular instability advances, and Markstein lengths decrease, indicating a rise in hydrodynamic instability. Flame instability is unaffected by initial temperature. For different diluents, the product of thermal diffusion and chemical reaction terms show the same tendency as the LBV as a function of the dilution ratio. The adiabatic flame temperature plays a most crucial role on the LBV in methane-air-diluent flames, followed by thermal diffusivity. Both temperature and pressure lowered the flame thickness, yet pressure demonstrated more impact on the flame thickness than temperature. At the cost of higher turbulence intensity, a drop in flame velocity caused by exhaust gas recirculation can be partially compensated by increasing turbulence intensity.
Acknowledgments
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Author Contribution
All authors approved the final version.
Abbreviation List
LBV, Laminar burning velocity
References
[1] Crispin TS. Burning velocity and the influence of flame stretch. unpublished Ph.D. thesis. Department of Fuel and Energy University of Leeds; 1991.
[2] Strahle WC. An Introduction to Combustion Science and Technology, 1st ed. Gordon and Breach Science Publishers, 1993.
[3] Jerzembeck S, Peters N, Pepiot-Desjardins P et al. Laminar burning velocities at high pressure for primary reference fuels and gasoline: Experimental and numerical investigation. Combust Flame, 2009; 156: 292-301. DOI: 10.1016/j.combustflame.2008.11.009.
[4] Saeed K, Stone CR. The modelling of premixed laminar combustion in a closed vessel. Combust Theor Model, 2004; 8: 721.
[5] Marshall SP, Taylor S, Stone CR et al. Laminar burning velocity measurements of liquid fuels at elevated pressures and temperatures with combustion residuals. Combust Flame, 2011, 158(10): 1920-1932. DOI: 10.1016/j.combustflame.2011.02.016.
[6] Natarajan J, Lieuwen T, Seitzman J. Laminar flame speeds of H2/CO mixtures: Effect of CO2 dilution, preheat temperature, and pressure.Combust Flame, 2007; 151: 104-119. DOI: 10.1016/j.combustflame.2007.05.003.
[7] Liao SY, Jiang DM, Gao J et al. Measurements of Markstein numbers and laminar burning velocities for natural gas-air mixtures. Energ Fuel, 2004; 18: 316-326. DOI: 10.1021/ef034036z.
[8] Hu E, Huang Z, He J et al. Experimental and numerical study on laminar burning velocities and flame instabilities of hydrogen-air mixtures at elevated pressures and temperatures. Int J Hydrogen Energ, 2009; 34: 8741-8755. DOI: 10.1016/j.ijhydene.2009.08.044.
[9] Tang C, He J, Huang Z et al. Measurements of laminar burning velocities and Markstein lengths of propane–hydrogen–air mixtures at elevated pressures and temperatures. Int J Hydrogen Energ, 2008; 33: 7274-7285. DOI: 10.1016/j.ijhydene.2008.08.053.
[10] Bradley D, Hicks RA, Lawes M et al. The measurement of laminar burning velocities and Markstein numbers for iso-octane-air and iso-octane-n-heptane-air mixtures at elevated temperatures and pressures in an explosion bomb. Combust Flame, 1998; 115: 126-144. DOI: 10.1016/S0010-2180(97)00349-0.
Copyright ©2022 The Author(s). This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.





 Copyright ©
Copyright ©