Structure, Physic-chemical Characterizations and Hirshfeld Surface Analysis of [C7ON2H18]3V10O28·2H2O Compound
Regaya Ksiksi1,2*, Mohamed Faouzi Zid1, Mohsen Graia3
1Laboratory of Materials, Faculty of Sciences of Tunis, University of Tunis El Manar, Tunis, Tunisia
2High Institute of Preparatory Studies in Biologyand Geology (ISEP-BG) of Soukra, University of Carthage, Amilcar, Tunisia
3Solid State Laboratory, Faculty of Sciences of Sfax, University of Sfax, Sfax, Tunisia
*Correspondence to: Regaya Ksiksi, PhD, Associate Professor, Laboratory of Materials, Faculty of Sciences of Tunis, University of Tunis, 2092 El Manar II, Tunis 1007, Tunisia; Email: rksiksi@gmail.com
DOI: 10.53964/jmn.2022009
Abstract
Objective: Structural study and physico-chemical characterization of a new decavanadate compound.
Methods: The compound was synthesized using distilled water as solvent. By stirring and magnetic heating and by evaporation at room temperature, a good quality crystal was chosen for single crystal X-ray diffraction.
Results: A new decavanadate compound, tri[2-[2-(dimethylamino) ethyl-methylaminium]ethanol] decavanadate dehydrate, was synthesized by slow evaporation, the formula unit is composed by one decavanadate cluster, three [C7ON2H18]2+ organic cations and two water molecules. Different characterization techniques are used such as: crystal X-ray diffraction, Scanning electron microscopy with energy dispersive X-Ray analysis and thermal analysis. The X-ray structure determination revealed that the compound crystallizes in the triclinic system, space group P-1 with the cell parameters: a=10.815 (9) Å, α=112.97 (3)°, b=11.624 (4) Å, β=111.73 (7)°, c=11.840 (5) Å, γ= 96.86 (6)° and V=1195.8 (6)Å3. The cohesion of the structure is ensured by hydrogen bonds of the type O-H...O, N-H...O and van der Waals interactions. The study of the Hirshfeld surface of the decavanadate compound shows us that it is dominated by O…H/H…O (56.7%) and H…H (30.2%) type contacts.
Conclusion: A new decavanadate compound was synthesized and characterized by different physico-chemical techniques. The detailed structural study shows that the different groupings of the structure pile up in layers parallel to the planes (010). The cohesion of the structure is ensured by hydrogen bonds and van der Waals interactions.
Keywords: decavanadate, synthesis, physico-chemical characterizations, hirshfeld surface analysis
1 INTRODUCTION
Polyoxametallates have potential applications in various fields of science and technology, catalytic, magnetic and electrochemical, due to their properties such as thermal stability, selective inhibitors of an impressive variety of enzymes redox activity as well as solubility[1-3]. Among the various polyoxometalates, we have focused our attention on polyoxovanadates due to the relatively low toxicity of vanadium in biological media[4]. Vanadium acts as an inorganic cofactor in haloperoxidases and alternative azoteases, exhibits insulin-mimetic activities related to diabetes mellitus, and exhibits anticancer activities in several cell lines[5]. The decavanadate compounds have shown a strong affinity for certain kinases, phosphorylase and reverse transcriptase[6]. In previous works we have published several decavanadate structures such as: Na5.22Li0.78[V10O28]20H2O[7], [Zn(H2O)6][Zn2V10O28(H2O)10]·6H2O[8], we have also published articles which show the important biological activity of decavanadate compounds studied by Aissa et al.[9], Ksiksi et al.[10] and Louati et al[11]. The study of the antitumor activities of these compounds encouraged us to synthesize new pure decavanadate phases and to study their structures.
In this work we will study: Structure, physico-chemical characterizations and Hirshfeld surface analysis of [C7ON2H18]3V10O28·2H2O compound.
2 MATERIALS AND METHODS
2.1 Materials and Physical Measurements
Scanning electron microscope coupled to an X-ray energy dispersive analysis spectrometer is performed using an FEI Quanta 200 environmental apparatus (LEUVEN Belgium, PHILIPS/FEI). The Thermogravimetry (TG) and Differential thermal analysis (DTA) analysis were carried out using a SETERAM LabsysTM typeapparatus (France, Setaram Instrumentation), by exploring the temperature range between ambient and 300°C.
2.2 Synthesis of [C7ON2H18]3V10O28·2H2O Compound
The phase was prepared with a vanadium oxide V2O5 and 2-[2(Dimethylamino) ethyl]-methylamino] ethanol in 50mL of water with the following molar proportion 1:3. The solution is placed under magnetic stirring and with slight heating for 3h. After filtration, the mixture is brought to room temperature. After a few days, there is appearance of crystals of good quality for X-ray diffraction.
2.3 Hirshfeld Surface Study
The study of the Hirshfeld surface analysis was done using the software Crystal Explorer[12,13].
2.4 X-ray Study
A good quality orange single crystal was chosen for single crystal X-ray diffraction. An Enraf-Nonius CAD4 type automatic four-circle diffractometer[14] was used for data collection using Mo-Kα radiation (λ=0.71073 Å). The resolution of the structure was carried out by Farrugia[15]. The SHELXS-97 program[16] was used for the structure resolution and the SHELXL-2014[17] for the refinement. The absorption correction was performed by psi-scan method[18]. The figures were represented using the program Diamond[19]. The HFIX statement for fixing hydrogen atoms. The X-ray crystallographic data are given in Table 1.
Table 1. Crystallographic Characteristics, X-ray Data Collection, and Structure-Refinement Parameters for [C7ON2H18]3V10O28·2H2O Compound
Crystal Data |
|
Chemical formula |
[C7ON2H18]3V10O28·2H2O |
Formula weight Mr (g.mol−1) |
1432.13 |
Crystal system, space group |
Triclinic, P-1 |
T (K) |
298(2) |
a b c (Å) |
10.815 (9), 11.624 (4), 11.840 (5) |
α, β, γ (°) |
112.97 (3), 111.73 (4), 96.86 (6) |
V(Å3) , Z |
1195.8(6), 1 |
Radiation λ (Å) |
Mo-Kα 0.71073 |
Crystal size (mm3) |
0.16 x 0.31 x 0.55 |
μ (mm−1) |
1.989 |
F (000) |
720 |
Data collection |
|
Diffractometer |
Enraf-Nonius CAD4 |
Absorption correction |
Ψscan |
Tmin,Tmax |
0.3337, 0.7232 |
Range for data collection (°) |
2≤θ≤ 27 |
h, k, l ranges |
-1≤ h ≤13, -14≤ k ≤14, -15≤ l ≤14 |
Scan mode |
ω/2θ |
No. of measured, independent, and observed |
6018, 5181, 3298 |
[I >2σ(I)]] reflections |
|
Rint |
0.0713 |
Refinement |
|
R1 [F2 > 2 σ (F2)] |
0.077 |
wR2(F2) |
0.2241 |
S |
1.013 |
No. of parameters |
375 |
Maximum residual electron density |
1.218 |
Δρmax,(e.Å−3) |
|
Minimum residual electron density |
-1.075 |
Δρmin (e.Å−3) |
|
Notes: a, b, c, α, β, γ: cells; F (000): Structure factor; V: Volume; μ: Absorption coefficient; R: Reliability factors; T: Absorption transmission factor; wR: Weighted R factor; h,k,l: Miller Indices, S: GOOF on F2.
3 RESULTS AND DISCUSSION
3.1 Crystal Structure
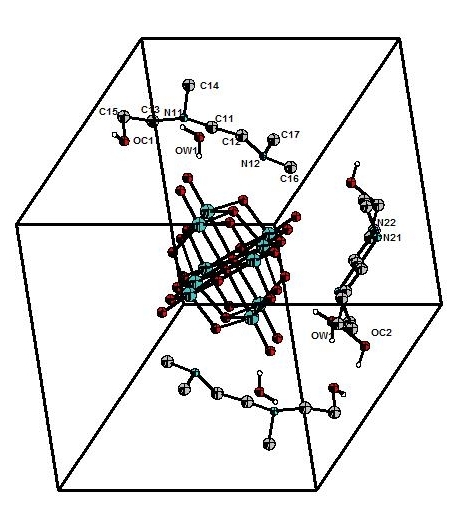
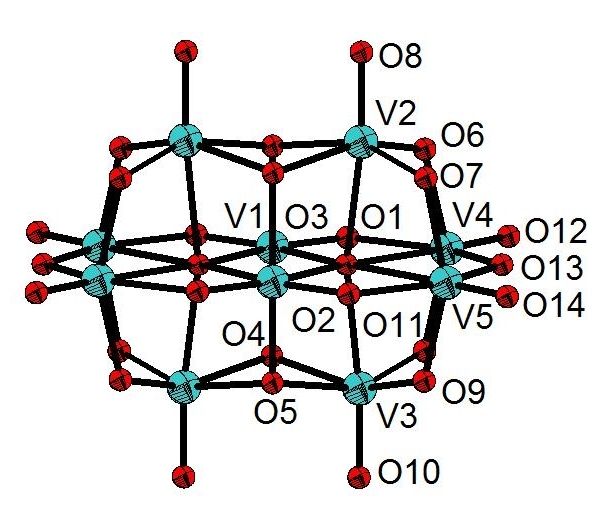
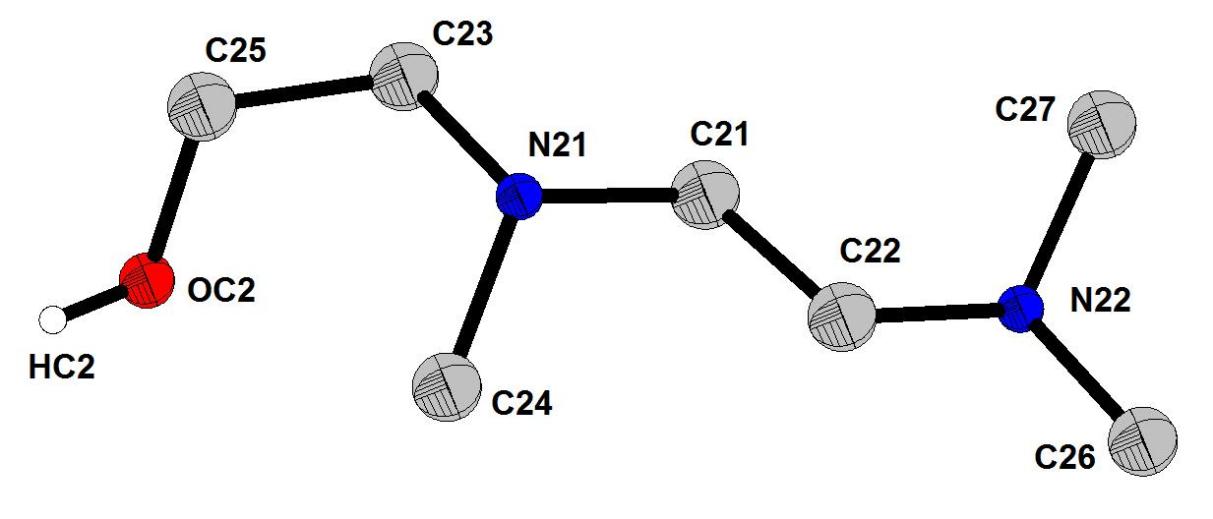
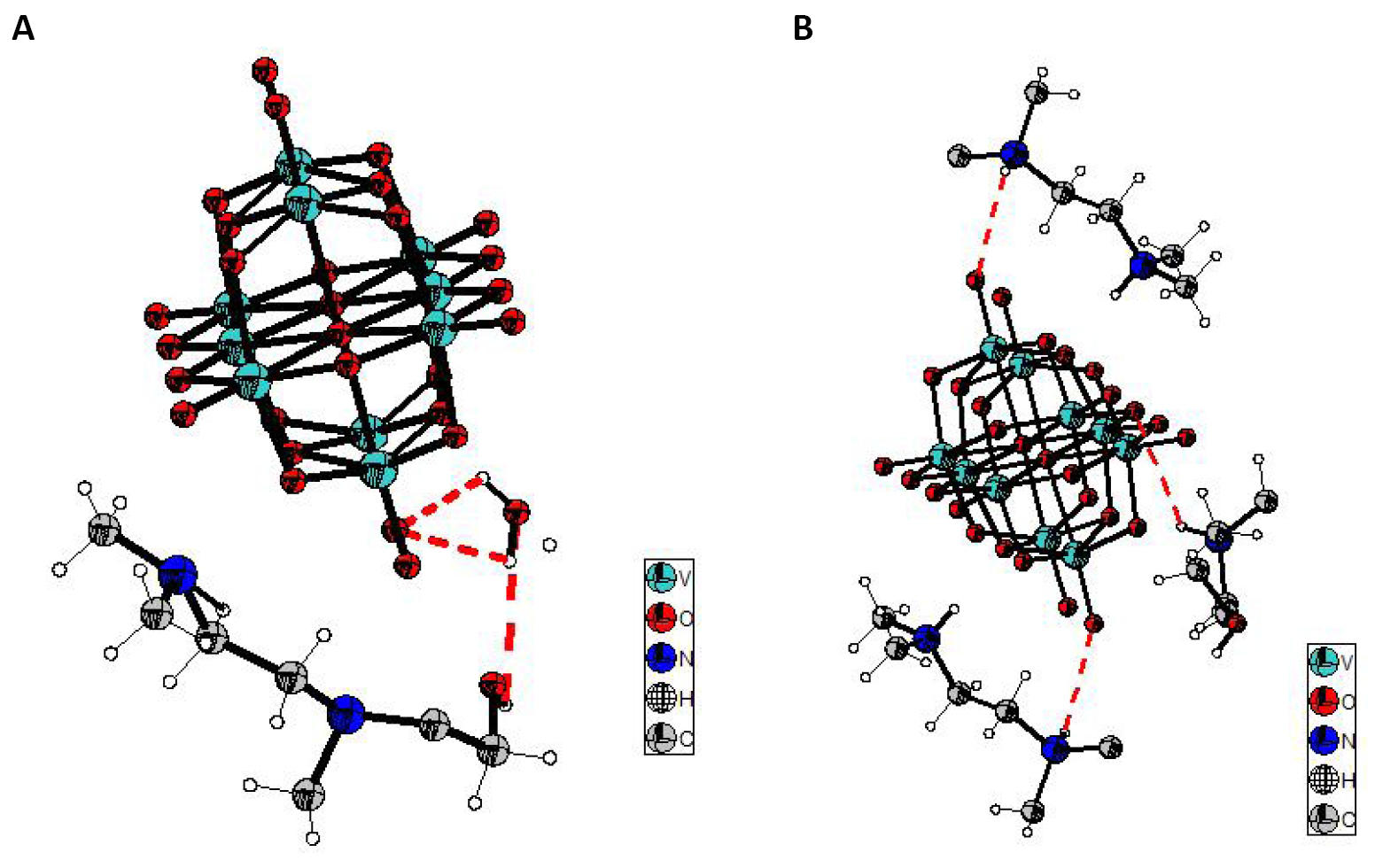
The structure of the decavanadate compound, [C7ON2H18]3V10O28·2H2O, is composed by the centrosymmetric decavanadate group [V10O28]6-, two non-centrosymmetric 2-[2-(Dimethylamino) ethyl]-methylaminium] ethanol [C7ON2H18]2+ cations, one centrosymmetric cation [C7ON2H18]2+ and two water molecules (Figure 1). The centrosymmetric cluster [V10O28]6- is formed by ten VO6 octahedra interconnected by edge sharing. There are different types of V-O distances: they are between 1.598 (4) and 1.612 (4) Å for the terminal oxygen atoms, between 1.795 (4) and 1.879 (4) Å for doubly coordinated oxygen atoms, between 1.942 (3) and 2.023 (4) Å for triply coordinated oxygen atoms and between 2.097 (4) and 2.353 (4) Å for oxygen atoms hexacoordinated (Figure 2). The distances V-V vary from 3.066 (1) to 3.119 (1) Å. The distances and angles of bond in this group are in agreement with other similar compounds found in the bibliography[9, 11].The representation of the organic cation 2-[2-(Dimethylamino) ethyl]-methylaminium] ethanol [C7ON2H18]2+ is given in Figure 3.
|
Figure 1. Formula unit of [C7ON2H18]3V10O28·2H2O compound.
|
Figure 2. Decavanadate group in [C7ON2H18]3V10O28·2H2O compound.
|
Figure 3. Representation of organic cation 2-[2-(Dimethylamino)ethyl]-methylaminium] ethanol [C7ON2H18]2+.
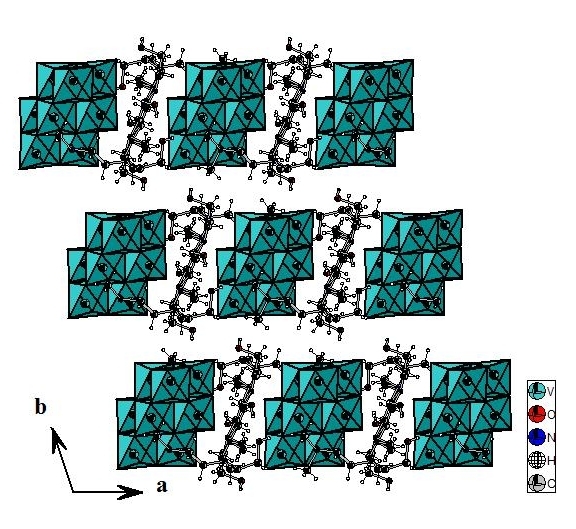
Decavanadate groups, organic cations and water molecules stack up in layers parallel to the (010) plane. Cohesion between layers is ensured by van der Waals interactions (Figure 4).
|
Figure 4. Projection along (a, b) plane of [C7ON2H18]3V10O28.2H2O compound.
In one layer, the [C7ON2H18]2+ cations and water molecules form a two-dimensional network in which the decavanadate groups are nested. The cohesion of the structure is ensured by O-H…O, N-H...O hydrogen bonds (Table 2) which engage the [C7ON2H18]2+ cations, water molecules and decavanadate groups (Figure 5).
Table 2. Hydrogen Bonds Length in (C4H7N2)6V10O28·3H2O [C7ON2H18]3V10O28·2H2O Compound
D-H···A |
D (D-H) (Å) |
d (H···A) (Å) |
d (D···A) (Å) |
<D-H···A> (°) |
N11-H11...OW1i |
0.91 |
2.148 |
2.905 |
139.97 |
N11-H11...O8 |
0.91 |
2.522 |
3.015 |
114.52 |
N12-H12...O5 |
0.91 |
1.758 |
2.665 |
173.49 |
OC1-HC1...O6i |
0.82 |
1.892 |
2.71 |
175.24 |
N21-H21...O4ii |
0.91 |
1.705 |
2.606 |
169.74 |
N22-H22...O4iii |
0.91 |
1.865 |
2.77 |
172.97 |
OW1-HW1...O7 |
0.824 |
2.158 |
2.947 |
160.14 |
OW1-HW1 O8 |
0.824 |
2.574 |
3.025 |
115.81 |
OW1-HW2…OC1 |
0.815 |
2.025 |
2.836 |
173.19 |
Notes: Symmetry codes: i: -x, -y+1, -z; ii : x+1, y, z ; iii : -x+1, -y+1, -z+1
|
Figure 5. Cohesion of the structure by hydrogen bonds. A: O-H…O hydrogen bonds; B: N-H...O hydrogen bonds.
3.2 Comparison of the Structure Studied with (NH4)4Li2V10O28·10H2O Compound
The projection according to c of the structure studied in this work and of the structure (NH4)4Li2V10O28·10H2O[10] shows that the decavanadate groups, organic cations and water molecules stack up in layers parallel to the (010) plane and in the decavanadate structure of ammonium and lithium the decavanadate groups and the Li2(H2O)102+ dimers stack up in layers parallel to the (010) plane. The cohesion of the two structures is ensured by hydrogen bonds and van der Waals interactions.
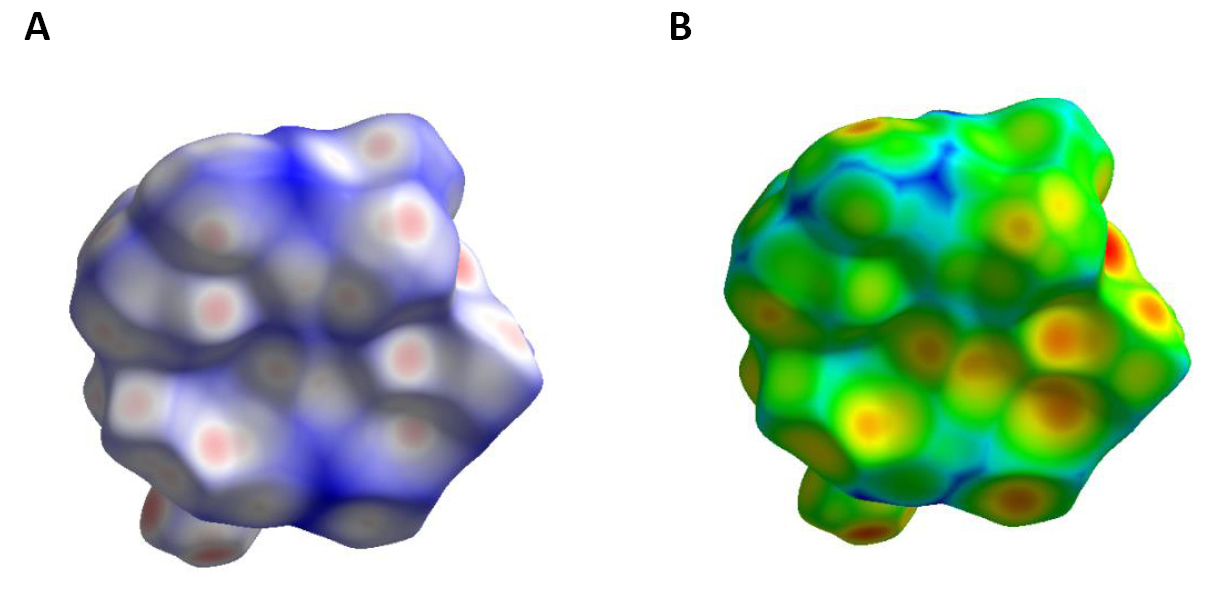
The Hirshfeld area of [C7ON2H18]3V10O28·2H2O compound was determined from the asymmetric unit of the compound. The Hirshfeld surfaces of [C7ON2H18]3V10O28·2H2O compound is studied in dnorm (normalized contact distance) and di(The both contact distances between nearest atoms present inside)(Figure 6). Figure 6 shows us the existence of O-H…O hydrogen bonds (red spots) they correspond to O⋯H/H⋯O bonds. H...H type interactions (white areas). Areas where neighboring atoms are too far apart to interact with each other (bluish areas).
|
Figure 6. Hirshfeld surface mapped over dnorm (A) and di (B) of [C7ON2H18]3V10O28.2H2O compound.
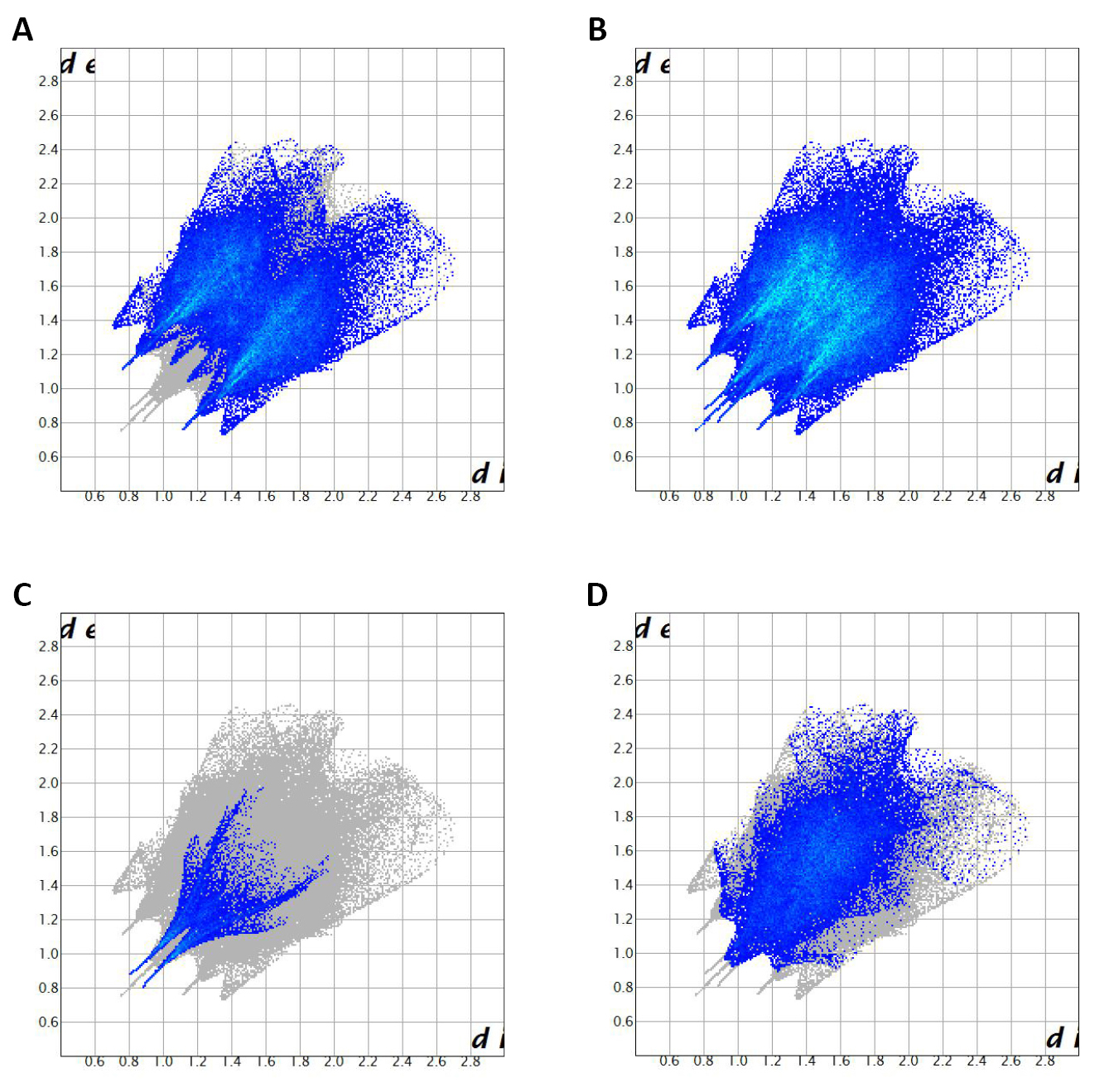
The contribution of intermolecular contacts to Hirshfeld surfaces in the structure of the compound [C7ON2H18]3V10O28·2H2O reveals that the structure is dominated by O…H/H…O (56.7%), H…H (30.2%), V...O/O...V (10.9%) (Figure 7). The comparison of the interactions O...H/ H...O, H...H, V...O/O...V between the studied compound and (NH4)4Li2V10O28·10H2O compound, shows a considerable increase in the H…H interactions in the structure of the compound studied (Table S1)[10] (30.2℅ relative to 18.4%). This increase can be the cause of the increase in the mesh parameters as well as the volume in our compound.
|
Figure 7. 2D fingerprint plots of the studied compound resolved into all contacts (A), O…H/H…O (56.7 ℅) (B), H…H (30.2 ℅) (C), and O…V/V…O (7.1 ℅) (D)contacts.
3.3 EDX (Energy Dispersive X-ray) Analysis
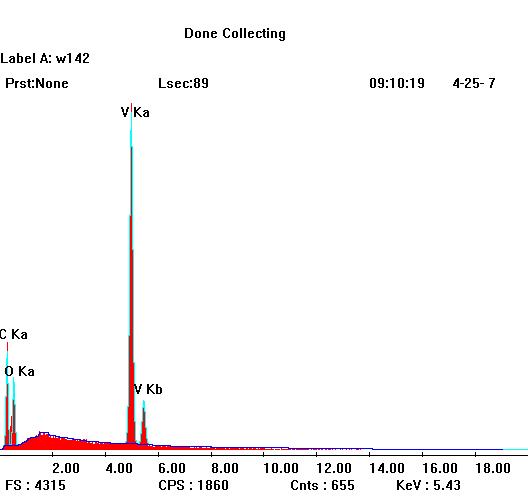
The EDX analysis of [C7ON2H18]3V10O28·2H2O compound is given in Figure 8. This figure shows the presence of the following chemical elements: vanadium, oxygen and carbon.
|
Figure 8. EDX of [C7ON2H18]3V10O28.2H2O compound.
3.4 DTA and TG Curve of [C7ON2H18]3V10O28·2H2O Compound
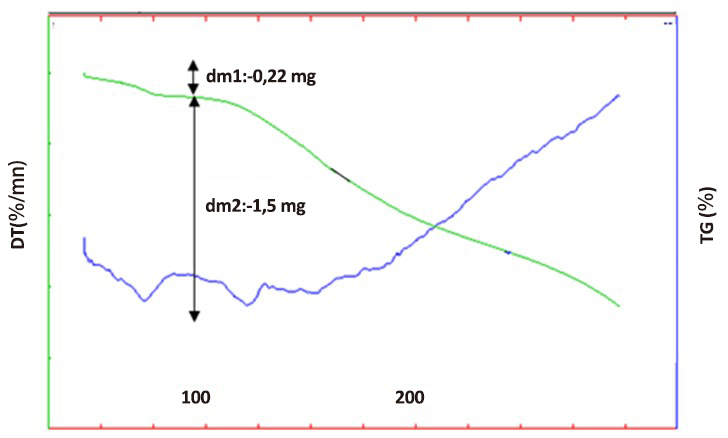
The DTA-TG analysis of this compound was carried out on a sample with a mass of 9.01mg placed in a platinum crucible.
The TG curve of [C7ON2H18]3V10O28·2H2O compound (Figure 9) reveals that the departure of water molecules begins around 60°C and ends around 80°C. The loss percentage is 2.4%. This is reflected on the DTA curve by an endothermic peak around 70°C. Theoretically, the departure of two water molecules would correspond to a loss percentage of 2.5%.
The second mass loss on the TG curve of the order of 16.7% concerns the departure of CO2, it begins around 110°C. The theoretical percentage of CO2 departure is 16.14%.
|
Figure 9. DTA and TG of [C7ON2H18]3V10O28.2H2O compound.
4 CONCLUSION
A new decavanadate compound was synthesized and characterized by different physico-chemical techniques: [C7ON2H18]3V10O28·2H2O. [C7ON2H18]2+ cations, water molecules and decavanadate groups stack in layers parallel to the (010) plane. The cohesion of the structure is ensured by hydrogen bonds and van der Waals interactions.
The EDX analysis of [C7ON2H18]3V10O28·2H2O confirms the presence of the following chemical elements: vanadium, nitrogen, oxygen and carbon. The thermal analysis informs us about the stability of the studied compound.
In perspective we will study the antitumor activity of [C7ON2H18]3V10O28·2H2O compound against the three cell lines: U87, IGR39 and MDA-MB-231 and the compared with the antitumor activity of (NH4)4Li2V10O28·10H2O compound that we have already compared the two structures.
Acknowledgements
Financial support from the Ministry of Higher Education and Scientific Research of Tunisia is gratefully acknowledged. This work is done as part of a federated research project under the code PRF2019-D3P2.
Conflicts of Interest
There was no conflict of interest between the authors of this article.
Author Contribution
Ksiksi R was responsible for data curtion, writing, and original-draft. Graia M was responsible for review. Zid MF was responsible for supervision.
Abbreviation List
a, b, c, α, β, γ, cells
di, The both contact distances between nearest atoms present inside.
dnorm, Normalized contact distance
DTA, Differential thermal analysis
EDX, Energy dispersive X-ray
F(000), Number of electrons per cell
TG, Thermogravimetry
References
[1] Avila PF, Ripplinger TJ, Kemper DJ et al. Features of vibrational and electronic structures of decavanadate revealed by resonance raman spectroscopy and density functional theory. J Phys Chem Lett, 2019; 10: 6032-6037. DOI: 10.1021/acs.jpclett.9b02362
[2] Arefian M, Mirzaei M, Eshtiagh-Hosseini H et al. A survey of the different roles of polyoxometalates in their interaction with amino acids, peptides and proteins. Dalton Trans, 2017; 46: 6812-6829. DOI: 10.1039/C7DT00894E
[3] Judd DA, Nettles JH, Nevins N et al. Polyoxometalate HIV-1 protease inhibitors: A new mode of protease inhibition. J Am Chem Soc, 2001; 123: 886-897. DOI: 10.1021/ja001809e
[4] Treviño S, Díaz A, Sánchez-Lara E et al. Vanadium in biological action: Chemical, pharmacological aspects, and metabolic implications in diabetes mellitus. Biol Trace Elem Res, 2019; 188: 68-98. DOI: 10.1007/s12011-018-1540-6
[5] Srivastava AK, Mehdi MZ. Insulino-mimetic and anti-diabetic effects of vanadium compounds. Diabet Med, 2005; 22: 2-13. DOI: 10.1111/j.1464-5491.2004.01381.x
[6] Zhai F, Wang X, Li D, et al. Synthesis and biological evaluation of decavanadate Na4Co(H2O)6V10O28·18H2O. Biomed Pharmacother, 2009; 63: 51-55. DOI: 10.1016/j.biopha.2008.01.006
[7] Ksiksi R, Graia M, Jouini T. Sel mixte de lithium et de sodium eicosahydrate, Na5.22Li0.78[V10O28]·20H2O. Acta Crystallogr Sect E Struct Rep Online, 2005; 61: i177-i179. DOI: 10.1107/S160053680502060X
[8] Graia M, Ksiksi R, Driss A. Preparation and characterization of [Zn(H2O)6][Zn2V10O28(H2O)10]·6H2O single crystals with a novel metallic heteropolyoxoanion. J Chem Crystallogr, 2008; 38: 855-859. DOI: 10.1007/s10870-008-9410-2
[9] Aissa T, Ksiksi R, Elbini-Dhouib I et al. Synthesis of a new vanadium complex (V), hexa [4-methylimidazolium] decavanadate trihydrate (C4H7N2)6V10O28·3H2O: Physico-chemical and biological characterizations. J Mol Struct, 2021; 1236: 130331. DOI: 10.1016/j.molstruc.2021.130331
[10] Ksiksi R, Abdelkafi-Koubaa Z, Mlayah-Bellalouna S et al. Synthesis, structural characterization and antitumoral activity of (NH4)4Li2V10O28·10H2O compound. J Mol Struct, 2021; 1229: 129492. DOI: 10.1016/j.molstruc.2020.129492
[11] Louati M, Ksiksi R, Elbini-Dhouib I et al. Synthesis, structure and characterization of a novel decavanadate, Mg(H2O)6(C4N2H7)4V10O28·4H2O, with a potential antitumor activity. J Mol Struct, 2021; 1242: 130711. DOI: 10.1016/j.molstruc.2021.130711
[12] Wolff SK, Grimwood DJ, McKinnon JJ et al. Crystal explorer. University of Western Australia: Perth, Australia, 2012.
[13] Hirshfeld FL. Bonded-atom fragments for describing molecular charge densities. Theor Chim Acta, 1977; 44: 129-138. DOI: 10.1007/BF00549096
[14] Harms K, Wocadlo S. XCAD4. University of Marburg Germany: Marburg, Germany, 1995.
[15] Farrugia LJ. WinGX and ORTEP for windows. J Appl Crystallogr, 2012; 45: 849-854. DOI: 10.1107/S0021889812029111
[16] Sheldrick G.M. A short history of SHELX. Acta Crystallogr A, 2008; 64: 112-122. DOI: 10.1107/S0108767307043930
[17] Sheldrick GM. Crystal structure refinement with SHELXL. Acta Crystallogr C, 2015; 71: 3-8. DOI: 10.1107/S2053229614024218
[18] North ACT, Phillips DC, Mathews FS. A semi-empirical method of absorption correction. Acta Crystallogr A, 1968; 24: 351-359. DOI: 10.1107/S0567739468000707
[19] Brandenburg K. DIAMOND Version 2.1e. Crystal Impact GbR: Bonn, Germany, 2001.
Copyright © 2022 The Author(s). This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.





 Copyright ©
Copyright ©