The Role of Advanced Imaging in Neurosurgical Diagnosis
Kyril L Cole1, Matthew C Findlay1, Mrinmoy Kundu2, Chase Johansen3, Cameron Rawanduzy4, Brandon Lucke-Wold5*
1School of Medicine, University of Utah, Salt Lake City, UT, USA
2Institute of Medical Sciences & Sum Hospital, Bhubaneswar, India
3Albany Medical College, Albany, NY, USA
4Department of Neurosurgery, University of Utah, Salt Lake City, UT, USA
5Department of Neurosurgery, University of Florida, Gainesville, FL, USA
*Correspondence to: Brandon Lucke-Wold, PhD, MCTS, Department of Neurosurgery, University of Florida, 2046 NE Waldo Rd Ste 1150, Gainesville, FL 32609, USA; Email: Brandon.Lucke-Wold@neurosurgery.ufl.edu
Abstract
Neurosurgery as a specialty has developed at a rapid pace as a result of the continual advancements in neuroimaging modalities. With more sophisticated imaging options available to the modern neurosurgeon, diagnoses become more accurate and at a faster rate, allowing for greater surgical planning and precision. Herein, the authors review the current heavily used imaging modalities within neurosurgery, weighing their strengths and weaknesses, and provide a look into new advances and imaging options within the field. Of the many imaging modalities currently available to the practicing neurosurgeon, magnetic resonance imaging (MRI), computed tomography, positron emission tomography, and ultrasonography are used most heavily within the field for appropriate diagnosis of neuropathologies in question. For each, their strengths are weighed regarding appropriate capabilities in accurate diagnosis of cranial or spinal lesions. Reasoning for choosing one over the other for various pathologies is also reviewed. Current limitations of each is also assessed, providing insight for possible improvement for each. New advancements in imaging options are subsequently reviewed for best uses within neurosurgery, including the new utilization of FIESTA sequencing, glymphatic mapping, black-blood MRI, and functional MRI. The specialty of neurosurgery will continue to heavily rely on improvements within imaging options available for improved diagnosis and greater surgical outcomes for the patients treated. The synthesis of techniques provided herein may provide meaningful guidance for neurosurgeons in effectively diagnosing neurological pathologies while also helping guide future efforts in neuroimaging developments.
Keywords: advanced imaging, neurosurgery, magnetic resonance imaging, computed tomography, ultrasound, positron emission tomography
1 INTRODUCTION
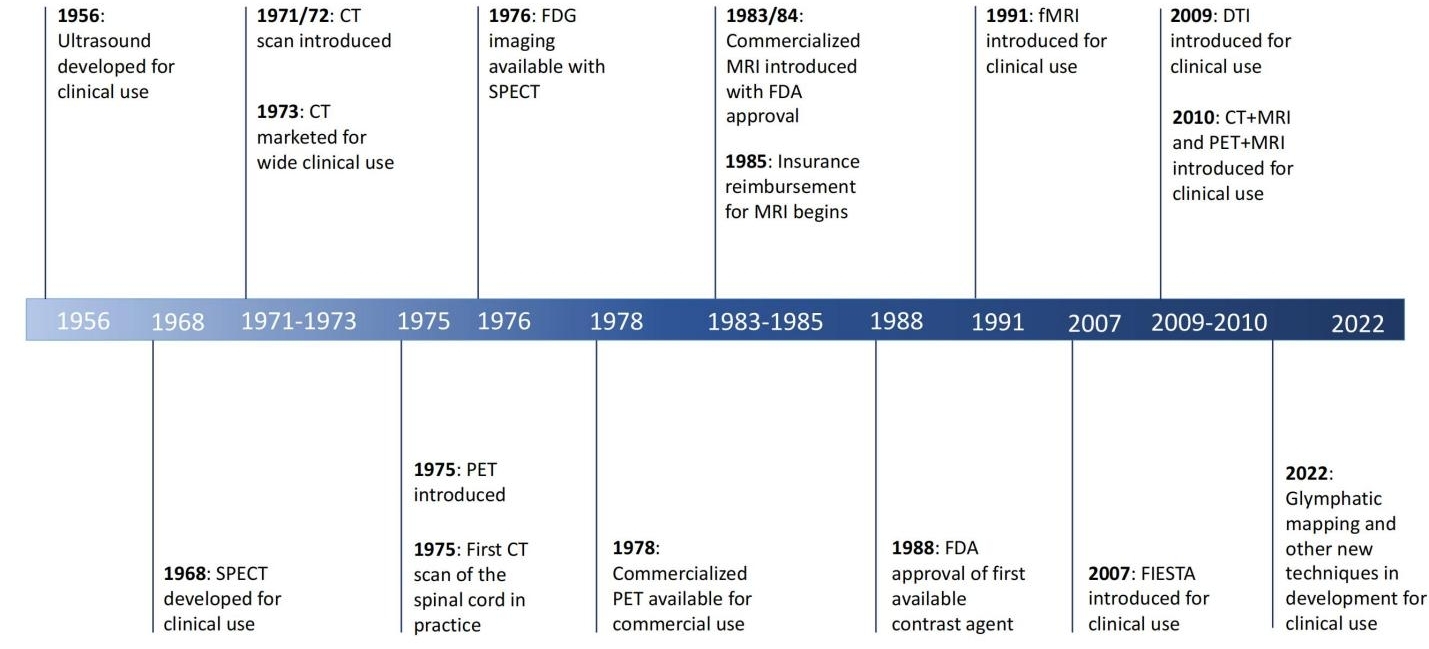
At the turn of the twentieth century, the ability to localize a neurologic lesion and determine the best operative approach relied solely on the surgeon's brain and clinical reasoning alone[1]. Since then, significant advancements have been made at an astounding pace, enhancing the technology and tools available to the modern neurosurgeon (Figure 1). Among these, advancements in neuroimaging have been demonstrated to be of particular importance, providing neurosurgeons with a greater wealth of information to inform diagnosis and subsequent management, selecting the appropriate patients and surgical targets, as well as optimal surgical approaches.2 Improvements in neuroimaging options have also been instrumental in improving patient care and reducing the morbidity and mortality from neurosurgical procedures while allowing for more reliable diagnoses and surgical decision-making within the specialty[2]. Similarly, the speed at which a neurosurgeon can now accurately diagnose and treat pathology continues to increase with new advancements in imaging modalities.
|
Figure 1. Timeline of imaging modality development and use in the neurosurgical specialty.
With these imaging advancements, the challenge thus becomes the appropriate utilization of the available imaging modalities, as needed, to produce the most effective efforts for patients. Herein, we summarize the current roles and advancements of heavily used imaging techniques in neurosurgery, including magnetic resonance imaging (MRI), computed tomography (CT), positron emission tomography (PET), and ultrasound. We also discuss emerging imaging modalities available to neurosurgeons for greater diagnostic capabilities, highlighting the future of imaging in neurosurgery.
2 MRI
MRI is an invaluable core tool in diagnosing cranial and spinal pathologies[3]. Considering the significant risk imposed with the biopsy of pathological tissue within these critical regions, current standards of care for intracranial/spinal disease involve a conventional MRI with the basic T1, T2, and FLAIR sequences[4]. These sequences, their merits, and pathologies well-visualized therein are discussed below and in Table 1.
Table 1. Common Neuropathologies Diagnosed within Neurosurgery by MRI
MRI Modality |
Cranial |
Spine |
T1 pre-contrast |
β-amyloid deposits |
Intraspinal lesions |
|
Vascular malformations |
Fatty marrow changes |
|
Cerebral venous thrombosis |
Vertebral body anatomy |
|
Metastatic disease |
Infection |
|
Intracranial lesions |
|
|
Mineral-laden structures |
|
T1-post contrast |
Intracranial lesions |
Vascular malformation |
|
Ischemic changes |
Metastatic lesions |
|
Demyelination |
Infection |
|
Radionecrosis |
|
|
Tumor recurrence |
|
T2 |
Vascular malformations |
Disc disease |
|
Intertumoral hemorrhage |
Nerve root compression |
|
Thrombosed aneurysm |
Stenosis |
FLAIR |
Multiple sclerosis |
Multiple sclerosis |
|
Superficial lesions |
Pathology near CSF |
|
Pathology near CSF |
|
Notes: MRI, Magnetic resonance imaging; FLAIR, Fluid-attenuated inversion recovery; CSF, Cerebral spinal fluid.
2.1 T1 Pre-contrast
MRI imaging operates by administering a strong radiofrequency current into the body. Upon cessation of the radiofrequency field, the disequilibrated nuclei dissipate energy as they relax back to their preexcitation net magnetization vector[5]. Some tissues dissipate this energy quicker than others, and T1 imaging measures those differences in relaxation times between tissues[5]. For example, fat tissue reverts quickly to its preexcitation magnetization and is thus hyperintense in T1-weighted images[6]. Thus, the brain's white matter is brighter than gray matter on T1, and fluid is completely hypointense. Considering most intracranial pathologies are highly vascularized, they often present hypointense in T1-weighted imaging[7]. However, pathologies which include elevated concentrations of methemoglobin, melanin, lipid, protein, calcium, iron, copper, and manganese, have a shorter relaxation time and are therefore hyperintense[7]. Pathologies with concentrated methemoglobin include β-amyloid deposits, cavernous malformations, and cerebral venous thrombosis[7]. Melanin-containing lesions include metastatic melanoma, and lipid-dense lesions include lipomas, teratomas, dermoid cysts, and lipomatous ependymomas[7]. Protein-containing lesions such as colloid cysts, Rathke cleft cysts, and ectopic pituitary gland tissue may likewise be independently hyperdense, as will mineral-containing lesions[7].
Within the spine, non-contrast T1 imaging is commonly used in isolation or compounded with other MRI sequences, such as short tau inversion recovery, to assess bone marrow abnormalities[8]. For example, fatty marrow, Paget disease, bone marrow hemorrhage, melanoma, and other post-inflammatory focal marrow atrophy are all well-detected via T1-weighted non-contrast imaging.
2.2 T1 Post-contrast
Gadolinium-based contrast agents are commonly used to enhance brain or spine MRI imaging[9]. These contrast agents are composed of paramagnetic Gadolinium compounds, increasing MRI signal intensity by quickening the T1 relaxation rate and providing additional visual disparity between tissue types. Typical uses of T1 contrast imaging include the detection of inflammatory lesions, imaging of tumors, demyelination, and ischemic changes[7]. Furthermore, some contrast agents will bind specifically to specific tumor markers, which is particularly useful in visualizing intracranial tumor borders[10]. Radionecrosis following stereotactic radiosurgery and evidence of tumor recurrence is also often assessed using T1-post contrast[11].
Within the spine, T1-weighted post-gadolinium imaging provides enhanced visualization of enhancing primary or metastatic lesions within the spine and is otherwise helpful for spinal infection and vascular malformation detection[9,12]. However, some pathologies-especially those without increased vasculature-will persist as hypodense, despite the addition of contrast.
2.3 T2
As one of the basic pulse sequences of MRI, T2 is helpful as a broad-brush approach that is sensitive to many pathological states. In contrast to T1 imaging, fat tissue will be only moderately bright, whereas fluid will be hyperintense. Thus, intracranial pathology will likely be hyperintense upon T2 due to vascularization, and white matter will be darker than gray[13]. This can increase visualization of intertumoral hemorrhage[14]. Other pathologies of a vascular nature, such as cerebral hemorrhage, thrombosed aneurysm, arteriovenous malformation, cavernoma, and microhemorrhage, are also seen in T2-weighted images[14]. Unfortunately, T2 is limited by the signal produced by CSF with partial volume effects and flow artifacts which significantly inhibit periventricular and subcortical brain region evaluation[15]. Within the spine, T2-weighted imaging is helpful in the detection and diagnosis of stenosis, nerve root compression, disc disease, or infection[16].
2.4 FLAIR
More recently developed, fluid-attenuated inversion recovery (FLAIR) has emerged as an adjunct or replacement sequence for traditional T2-weighted imaging and is more informative than T1 with contrast[17]. Producing strong T2 weighting while suppressing signals from cerebrospinal fluid, FLAIR allows strong visualization of lesions, such as cerebral cortical tumors, which lie close to CSF[18,19]. While FLAIR is particularly effective in evaluating supratentorial/superficial brain lesions, it has less diagnostic power for pathology in the posterior fossa or spinal cord[18,20]. However, due to the enhanced visualization of white matter, FLAIR of the head and spine is particularly effective in diagnosing multiple sclerosis[20-22].
2.5 Additional Sequences
Numerous additional MRI sequences have been developed and have their advantages within clinical practice. Particularly prevalent sequences include diffusion-weighted imaging and apparent diffusion coefficient sequences, which are often used to assess response to treatment and disease progression or detection of stroke or differentiation of brain tumors, respectively[23,24]. Additional MRI imaging sequences are being consistently introduced, continually improving spatial resolution and signal contrast[25]. Thus, there is likely more evolution to come regarding sequence choice in the assessment of intracranial and spinal disease with MRI imaging.
3 CT
Developed in the 1970s, CT has numerous applications within many areas of medicine; however, it is especially invaluable within neuropathological settings. This noninvasive diagnostic modality uses repeated X-rays and computer algorithms to produce images of body tissues. Compared with MRI, which records various hyperdensities per tissue as dependent upon the MRI sequence, CT is unidimensional as tissues with increased radiodensity will always present hyperdense. While MRI is considered superior to CT in the workup for intracranial abnormalities, CT is more commonly performed due to its widespread availability in acute care settings, shorter exam, and less expensive cost[26,27]. CT is also preferred over MRI to assess pathology involving the bony structures of the head and spine, such as for pathologic or traumatic fractures[26]. Intracranial pathologies commonly evaluated via CT and its variations include bone abnormalities, brain tumors, fluid collection, hemorrhage, hydrocephalus, stroke, trauma, and epilepsy[28]. Within the spine, CT offers excellent visualization of vertebral bodies, intervertebral discs, structural anomalies, and vascular malformations within the spine[29]. Soft spine tissues can also be enhanced on CT imaging with contrast[30]. A broad overview of the clinical uses of CT imaging in neurosurgery can be seen in Table 2.
Table 2. Common Neuropathologies Diagnosed within Neurosurgery by CT
CT Modality |
Cranial |
Spine |
|
General CT |
Osseus pathologies/trauma |
Vertebral body integrity |
|
|
Primary/secondary tumors |
Disc disease |
|
|
Fluid collection |
Structural anomalies |
|
|
Hemorrhage |
Osseus trauma |
|
|
Stroke |
|
|
|
Infection |
|
|
CT Angiography |
Intracranial vasculature integrity |
Vascular malformations |
|
|
Aneurysms |
Carotid stenosis |
|
|
Intracranial hemorrhage |
Anterior spinal artery syndrome |
|
|
Brain death assessment |
Carotid artery dissection |
|
Perfusion CT |
Tissue perfusion status |
|
|
|
Blood-brain barrier integrity |
|
|
|
Acute stroke |
|
|
|
Thrombolytic injury |
|
|
|
Vasospasm |
|
|
|
Outcomes of neuroendovascular therapies |
|
|
Notes: CT, Computed tomography.
3.1 CT Angiography (CTA)
A specialized variant of CT is CTA which augments traditional CT with intravenous contrast to produce images of blood vessels and tissues. Due to its ability to clearly track the course of blood products, CTA is often used as the first-line assessment of intracranial vasculature integrity, aneurysms, and hemorrhage[31]. Additionally, CTA may be used as radiographic support for diagnosing brain death following catastrophic brain injury[32]. The image reconstruction in CTA has developed to a diagnostic quality similar to MRI and can be used as a replacement for patients with metallic implants or other conditions which make them unfit for an MRI scan[33].
3.2 Perfusion CT
Like CTA, perfusion CT employs intravenous contrast to evaluate intracranial vasculature. However, CT perfusion is specially designed to test tissue perfusion status and is particularly useful in assessing the blood-brain barrier, acute stroke, thrombolytic injury, vasospasm, and the outcome of neuroendovascular therapies[34]. One of the particular advantages of perfusion CT is its ability to identify potentially salvageable tissue in the setting of acute ischemic stroke and can therefore predict the effectiveness of thrombolytic interventions and inform long-term outcomes following treatment[35].
3.3 Contraindications
It warrants observation that CT carries uniquely elevated risks compared to other imaging modalities as it imparts comparatively large doses of radiation. Such high radiation doses and the associated risks may preclude CT imaging within certain contexts, and the benefits of imaging have to be weighed against the dangers of radiation and intravenous contrast. For example, high-dose radiation in pregnant patients, particularly in the first trimester, has been associated with fetal developmental abnormalities[36]. Thus, CT imaging in pregnant patients must only be performed in critical situations and after carefully considering the risks. Additionally, the radiation of CT imaging use has been linked to the development of hematological and solid tumor cancers[37]. Thankfully, newer CT imaging techniques are being developed which use iterative reconstruction algorithms to acquire diagnostic-quality images while requiring diminished radiation doses for image acquisition[38].
Other adverse effects of CT imaging have also been reported due to the contrast agents used. Therefore, CT may likewise be contraindicated among patients with conditions such as allergies to the contrast agents, hyperthyroidism, pheochromocytoma, myasthenia gravis, metformin use, and chronic or acutely worsening renal disease[36].
4 PET
PET is a modern imaging technology often used to investigate neurological pathologies. It involves the intravenous administration of radioactive pharmaceutical compounds, which are systemically distributed and metabolized by cells. PET identifies metabolism variation by tissue types based on three-dimensional mapping of positron-emitting pharmaceuticals[39]. For neurosurgeons, PET is often used to create diagnostic images of regional cerebral functions such as blood flow, metabolism, and the binding of specific receptors[40]. Concomitantly, because of basic metabolic differences in neoplastic and non-neoplastic cells, PET is often used to discern cancerous masses from normal or inflamed tissue[41]. Within cancerous cells, radiolabeled FDG (fluorodeoxyglucose) is converted to FDG-6, which accumulates and undergoes no further metabolism[42]. This accumulation of radioactive compound can be quantified and used for a diagnosis.
Moreover, PET sometimes bears the capacity to inform the process of tumor classification for investigating neurosurgeons[43]. Aside from cases of neoplasia, PET is utilized to evaluate neurodegenerative diseases such as idiopathic normal pressure hydrocephalus and Parkinson’s disease[44,45]. Additionally, its use extends to inflammatory diseases, such as Rasmussen's encephalitis and refractory epilepsy[46-48]. PET has applications beyond cranial imaging, as well. Some studies suggest that it might predict operative outcomes in cases of cervical spine stenosis[49]. Others implicate its possible combination with CT images in diagnosing surgical site spine infections when other imaging modalities prove inadequate[50,51]. As with intracranial tumors, masses of the spine are also regularly analyzed by way of PET. Of particular note, PET is a well-established imaging technique for investigating applications in epilepsy, neuro-oncology, and cerebrovascular disease, offering physiological data about brain metabolism (Table 3).
Table 3. Common Neuropathologies Diagnosed within Neurosurgery by PET
PET Modality |
Cranial |
Spine |
Radiolabeled FDG |
Regional function assessment |
Cervical spine stenosis |
|
Metabolic demand assessment |
Surgical site infections |
|
Regional traumatic inflammation |
All primary tumors of the spine |
|
Rasmussen’s encephalitis |
Spine metastases |
|
Refractory epilepsy |
Spinal cord infections |
|
Idiopathic normal pressure hydrocephalus |
Chronic osteomyelitis |
|
Parkinson’s disease |
Spondylodiscitis |
|
All primary tumors of the cranium |
|
|
Glioblastoma |
|
|
Meningioma |
|
|
Brain metastases |
|
|
Cerebrovascular disease |
|
Notes: PET, Positron emission tomography; FDG, Fluorodeoxyglucose.
4.1 Applications
Epilepsy: Blood flow, metabolism, transport rates, the production of protein and DNA, and receptor density are just a few of the different physiological activities that may be seen and quantified using PET scans of the brain for diagnosis of epilepsy. PET radiotracers are made of positron-emitting isotopes, which produce paired gamma photons that the PET scanner detects via coincidence detection, giving PET millimeter-level spatial resolution. As a result, functional imaging data from PET are produced with a spatial resolution that is better than that of single-photon emission computerized tomography. Although several PET radiotracers are available to identify the epileptogenic focus, FDG is still the radiotracer for epilepsy most frequently utilized in clinical settings.
Interictal temporal lobe epilepsy (TLE) and extra-temporal lobe epilepsy (ETLE) epileptogenic foci are linked to a region of impaired glucose metabolism that often extends outside the seizure start zone[52,53]. The evidence supports that PET interictal hypometabolism is better for lateralizing seizure foci and regionalizing the epileptic focus on the lobar level but not for identifying a seizure's starting loci. Therefore, studying the patient's clinical data is essential for accurate image interpretation[54,55]. FDG PET has been demonstrated to be useful in locating hypometabolic areas in non-lesioned epileptogenic lesions and in individuals with modest structural imaging[56,57]. Studies have similarly demonstrated TLE with normal imaging present also to get the seizure focus appropriately lateralized using FDG PET data[58].
Neurooncology: Regarding tumor identification for possible neurosurgical intervention, PET scans are highly predictive of brain tumor presence, often used to identify gliomas[59,60] or brain metastases[61]. For neoplastic pathologies, the diagnostic performance of FDG PET is moderate, with a sensitivity and specificity of 71% and 77%, respectively[62]. For the diagnosis of primary brain tumors, FET PET demonstrates a pooled sensitivity and specificity of over 80%[60]. Somatostatin receptor positron PET imaging may also be useful for meningioma identification, and although less utilized than other imaging modalities in the initial diagnosis of meningiomas, PET is important since MRI alone demonstrates difficulties detecting meningiomas that were close to the falx cerebri or at the base of the skull, had trans-osseous extension, or had ambiguous imaging signals caused by calcifications or aberrations[63-66].
In oncology of the spine, primary and secondary tumor imaging is increasingly being done clinically via PET modalities, particularly with the new technological advancements in this area. These advancements in technology have made it possible for neurosurgeons to evaluate a variety of physiological parameters in vivo, which are crucial for (a) Correctly grading primary and secondary spinal tumors, (b) Tissue characterization, (c) Determining the extent and infiltration of tumors, (d) Surgical planning, and (e) Monitoring therapy. However, due to the decreased sensitivity for identifying hypermetabolic lesions smaller than 2.5x the scanner spatial resolution, evaluation of the spinal cord with PET is partially constrained[67]. However, this technological challenge is becoming less of an issue with newer PET scanners, which have increased clinical interest in spinal PET.
Cerebrovascular disease: Cerebral blood flow in reversible cerebral ischemia has been investigated using 15O-H2O PET and perfusion-weighted imaging (PWI)[68]. Adoption of PWI methods, including DSC and ASL, is still lacking for regular clinical usage due to the lack of consistency and unpredictability in the values the techniques produce. The best imaging method for studying cerebral vascular perfusion continues to be PET and MRI[69]. However, because it is difficult to provide a radiotracer with a brief half-life in the right amount of time to examine brain hemodynamics during an acute stroke, the practical use of 15O-H2O PET in stroke remains a challenge.
Spinal infection: While MRI is the imaging technique of choice for spinal infections, 18F-FDG PET can be a complementary tool in properly diagnosing these pathologies. Studies have found PET to be of clinical use when investigating chronic osteomyelitis using 18F-FDG PET and suspected spondylodiscitis[70-72]. 18F-FDG PET has also demonstrated 100% diagnostic precision in axial bone compared to radiolabeled leukocyte scintigraphy[73].
4.2 Ultrasound
Ultrasound is a highly versatile, real-time imaging technology that relies on sound waves' properties to generate images. It functions via the transduction of sound waves into media, which, in turn, reflect towards the probe with variable shape and intensity. Bone does not allow the transduction of sufficient sound waves; therefore, the cranial-neuropathological application of ultrasound in neurosurgery is often limited to neonates and infants whose cranial sutures have yet to fuse. In these cases, cranial access is most commonly provided by the anterior fontanelle[42]. Physicians often utilize this modality for the observation of both brain maturation and lesion development by way of serial scans[74].
Occasionally, ultrasound use leads to the incidental discovery of abnormalities in cranial structure, such as craniosynostosis and plagiocephaly[75-77]. Ultrasound technology is often used to characterize soft-tissue defects, lesions, and malformation pathologies of the spine, such as incomplete ossification seen in spina bifida[78-80]. To a lesser degree, ultrasound can be used in adults to visualize blood vessels of the neck and monitor blood flow in the brain and help identify potential stroke, brain tumors, hydrocephalus, and vascular issues. Ultrasound can also help neurosurgeons determine tears in ligaments, muscles, tendons, and other soft tissue masses in the back, often more clearly than an X-ray alone, to assist in the differential diagnosis of chronic back pain. Common uses of ultrasound in neurosurgical practice can be found in Table 4.
Table 4. Common Neuropathologies Diagnosed within Neurosurgery by US
US Modality |
Cranial |
Spine |
General |
Carotid artery occlusion |
Spinal dysraphism |
|
Carotid artery stenosis |
Caudal regression syndrome |
|
Intracranial teratoma |
VATER syndrome |
|
Choroid plexus papilloma |
Tethered cords |
|
Craniopharyngioma |
Spina bifida |
|
Hydrocephalus |
Incomplete ossification |
|
Craniosynostosis |
Congenital malformations |
|
Encephalitis |
Soft-tissue defects/damage |
|
Meningitis |
Chronic back pain |
|
Intracranial bleeds (infants) |
|
Ultrasound, US.
4.3 Additional Applications
Diagnosis of fetal brain tumors: The first report of a brain teratoma was discovered by ultrasonography (US) in a fetus at 28 weeks of gestation, published in 1980 by Hoff and Mackay[81]. Since then, further pregnancies with congenital brain tumors have been documented; most commonly gliomas and teratomas[82]. Early detection of malignant cancers during the prenatal period is made possible by modern US devices and routine US screening during pregnancy, further assisting neurosurgeons in diagnosing and acting earlier on the progression of tumor growth[83]. Of note, teratomas make up around 62% of all brain tumors discovered during pregnancy, primarily during the second or third trimester[84].
Choroid plexus papilloma (CPP): The third, fourth, and lateral ventricles are all potential sites for CPP development, with the third trimester being its typical discovery[85]. To aid neurosurgeons in the differential diagnosis of intraventricular hemorrhage, color doppler imaging may help demonstrate vascularization in the lesion[85].
Craniopharyngioma: Craniopharyngiomas make up roughly 2% to 5% of all congenital CNS tumors and are benign. They are most frequently seen in the suprasellar area and arise from squamous cell remnants from Rathke's pouch[86]. US can diagnose this type of large echogenic mass without requiring further imaging.
Hydrocephalus: When the anterior fontanelle is patent throughout the first few months of life, ultrasound is incredibly helpful in assessing the brain and ventricles. Ultrasound is employed for the primary bedside screening of intraventricular hemorrhage and hydrocephalus in newborns[87]. Although the test is sufficient for a general evaluation of the ventricles, it might be difficult to observe specifics regarding the surrounding parenchyma and the third and fourth ventricles. When treating pediatric hydrocephalus, often early in life when the anterior fontanelle is patent, ultrasound works best as a screening or surveillance test for follow-up.
Spinal dysraphism: The range of skin-covered and non-skin-covered abnormalities in neural tube development and closure that make up spinal dysraphism is wide. The dysraphic bone structures may be recognized using ultrasound, and the posterior masses' contents can be visualized[88-90]. Imaging of non-skin-covered abnormalities prior to surgery is typically of little clinical use, as the diagnosis is generally straightforward, with quick skin closure being the key to preventing infection and additional neurological harm. Although US has been proven to be a helpful modality in this scenario, MRI is often used to assess postoperative problems such as retethering[91].
Caudal regression syndrome: Before the fourth week of gestation, caudal cell mass and cloaca injuries can cause various malformations in the caudal spine and pelvic viscera. Although MRI will be necessary for a full examination, US can be utilized to assess the spinal cord and assess the breadth of involvement. Due to short sacral nerve roots and the loss of anterior horn cells, a truncated or blunted cord is frequently observed[92,93].
VATER syndrome: The notochord has an impact on the development of the thoracic and abdominal viscera as well as the vertebral bodies. The VATER syndrome range of visceral and vertebral abnormalities reflects this. Usually, while the newborn is still in the neonatal critical care unit, US is the imaging technique utilized to check for the many visceral defects that might arise. Thus, during the patient's initial examination, abnormalities of the spine and spinal cord may be checked for, and the requirement for any additional imaging can be established. Similarly, dysraphic spinal abnormalities and tethered cords are more common in individuals with urogenital and anorectal malformations[94], and these conditions may be detected with ultrasound.
5 EMERGING DIAGNOSTIC IMAGING
From the viewpoint of current and upcoming neurosurgeons, finding new imaging techniques that provide greater accuracy in demonstrating the anatomy of the central nervous system at microscopic resolutions, in addition to allowing clear visualization of the function or metabolism of the brain, is of the greatest importance moving forward[95]. Several emerging imaging modalities promise to provide such clarity for neurosurgeons to better diagnose various neuropathologies. These include FIESTA sequencing, glymphatic mapping, black-blood MRI, and fMRI changes, among others, which will be discussed here.
5.1 FIESTA Sequencing
Fast imaging employing steady-state acquisition (FIESTA) sequencing is a high-resolution T2-weighted MRI sequence that provides outstanding image contrast[96]. Compared to other steady-state pulse sequences, FIESTA sequence does not suffer from excessive signal saturation or motion artifacts, offering a great signal-to-noise ratio (SNR)[97]. Acting as an excellent adjunct to conventional T1- and T2-weighted sequences, FIESTA sequence has been demonstrated to be of great diagnostic benefit in evaluating cerebral spinal fluid (CSF) within dural reflections of the posterior fossa cranial nerves[98], providing a superior assessment of the effect of tumors on cranial nerve anatomy[99], preoperative localization of the anterior optic pathways in patients with large suprasellar tumors[100], discerning fat graft enhancement from residual or recurrent tumor on delayed postoperative imaging after vestibular schwannoma resection[101], and detecting oculomotor nerve compression in the presence of parasellar lesions[102]. FIESTA sequencing has also been demonstrated to greatly assist in radiosurgical planning and treatment of trigeminal neuralgia[103].
5.2 Glymphatic Mapping
Glial-lymphatic or glymphatic cerebrospinal fluid flow 3D mapping in the brain is accomplished via dynamic contrast-enhanced MRI. The glymphatic system is a fluid-waste clearance pathway recently identified that subserves the flow of CSF into the brain, brain interstitium, and later venous perivascular and perineuronal spaces, ultimately clearing potentially toxic metabolites from neuropil into meningeal and cervical lymphatic drainage vessels[104]. Dysfunction of the glymphatic pathways may be the source of many neuropathologies diagnosed and cared for by neurosurgeons. Although primarily limited to rodent studies, it is understood that these pathways play an important role in CNS health, with glymphatic dysfunction evident in traumatic brain injury[105,106]. Alzheimer’s disease and other dementias[107,108], and micro-infarct disease or stroke[109-111]. If glymphatic mapping proves to be significant for human brain homeostasis, as demonstrated in rodents, it would provide neurosurgeons with new prognostic and diagnostic tools, particularly in TBI and neurovascular care[104].
5.3 Black-blood MRI
MRI has been heavily used in neurosurgery since its inception due to its high versatility with various options for tissue contrast. One of new importance for neurosurgeons, termed black-blood contrast, allows the user to suppress the blood signal in the brain to visualize the vessel wall or better characterize tissue adjacent to the blood pool[102]. Black-blood sequencing has been demonstrated to be superior to conventional T1-sequencing in the automated detection of brain metastases by convolutional neural networks[113,114]. Black-blood imaging has also demonstrated great value in diagnosing and determining the management of patients with intracranial neurovascular pathologies, including intracranial atherosclerosis, aneurysms, vasculitis and vasculopathy, moyamoya disease, dissection, and vertebrobasilar hypoplasia[115].
5.4 Functional MRI (FMRI)
FMRI is a non-invasive option that measures brain activity by detecting changes associated with the blood flow throughout the brain, such as increased movement to the left motor cortex for the movement a patient’s right hand. It allows for determining the spatial relationships between tumor tissue and eloquent brain areas, requiring as a minimum a high-power 3 Tesla MRI unit to reliably detect exact blood flow[116]. FMRI has increasingly been used for preoperative counseling and planning among neurosurgeons in neuro-oncology patients, in addition to intraoperative guidance for tumor resection, particularly in the eloquent cortex[117]. FMRI demonstrates particular importance in neuro-oncology, as it is a much less invasive technique neurosurgeons can use compared to traditional functional studies, such as direct electrical stimulation, while also allowing for more areas of the brain to be mapped for pre- and postoperative decision-making[118].
5.5 Additional Upcoming Advanced Diagnostic Options
Several newer imaging options have come into existence and practices around the globe for a neurosurgeon’s improved diagnostic abilities and subsequent surgical planning. For improved tumor differentiation and characterization, newer techniques include susceptibility-weighted imaging, providing greater sensitivity to the magnetic susceptibility of different tissues, as well as magnetic resonance spectroscopy, allowing for greater evaluation of tumor biochemical and metabolic profile[121]. Newer options for improved perfusion imaging include dynamic susceptibility contrast (DSC), dynamic contrast enhanced (DCE) imaging, arterial spin labeling (ASL), and amino acid positron emission tomography. DSC, DCE, and ASL focuse on cerebral blood volume, permeability, and blood flow, allowing for improved grading of tumors and the prediction of tumor recurrence risk[121]. For improved preoperative planning, diffusion tensor imaging, in addition to the described FMRI, are newer options to better examine the white matter tracts relative to infiltrative tumor material.
6 FUTURE DIRECTIONS
While much progress has been made in neuroimaging modalities, there is surely more to come. One area within neuroimaging which shows great promise involves the development of artificial intelligent systems, machine deep-learning algorithms, radiomics, and ever-increasingly sophisticated imaging analytic methods. Another area of interest is the use of image fusion, or exactly overlapping scans in three-dimensional space, to further assist the neurosurgeon past the diagnostic stage into the operating room, for a more successful operation. These advances are being explored as augmentative tools to enhance the diagnosing capacities of current providers by distinguishing characteristics of various pathologies heretofore invisible to the naked human eye[119]. It is likely that artificially intelligent systems will be increasingly useful to neurosurgeons in deciphering radiographical data to inform patient care in the coming years[120].
7 CONCLUSION
The progression of neurosurgery as a specialty remains heavily reliant on advances in radiology and available neuroimaging options. The authors have provided a clear review of the commonly used imaging modalities and their uses in diagnosis within neurosurgery, including MRI, CT, PET, and US, with important advantages and inherent limitations for the surgeon and patient discussed for each. In addition, several new advancements in neuroimaging tools have come to fruition in recent years, providing a glimpse into what neurosurgeons can expect within the next decade. The synthesis of techniques provided herein may provide meaningful guidance for neurosurgeons in effectively diagnosing neurological pathologies while also helping guide future efforts in neuroimaging developments.
Acknowledgements
Not applicable.
Conflicts of Interest
The authors reported no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Author Contribution
Conception and Design: Cole KL, Lucke-Wold B. Drafting the article: Cole KL, Findlay MC, Kundu M, Johansen C, Rawanduzy C, Lucke-Wold B. Critically revising the article: Cole KL, Findlay MC, Lucke-Wold B. Administrative/technical/material support: Lucke-Wold B.
Abbreviation List
ASL, Arterial spin labeling
CPP, Choroid plexus papilloma
CSF, Cerebral spinal fluid
CT, Computed tomography
CTA, CT angiography
DCE, Dynamic contrast enhanced
DSC, Dynamic susceptibility contrast
ETLE, Extra-temporal lobe epilepsy
FLAIR, Fluid-attenuated inversion recovery
FMRI, Functional MRI
MRI, Resonance imaging
PET, Positron emission tomography
PWI, Perfusion-weighted imaging
TLE, Temporal lobe epilepsy
US, Ultrasonography
References
[1] Preul MC, Feindel W. A history of brain imaging technology in neurosurgery. Neurosurg Clin N Am, 2001; 12: 127-143. DOI: 10.1016/S1042-3680(18)30072-X
[2] Kirkman MA. The role of imaging in the development of neurosurgery. J Clin Neurosci, 2015; 22: 55-61. DOI: 10.1016/j.jocn.2014.05.024
[3] Langen KJ, Galldiks N, Hattingen E, Shah NJ. Advances in neuro-oncology imaging. Nat Rev Neurol, 2017; 13: 279-289. DOI: 10.1038/nrneurol.2017.44
[4] Vagvala S, Guenette JP, Jaimes C et al. Imaging diagnosis and treatment selection for brain tumors in the era of molecular therapeutics. Cancer Imaging, 2022; 22: 19. DOI: 10.1186/s40644-022-00455-5
[5] Serai SD. Basics of magnetic resonance imaging and quantitative parameters T1, T2, T2*, T1rho and diffusion-weighted imaging. Pediatr Radiol, 2022; 52: 217-227. DOI: 10.1007/s00247-021-05042-7
[6] De Kerviler E, Cuenod CA, Clément O et al. What is bright on T1 MRI scans? J Radiol, 1998; 79: 117-126.
[7] Zimny A, Zińska L, Bladowska J et al. Intracranial lesions with high signal intensity on T1-weighted MR images-review of pathologies. Pol J Radiol, 2013; 78: 36-46. DOI: 10.12659/PJR.889663
[8] Golfieri R, Baddeley H, Pringle JS et al. The role of the STIR sequence in magnetic resonance imaging examination of bone tumours. Br J Radiol, 1990; 63: 251-256. DOI: 10.1259/0007-1285-63-748-251
[9] StatPearls. Accessed 2022. Available at https://www.statpearls.com
[10] Zhou Z, Lu ZR. Gadolinium-based contrast agents for magnetic resonance cancer imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol, 2013; 5: 1-18. DOI: 10.1002/wnan.1198
[11] Wang B, Zhang Y, Zhao B et al. Postcontrast T1 Mapping for Differential Diagnosis of Recurrence and Radionecrosis after Gamma Knife Radiosurgery for Brain Metastasis. AJNR Am J Neuroradiol, 2018; 39: 1025-1031. DOI: 10.3174/ajnr.A5643
[12] Hu HH, Benkert T, Smith M et al. Post-contrast T1-weighted spine 3T MRI in children using a golden-angle radial acquisition. Neuroradiology, 2019; 61: 341-349. DOI: 10.1007/s00234-019-02165-5
[13] Zimny A, Neska-Matuszewska M, Bladowska J et al. Intracranial Lesions with Low Signal Intensity on T2-weighted MR Images - Review of Pathologies. Pol J Radiol, 2015; 80: 40-50. DOI: 10.12659/PJR.892146
[14] Chavhan GB, Babyn PS, Thomas B et al. Principles, techniques, and applications of T2*-based MR imaging and its special applications. Radiographics, 2009; 29: 1433-1449. DOI: 10.1148/rg.295095034
[15] Pan JW, Moon CH, Hetherington HP. Cerebrospinal fluid-suppressed T. Magn Reson Med, 2019; 81: 2924-2936. DOI: 10.1002/mrm.27598
[16] Keerthivasan MB, Winegar B, Becker JL et al. Clinical Utility of a Novel Ultrafast T2-Weighted Sequence for Spine Imaging. AJNR Am J Neuroradiol, 2018; 39: 1568-1575. DOI: 10.3174/ajnr.A5713
[17] Lee EK, Lee EJ, Kim S et al. Importance of Contrast-Enhanced Fluid-Attenuated Inversion Recovery Magnetic Resonance Imaging in Various Intracranial Pathologic Conditions. Korean J Radiol, 2016; 17: 127-141. DOI: 10.3348/kjr.2016.17.1.127
[18] Bakshi R, Ariyaratana S, Benedict RH et al. Fluid-attenuated inversion recovery magnetic resonance imaging detects cortical and juxtacortical multiple sclerosis lesions. Arch Neurol, 2001; 58: 742-748. DOI: 10.1001/archneur.58.5.742
[19] Kates R, Atkinson D, Brant-Zawadzki M. Fluid-attenuated inversion recovery (FLAIR): clinical prospectus of current and future applications. Top Magn Reson Imaging, 1996; 8: 389-396.
[20] Stevenson VL, Gawne-Cain ML, Barker GJ et al. Imaging of the spinal cord and brain in multiple sclerosis: a comparative study between fast FLAIR and fast spin echo. J Neurol, 1997; 244: 119-124. DOI: 10.1007/s004150050060
[21] Filippi M, Yousry TA, Alkadhi H et al. Spinal cord MRI in multiple sclerosis with multicoil arrays: a comparison between fast spin echo and fast FLAIR. J Neurol Neurosurg Psychiatry, 1996; 61: 632-635. DOI: 10.1136/jnnp.61.6.632
[22] Sati P, George IC, Shea CD et al. FLAIR*: a combined MR contrast technique for visualizing white matter lesions and parenchymal veins. Radiology, 2012; 265: 926-932. DOI: 10.1148/radiol.12120208
[23] Baliyan V, Das CJ, Sharma R et al. Diffusion weighted imaging: Technique and applications. World J Radiol, 2016; 8: 785-798. DOI: 10.4329/wjr.v8.i9.785
[24] Mascalchi M, Filippi M, Floris R et al. Diffusion-weighted MR of the brain: methodology and clinical application. Radiol Med, 2005; 109: 155-197.
[25] Cotten A, Kermarrec E, Moraux A et al. New MRI sequences. Joint Bone Spine, 2009; 76: 588-590. DOI: 10.1016/j.jbspin.2009.09.002
[26] Lee B, Newberg A. Neuroimaging in traumatic brain imaging. NeuroRx, 2005; 2: 372-383. DOI: 10.1602/neurorx.2.2.372
[27] Mushlin AI, Mooney C, Holloway RG et al. The cost-effectiveness of magnetic resonance imaging for patients with equivocal neurological symptoms. Int J Technol Assess Health Care, 1997; 13: 21-34. DOI: 10.1017/s0266462300010205
[28] Ugwuanyi DC, Sibeudu TF, Irole CP et al. Evaluation of common findings in brain computerized tomography (CT) scan: A single center study. AIMS Neurosci, 2020; 7: 311-318. DOI: 10.3934/Neuroscience.2020017
[29] van Rijn RM, Wassenaar M, Verhagen AP et al. Computed tomography for the diagnosis of lumbar spinal pathology in adult patients with low back pain or sciatica: a diagnostic systematic review. Eur Spine J, 2012; 21: 228-239. DOI: 10.1007/s00586-011-2012-2
[30] Tins B. Technical aspects of CT imaging of the spine. Insights Imaging, 2010; 1: 349-359. DOI: 10.1007/s13244-010-0047-2
[31] Alshumrani G, Al Abo Nasser B, Alzawani Aet al. The role of computed tomography angiogram in intracranial hemorrhage. Do the benefits justify the known risks in everyday practice? Clin Neurol Neurosurg, 2021; 200: 106379. DOI: 10.1016/j.clineuro.2020.106379
[32] Brasil S, Bor-Seng-Shu E, de-Lima-Oliveira M et al. Computed tomography angiography accuracy in brain death diagnosis. J Neurosurg, 2019; 2019: 1-9. DOI: 10.3171/2019.6.JNS191107
[33] Katz DA, Marks MP, Napel SA et al. Circle of Willis: evaluation with spiral CT angiography, MR angiography, and conventional angiography. Radiology, 1995; 195: 445-449. DOI: 10.1148/radiology.195.2.7724764
[34] Sabarudin A, Subramaniam C, Sun Z. Cerebral CT angiography and CT perfusion in acute stroke detection: a systematic review of diagnostic value. Quant Imaging Med Surg, 2014; 4: 282-290. DOI: 10.3978/j.issn.2223-4292.2014.07.10
[35] Silvennoinen HM, Hamberg LM, Lindsberg PJ et al. CT perfusion identifies increased salvage of tissue in patients receiving intravenous recombinant tissue plasminogen activator within 3 hours of stroke onset. AJNR Am J Neuroradiol, 2008; 29: 1118-1123. DOI: 10.3174/ajnr.A1039
[36] Rawson JV, Pelletier AL. When to Order a Contrast-Enhanced CT. Am Fam Physician, 2013; 88: 312-316.
[37] Royal HD. Effects of low level radiation-what's new? Semin Nucl Med, 2008; 38: 392-402. DOI: 10.1053/j.semnuclmed.2008.05.006
[38] Power SP, Moloney F, Twomey M et al. Computed tomography and patient risk: Facts, perceptions and uncertainties. World J Radiol, 2016; 8: 902-915. DOI: 10.4329/wjr.v8.i12.902
[39] Lameka K, Farwell MD, Ichise M. Positron Emission Tomography. Handb Clin Neurol, 2016; 135: 209-227. DOI: 10.1016/B978-0-444-53485-9.00011-8
[40] Mishina M. Positron emission tomography for brain research. J Nippon Med Sch, 2008; 75: 68-76. DOI: 10.1272/jnms.75.68
[41] Hasnain N, Mustafa RM, Bakhshi SK et al. Efficacy of Positron Emission Tomography in distinguishing brain tumours from inflammation. J Pak Med Assoc, 2020; 70: 2291-2293.
[42] Glick Y. Head Ultrasound. Radiopedia 2022. DOI: 10.53347/rID-73886
[43] Pronin IN, Khokhlova EV, Konakova TA et al. Positron emission tomography with 11C-methionine in primary brain tumor diagnosis. Zh Nevrol Psikhiatr Im S S Korsakova, 2020; 120: 51-56. DOI: 10.17116/jnevro202012008151
[44] Mattoli MV, Treglia G, Calcagni ML et al. Usefulness of Brain Positron Emission Tomography with Different Tracers in the Evaluation of Patients with Idiopathic Normal Pressure Hydrocephalous. Int J Mol Sci, 2020; 21: 1. DOI: 10.3390/ijms21186523
[45] Tatsch K. Positron emission tomography in diagnosis and differential diagnosis of Parkinson's disease. Neurodegener Dis, 2010; 7: 330-340. DOI: 10.1159/000314499
[46] Shetty-Alva N, Novotny EJ, Shetty T et al. Positron emission tomography in Rasmussen's encephalitis. Pediatr Neurol, 2007; 36: 112-114. DOI: 10.1016/j.pediatrneurol.2006.07.012
[47] Chan TLH, Romsa J, Steven DA et al. Refractory Epilepsy: The Role of Positron Emission Tomography. Can J Neurol Sci, 2018; 45: 30-34. DOI: 10.1017/cjn.2017.244
[48] Burneo JG, Poon R, Kellett S et al. The Utility of Positron Emission Tomography in Epilepsy. Can J Neurol Sci, 2015; 42: 360-371. DOI: 10.1017/cjn.2015.279
[49] Segawa T, Koga H, Oshina M et al. The Diagnostic Value of Fluorine-18 Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for the Detection of Surgical Site Infection after Spine Surgery. Spine (Phila Pa 1976), 2021; 46: E602-E610. DOI: 10.1097/BRS.0000000000003847
[50] Eicker SO, Langen KJ, Galldiks N et al. Clinical value of 2-deoxy-[18F]fluoro-D-glucose positron emission tomography in patients with cervical spondylotic myelopathy. Neurosurg Focus, 2013; 35: E2. DOI: 10.3171/2013.3.FOCUS1379
[51] Uchida K, Nakajima H, Yayama T et al. High-resolution magnetic resonance imaging and 18FDG-PET findings of the cervical spinal cord before and after decompressive surgery in patients with compressive myelopathy. Spine (Phila Pa 1976), 2009; 34: 1185-1191. DOI: 10.1097/BRS.0b013e31819e2919
[52] Theodore WH, Sato S, Kufta C et al. Temporal lobectomy for uncontrolled seizures: the role of positron emission tomography. Ann Neurol, 1992; 32: 789-794. DOI: 10.1002/ana.410320613
[53] Mendes Coelho VC, Morita ME, Amorim BJ et al. Automated Online Quantification Method for. Front Neurol, 2017; 8: 453. DOI: 10.3389/fneur.2017.00453
[54] Takaya S, Hanakawa T, Hashikawa K et al. Prefrontal hypofunction in patients with intractable mesial temporal lobe epilepsy. Neurology, 2006; 67: 1674-1676. DOI: 10.1212/01.wnl.0000242628.26978.e2
[55] Wong CH, Bleasel A, Wen L et al. The topography and significance of extratemporal hypometabolism in refractory mesial temporal lobe epilepsy examined by FDG-PET. Epilepsia, 2010; 51: 1365-1373. DOI: 10.1111/j.1528-1167.2010.02552.x
[56] Henry TR, Sutherling WW, Engel J et al. Interictal cerebral metabolism in partial epilepsies of neocortical origin. Epilepsy Res, 1991; 10: 174-182. DOI: 10.1016/0920-1211(91)90010-d
[57] Schlaug G, Antke C, Holthausen H et al. Ictal motor signs and interictal regional cerebral hypometabolism. Neurology, 1997; 49: 341-350. DOI: 10.1212/wnl.49.2.341
[58] Carne RP, O'Brien TJ, Kilpatrick CJ et al. MRI-negative PET-positive temporal lobe epilepsy: a distinct surgically remediable syndrome. Brain, 2004; 127: 2276-2285. DOI: 10.1093/brain/awh257
[59] Rapp M, Heinzel A, Galldiks N et al. Diagnostic performance of 18F-FET PET in newly diagnosed cerebral lesions suggestive of glioma. J Nucl Med, 2013; 54: 229-235. DOI: 10.2967/jnumed.112.109603
[60] Dunet V, Rossier C, Buck A et al. Performance of 18F-fluoro-ethyl-tyrosine (18F-FET) PET for the differential diagnosis of primary brain tumor: a systematic review and Metaanalysis. J Nucl Med, 2012; 53: 207-214. DOI: 10.2967/jnumed.111.096859
[61] Unterrainer M, Galldiks N, Suchorska B et al. F-FET PET Uptake Characteristics in Patients with Newly Diagnosed and Untreated Brain Metastasis. J Nucl Med, 2017; 58: 584-589. DOI: 10.2967/jnumed.116.180075
[62] Zhao C, Zhang Y, Wang J. A meta-analysis on the diagnostic performance of (18)F-FDG and (11)C-methionine PET for differentiating brain tumors. AJNR Am J Neuroradiol, 2014; 35: 1058-1065. DOI: 10.3174/ajnr.A3718
[63] Galldiks N, Albert NL, Sommerauer M et al. PET imaging in patients with meningioma-report of the RANO/PET Group. Neuro Oncol, 2017; 19: 1576-1587. DOI: 10.1093/neuonc/nox112
[64] Delbeke D, Meyerowitz C, Lapidus RL et al. Optimal cutoff levels of F-18 fluorodeoxyglucose uptake in the differentiation of low-grade from high-grade brain tumors with PET. Radiology, 1995; 195: 47-52. DOI: 10.1148/radiology.195.1.7892494
[65] Afshar-Oromieh A, Giesel FL, Linhart HG et al. Detection of cranial meningiomas: comparison of ⁶⁸Ga-DOTATOC PET/CT and contrast-enhanced MRI. Eur J Nucl Med Mol Imaging, 2012; 39: 1409-1415. DOI: 10.1007/s00259-012-2155-3
[66] Li Y, Jin G, Su D. Comparison of Gadolinium-enhanced MRI and 18FDG PET/PET-CT for the diagnosis of brain metastases in lung cancer patients: A meta-analysis of 5 prospective studies. Oncotarget, 2017; 8: 35743-35749. DOI: 10.18632/oncotarget.16182
[67] Hoffman EJ, Huang SC, Phelps ME. Quantitation in positron emission computed tomography: 1. Effect of object size. J Comput Assist Tomogr, 1979; 3: 299-308. DOI: 10.1097/00004728-197906000-00001
[68] Heiss WD, Grond M, Thiel A et al. Tissue at risk of infarction rescued by early reperfusion: a positron emission tomography study in systemic recombinant tissue plasminogen activator thrombolysis of acute stroke. J Cereb Blood Flow Metab, 1998; 18: 1298-1307. DOI: 10.1097/00004647-199812000-00004
[69] Werner P, Saur D, Zeisig V et al. Simultaneous PET/MRI in stroke: a case series. J Cereb Blood Flow Metab, 2015; 35: 1421-1425. DOI: 10.1038/jcbfm.2015.158
[70] Guhlmann A, Brecht-Krauss D, Suger G et al. Chronic osteomyelitis: detection with FDG PET and correlation with histopathologic findings. Radiology, 1998; 206: 749-754. DOI: 10.1148/radiology.206.3.9494496
[71] Schmitz A, Risse JH, Grünwald F et al. Fluorine-18 fluorodeoxyglucose positron emission tomography findings in spondylodiscitis: preliminary results. Eur Spine J, 2001; 10: 534-539. DOI: 10.1007/s005860100339
[72] Guhlmann A, Brecht-Krauss D, Suger G et al. Fluorine-18-FDG PET and technetium-99m antigranulocyte antibody scintigraphy in chronic osteomyelitis. J Nucl Med, 1998; 39: 2145-2152.
[73] Meller J, Köster G, Liersch T et al. Chronic bacterial osteomyelitis: prospective comparison of (18)F-FDG imaging with a dual-head coincidence camera and (111)In-labelled autologous leucocyte scintigraphy. Eur J Nucl Med Mol Imaging, 2002; 29: 53-60. DOI: 10.1007/s00259-001-0661-9
[74] van Wezel-Meijler G, Steggerda SJ, Leijser LM. Cranial ultrasonography in neonates: role and limitations. Semin Perinatol, 2010; 34: 28-38. DOI: 10.1053/j.semperi.2009.10.002
[75] Yoon HK, Cho SW. Neonatal head ultrasound: systematic approach to congenital Central Nervous System anomalies. A pictorial essay. Med Ultrason, 2016; 18: 386-393. DOI: 10.11152/mu.2013.2066.183.hye
[76] Rozovsky K, Udjus K, Wilson N et al. Cranial Ultrasound as a First-Line Imaging Examination for Craniosynostosis. Pediatrics, 2016; 137: e20152230. DOI: 10.1542/peds.2015-2230
[77] Li P, Jiang X, Zhang G et al. Corrigendum to: Utilizing image and caption information for biomedical document classification. Bioinformatic, 2021; 37: i468-i476. DOI: 10.1093/bioinformatics/btab568
[78] Rees MA, Squires JH, Coley BD et al. Ultrasound of congenital spine anomalies. Pediatr Radiol, 2021; 51: 2442-2457. DOI: 10.1007/s00247-021-05178-6
[79] Ladino Torres MF, DiPietro MA. Spine ultrasound imaging in the newborn. Semin Ultrasound CT MR, 2014; 35: 652-661. DOI: 10.1053/j.sult.2014.08.001
[80] Coleman BG, Langer JE, Horii SC. The diagnostic features of spina bifida: the role of ultrasound. Fetal Diagn Ther, 2015; 37: 179-196. DOI: 10.1159/000364806
[81] Hoff NR, Mackay IM. Prenatal ultrasound diagnosis of intracranial teratoma. J Clin Ultrasound, 1980; 8: 247-249. DOI: 10.1002/jcu.1870080313
[82] Cavalheiro S, Moron AF, Hisaba W, Dastoli P, Silva NS. Fetal brain tumors. Childs Nerv Syst, 2003; 19: 529-536. DOI: 10.1007/s00381-003-0770-9
[83] Palo P, Penttinen M, Kalimo H. Early ultrasound diagnosis of fetal intracranial tumors. J Clin Ultrasound, 1994; 22: 447-450. DOI: 10.1002/jcu.1870220707
[84] Schlembach D, Bornemann A, Rupprecht T et al. Fetal intracranial tumors detected by ultrasound: a report of two cases and review of the literature. Ultrasound Obstet Gynecol, 1999; 14: 407-418. DOI: 10.1046/j.1469-0705.1999.14060407.x
[85] Meizner I. TUMORS OF THE BRAIN. In: Hill M ed. Ultrasonography of the Prenatal Brain. 2017; 393-406.
[86] Kawamata T, Kubo O, Hori T. Histological findings at the boundary of craniopharyngiomas. Brain Tumor Pathol. 2005; 22: 75-78. DOI: 10.1007/s10014-005-0191-4
[87] Nielsen N, Breedt A. Hydrocephalus. In: Cartwright C, Wallace D eds. Nursing Care of the Pediatric Neurosurgery Patient. Springer, Berlin: 2012; 37-84.
[88] Rubin JM, DiPietro MA, Chandler WF et al. Spinal ultrasonography. Intraoperative and pediatric applications. Radiol Clin North Am, 1988; 26: 1-27.
[89] Zieger M, Dörr U, Schulz RD. Pediatric spinal sonography. Part II: Malformations and mass lesions. Pediatr Radiol, 1988; 18: 105-111. DOI: 10.1007/BF02387552
[90] Korsvik HE. Ultrasound assessment of congenital spinal anomalies presenting in infancy. Semin Ultrasound CT MR, 1994; 15: 264-274. DOI: 10.1016/s0887-2171(05)80086-1
[91] Gerscovich EO, Maslen L, Cronan MS et al. Spinal sonography and magnetic resonance imaging in patients with repaired myelomeningocele: comparison of modalities. J Ultrasound Med, 1999; 18: 655-664. DOI: 10.7863/jum.1999.18.9.655
[92] Ball WS. Pediatric neuroradiology. AJNR Am J Neuroradiol, 2000; 21: 29-36.
[93] Barkovich AJ. Pediatric neuroimaging. Lippincott Williams & Wilkins, Philadelphia; 2000.
[94] Long FR, Hunter JV, Mahboubi S et al. Tethered cord and associated vertebral anomalies in children and infants with imperforate anus: evaluation with MR imaging and plain radiography. Radiology, 1996; 200: 377-382. DOI: 10.1148/radiology.200.2.8685329
[95] Oishi M, Uzuka T, Yoneoka Y et al. New diagnostic imaging methods in neurosurgery: advent of anatomical and functional neuroimaging. No Shinkei Geka, 2007; 35: 291-300.
[96] Cavusoglu M, Cılız DS, Duran S et al. Temporal bone MRI with 3D-FIESTA in the evaluation of facial and audiovestibular dysfunction. Diagn Interv Imaging, 2016; 97: 863-869. DOI: 10.1016/j.diii.2015.11.010
[97] Bhosale P, Ma J, Choi H. Utility of the FIESTA pulse sequence in body oncologic imaging: review. AJR Am J Roentgenol, 2009; 192(6 Suppl): S83-93. DOI: 10.2214/AJR.07.7062
[98] Noble DJ, Scoffings D, Ajithkumar T et al. Fast imaging employing steady-state acquisition (FIESTA) MRI to investigate cerebrospinal fluid (CSF) within dural reflections of posterior fossa cranial nerves. Br J Radiol, 2016; 89: 20160392. DOI: 10.1259/bjr.20160392
[99] Mikami T, Minamida Y, Yamaki T et al. Cranial nerve assessment in posterior fossa tumors with fast imaging employing steady-state acquisition (FIESTA). Neurosurg Rev, 2005; 28: 261-266. DOI: 10.1007/s10143-005-0394-5
[100] Watanabe K, Kakeda S, Yamamoto J et al. Delineation of optic nerves and chiasm in close proximity to large suprasellar tumors with contrast-enhanced FIESTA MR imaging. Radiology, 2012; 264: 852-858. DOI: 10.1148/radiol.12111363
[101] Kamal N, Reddy RK, Kohli G et al. The Role of Fast Imaging Employing Steady-State Acquisition (FIESTA) Magnetic Resonance Imaging for Assessment of Delayed Enhancement of Fat Graft Packing on Postoperative Imaging After Vestibular Schwannoma Surgery. World Neurosurg, 2018; 114: e1066-e1072. DOI: 10.1016/j.wneu.2018.03.147
[102] Yamauchi T, Kitai R, Neishi H et al. Detection of oculomotor nerve compression by 3D-FIESTA MRI in a patient with pituitary apoplexy and diabetes mellitus. No Shinkei Geka, 2014; 42: 137-142.
[103] Chávez GD, De Salles AA, Solberg TD et al. Three-dimensional fast imaging employing steady-state acquisition magnetic resonance imaging for stereotactic radiosurgery of trigeminal neuralgia. Neurosurgery, 2005; 56: E628. DOI: 10.1227/01.neu.0000154709.44776.50
[104] Rasmussen MK, Mestre H, Nedergaard M. The glymphatic pathway in neurological disorders. Lancet Neurol, 2018; 17: 1016-1024. DOI: 10.1016/S1474-4422(18)30318-1
[105] Iliff JJ, Chen MJ, Plog BA et al. Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury. J Neurosci, 2014; 34: 16180-16193. DOI: 10.1523/JNEUROSCI.3020-14.2014
[106] Plog BA, Dashnaw ML, Hitomi E et al. Biomarkers of traumatic injury are transported from brain to blood via the glymphatic system. J Neurosci, 2015; 35: 518-526. DOI: 10.1523/JNEUROSCI.3742-14.2015
[107] Iliff JJ, Wang M, Liao Y et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med, 2012; 4: 147ra111. DOI: 10.1126/scitranslmed.3003748
[108] Xu Z, Xiao N, Chen Y et al. Deletion of aquaporin-4 in APP/PS1 mice exacerbates brain Aβ accumulation and memory deficits. Mol Neurodegener, 2015; 10: 58. DOI: 10.1186/s13024-015-0056-1
[109] Gaberel T, Gakuba C, Goulay R et al. Impaired glymphatic perfusion after strokes revealed by contrast-enhanced MRI: a new target for fibrinolysis? Stroke, 2014; 45: 3092-3096. DOI: 10.1161/STROKEAHA.114.006617
[110] Goulay R, Flament J, Gauberti M et al. Subarachnoid Hemorrhage Severely Impairs Brain Parenchymal Cerebrospinal Fluid Circulation in Nonhuman Primate. Stroke, 2017; 48: 2301-2305. DOI: 10.1161/STROKEAHA.117.017014
[111] Zbesko JC, Nguyen TV, Yang T et al. Glial scars are permeable to the neurotoxic environment of chronic stroke infarcts. Neurobiol Dis, 2018; 112: 63-78. DOI: 10.1016/j.nbd.2018.01.007
[112] Henningsson M, Malik S, Botnar R et al. Black-Blood Contrast in Cardiovascular MRI. J Magn Reson Imaging, 2022; 55: 61-80. DOI: 10.1002/jmri.27399
[113] Kottlors J, Geissen S, Jendreizik H et al. Contrast-Enhanced Black Blood MRI Sequence Is Superior to Conventional T1 Sequence in Automated Detection of Brain Metastases by Convolutional Neural Networks. Diagnostics (Basel), 2021; 11: 6. DOI: 10.3390/diagnostics11061016
[114] Jun Y, Eo T, Kim T et al. Deep-learned 3D black-blood imaging using automatic labelling technique and 3D convolutional neural networks for detecting metastatic brain tumors. Sci Rep, 2018; 8: 9450. DOI: 10.1038/s41598-018-27742-1
[115] Chaganti J, Woodford H, Tomlinson S et al. Black blood imaging of intracranial vessel walls. Pract Neurol, 2020; 21: 91. DOI: 10.1136/practneurol-2020-002806
[116] Vlieger EJ, Majoie CB, Leenstra S et al. Functional magnetic resonance imaging for neurosurgical planning in neurooncology. Eur Radiol, 2004; 14: 1143-1153. DOI: 10.1007/s00330-004-2328-y
[117] Silva MA, See AP, Essayed WI et al. Challenges and techniques for presurgical brain mapping with functional MRI. Neuroimage Clin, 2018; 17: 794-803. DOI: 10.1016/j.nicl.2017.12.008
[118] Tharin S, Golby A. Functional brain mapping and its applications to neurosurgery. Neurosurgery, 2007; 60(4 Suppl 2): 185-201. DOI: 10.1227/01.NEU.0000255386.95464.52
[119] Yi Z, Long L, Zeng Y et al. Current Advances and Challenges in Radiomics of Brain Tumors. Front Oncol, 2021; 11: 732196. DOI: 10.3389/fonc.2021.732196
[120] Noh SH, Cho PG, Kim KN et al. Artificial Intelligence for Neurosurgery : Current State and Future Directions. J Korean Neurosurg Soc, 2022 [pre-print]. DOI: 10.3340/jkns.2022.0130
[121] Carrete LR, Young JS, Cha S. Advanced Imaging Techniques for Newly Diagnosed and Recurrent Gliomas. Front Neurosci, 2022; 16: 787755. DOI: 10.3389/fnins.2022.787755
Copyright © 2023 The Author(s). This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.




 Copyright ©
Copyright ©