Soft Magnetoelasticity for Mechanical Energy Harvesting
Minyan Ge1, Shumao Xu1,2*, Yurui Tang1, Yuchun Wang3, Xinyi Cui4, Weiqiang Zhang1,5*, Jing Wang1*
1Institute of Science and Technology for Brain-inspired Intelligence (ISTBI), Fudan University, Shanghai, China
2Department of Biomedical Engineering, The Pennsylvania State University, Pennsylvania, USA
3Huashan Hospital, Fudan University, Shanghai, China
4Shanghai University of Traditional Chinese Medicine, Shanghai, China
5Human Phenome Institute, Fudan University, Shanghai, China
*Correspondence to: Shumao Xu, PhD, Associate Professor, Institute of Science and Technology for Brain-inspired Intelligence (ISTBI), Fudan University, Shanghai, 201203, China; Department of Biomedical Engineering, The Pennsylvania State University, University Park, Pennsylvania, 16802, USA; E-mail: shumaoxu@fudan.edu.cn
Jing Wang, PhD, Associate Professor, Institute of Science and Technology for Brain-inspired Intelligence (ISTBI), Fudan University, Shanghai, 201203, China; E-mail: wangjing_@fudan.edu.cn
DOI: 10.53964/id.2025007
Abstract
In the rapid evolution of modern healthcare, the fusion of bioelectronics with mechanical energy harvesting techniques marks a significant advancement. Wearable biosensors and implantable devices, which are vital for enhancing patient care, enable the continuous monitoring of physiological and biomechanical activities, paving the way for reduced healthcare costs and improved patient quality of life. At the forefront of this progress is soft magnetoelasticity, which offers a revolutionary method for powering wearable and implantable healthcare devices. This technology harnesses the giant magnetoelastic effect in soft magnetic elastomers to transform biomechanical energy directly into electricity, overcoming the limitations of traditional electromagnetic generators including inadequate response to biomechanical stress and a mismatch in mechanical moduli with human tissues. Soft magnetoelastic generators boast superior energy conversion efficiency and better compatibility with human tissue mechanics, which show remarkable potential in applications that span from real-time wearable health monitoring to the operation of implantable devices, all without the need for battery replacements. This review on soft magnetoelasticity, from its fundamental concepts to its cutting-edge applications, aims to highlight its transformative impact on bioelectronics and its crucial role in advancing wearable and implantable medical technologies towards sustainable and autonomous healthcare solutions.
Keywords: soft magnetoelastic materials, mechanical energy harvesting, smart textile, electromagnetic generators
1 INTRODUCTION
In the evolving landscape of modern healthcare, bioelectronics have become crucial, transforming medical applications through wearable biosensors and implantable devices[1-6]. These technologies are vital for enhancing patient care by enabling continuous monitoring of physiological and biomechanical activities, thereby reducing healthcare costs and improving patients' quality of life. The development of advanced mechanisms for mechanical energy harvesting is imperative to ensure these devices operate sustainably and efficiently, minimizing the discomfort and risks associated with battery replacements in implantable devices[7-10]. Given that cardiovascular conditions stand as the primary reason for death globally[11], continuous monitoring techniques for heart rhythm and stroke volume are essential to prevent conditions like cardiac arrhythmias, which could lead to sudden death. The challenge lies in creating self-powered bioelectronic devices capable of continuous operation without high power consumption.
Current biomechanical energy conversion methods primarily use electromagnetic generators that generate energy from human motion through cutting magnetic movement like gear-and-generator and inertial induction systems[12-14]. However, the use of traditional magnetostrictive materials, like GaxFe1-x (Galfenol) and TbxDy1-xFe2 (Terfenol-D) has been limited in bioelectronics due to their insufficient response to biomechanical stress levels, the complexity of external magnetic field requirements, and a significant mismatch between their mechanical moduli and human tissues[15-18]. Soft magnetoelasticity offers a groundbreaking approach by altering magnetic properties in soft materials like elastomers under mechanical stress or strain, enabling more efficient biomechanical energy harvesting into electricity. This approach represents a shift from traditional, rigid metal alloys to flexible materials that can better interface with biological tissues. The giant magnetoelastic effect, first discovered in 1865[19], has typically been utilized in rigid metal alloys for vibration control applications in civil engineering. Recent advancements have demonstrated the potential of soft magnetoelastic materials to revolutionize mechanical energy harvesting, achieving magnetomechanical coupling coefficient reaching up to 7.19×10−8 T·Pa−1[15], far surpassing the performance of traditional metal alloys. This enhanced coupling factor not only enables the more efficient transformation of mechanical energy into electricity but ensures compatibility with the mechanical properties of human tissue, opening new avenues for the creation of self-powered bioelectronic devices. Soft magnetoelastic generators (MEGs) have been developed to harness this giant magnetoelastic effect in various applications, such as measuring human pulse signals under sweating[16-18] and enabling implantable power generation without the requirement for encapsulation[15,20,21], demonstrating their waterproof nature and the minimal impact of magnetic field attenuation through water[17,18,22-24].
This significant leap in soft magnetoelastic technology represents an essential milestone in mechanical energy harvesting, offering a sustainable and efficient power source for bioelectronic devices. By delving into the intricacies of soft magnetoelasticity, including its operational principles, and recent findings, this perspective aims to present a comprehensive overview of its potential to transform the field of bioelectronics. The detailed examination of these developments not only highlights the challenges overcome but also outlines the promising future applications of soft magnetoelasticity in powering the next evolution of medical bioelectronics.
2 MECHANICAL ENERGY HARVESTING
Soft magnetoelasticity is transforming the field of mechanical energy harvesting, which utilizes Faraday's principle of electromagnetic induction, adeptly converting mechanical forces into electrical energy in a manner that harmonizes with human biomechanics for wearable technologies. This section contrasts different energy conversion methods (Figure 1), highlighting MEG for its efficiency, ability to generate high current, and suitability for human motion.
|
Figure 1. Schematic Illustrations of Various Mechanical Energy Harvesting Methods. A: Triboelectric nanogenerator generating electricity through contact electrification and electrostatic induction. B: Piezoelectric nanogenerator converting mechanical stress to electrical energy using piezoelectric materials. C: Electromagnetic generator inducing electric current by moving a conductor through a magnetic field. D: Vibration work-function energy harvester. Reproduced from Ref.[105] with permission from Springer Nature. E: Electrostatic (capacitive) harvester, reproduced from ref.[106] Copyright 2012, under a Creative Commons CC-BY-NC-SA license. F: Mechanically assisted thermoelectric generator. (G) Hydrodynamic harvester. Reproduced from Ref.[113] with permission from Springer Nature, under a Creative Commons CC BY license. H: Electret generator, reproduced from Ref.[98] Copyright 2012, under a Creative Commons CC-BY-NC-SA license. I: Magnetoelastic generators producing electricity from the deformation of magnetostrictive materials. Reproduced from Ref.[116] with permission from Cell Press. J: Magnetoelastic generator sphere encompassing acrylic casing, a magnetoelastic composite wall as magnetomechanical coupling (MC) layer, a coil as magnetic induction (MI) layer, and a mobile glass sphere. K: shift in magnetic alignment and flux density under stress; L: application in converting vibrational energy into electricity. Reproduced from Ref.[24] with permission from American Chemical Society.
2.1 Triboelectric Nanogenerator
Triboelectric nanogenerator (TENG) that utilizes the triboelectric effect in combination with electrostatic induction has the capability to transform mechanical energy into electrical power (Figure 1A). This technology traces back to the observations of Thales, an early Greek philosopher from the pre-Socratic era, when rubbed with fur, could attract small objects like straw or hair, showcasing the triboelectric effect[25-28]. Although this phenomenon has a long history, it was only in recent times that the detailed mechanisms of triboelectrification were fully understood, that is, contact between two different materials can lead to the formation and separation of chemical bonds at their interface, enabling the transfer of charge (electrons, ions, or molecules) to balance the electrochemical potentials between them[29-31]. This results in one material carrying positive charge and the other negative charge[32]. TENGs utilize materials that are triboelectrically positive, such as polyamide (PA), metals, zinc oxide (ZnO), and indium tin oxide (ITO), and those that are triboelectrically negative, including polydimethylsiloxane (PDMS), polytetrafluoroethylene (PTFE), polyvinylidene fluoride (PVDF), polyethylene terephthalate (PET), and fluorinated ethylene propylene (FEP)[33-38]. These materials are chosen for their efficient charge transfer and separation capabilities under mechanical stress.
Characterized by four main operational modes-vertical contact-detach, lateral sliding, free-standing triboelectric-layer, and single-electrode mode-ENGs offer versatility, high energy conversion efficiency and large output power[29,39-44]. This makes them ideal for integration into everyday items, allowing them to capture energy from human movements and various mechanical sources[45-51]. Despite their advantages, TENGs encounter challenges including high impedance, pulse output, ignition risks, and potential friction damage, due to charge accumulation[52-57]. These issues underscore the need for continued research and development in this promising area of mechanical energy harvesting.
2.2 Piezoelectric Nanogenerator
Figure 1B showcases the piezoelectric nanogenerator (PENG), a device designed to exploit the piezoelectric effect, converting mechanical stress into electrical charges. This effect was first discovered by Curie in 1880 and has gained recognition as a source of renewable energy and a means to power active sensors[58]. The piezoelectric effect is triggered when piezoelectric material experiences external mechanical stress or pressure, causing it to deform[49,59-62]. This deformation leads to a shift in the negative and positive charge centers, leading to polarization in the piezoelectric materials. When the external stress is removed, the material returns to its original, neutral state. Piezoelectric materials are divided into two main categories: organic and inorganic. Inorganic piezoelectric materials, including quartz[63-65], ZnO[66-69], barium titanate (BaTiO3)[70-72], and lead zirconate titanate (PZT)[73-75], are noted for their strong piezoelectric responses. On the other hand, among organic ones, PVDF is distinguished by its significant piezoelectric coefficient, stability, and fabrication ease[76-81]. The development of piezoelectric composites by Newnham in 1978 was a breakthrough, providing enhanced piezoelectric and mechanical properties and more design flexibility than single-material ceramics[82]. These composites have grown increasingly important across various sectors, such as aerospace[83,84], healthcare[85-88], and advanced electronics[89-92], highlighting the significance of PENG as sophisticated smart materials. However, PENG encounter several challenges that affect their efficiency and practical application as energy sources. These issues include pulse-like output, relatively high impedance and low energy conversion efficiency[53,93]. By overcoming these obstacles, the performance of PENG can be enhanced, broadening its application in the development of renewable energy technologies and self-powered sensing systems.
2.3 Electromagnetic Generators
Electromagnetic generators (EMG) exemplify the use of electromagnetic induction to transform mechanical motion into electrical energy (Figure 1C). This process is rooted in Michael Faraday's groundbreaking discovery of electromagnetic induction in 1831[94]. At the heart of its operation is the interaction between coils and a magnet. The magnetic field changes with the relative position alternation of magnet and coils, altering the magnetic flux through the coil, which induces an electromotive force within coils, generating electricity.
EMG are known for their high efficiency in energy conversion, capable of producing large currents at relatively low voltages while exhibiting resistive impedance[95-98]. These features make EMG reliable and robust power sources, maintaining performance despite environmental changes such as temperature fluctuations, humidity, and friction[52,99]. Their durability and versatility are valuable in renewable energy systems, including wind and hydropower, where they convert natural kinetic energy into electrical power[100,101]. Furthermore, EMG play a crucial role in biomechanical energy harvesting by utilizing the negative work produced during human movement[102]. It occurs during deceleration phases of motion, where kinetic energy is typically lost. Although EMG hold promise for energy-harvesting applications, their practical use in personal energy systems is limited by challenges related to their size, weight, and complexity[103]. Inertial induction-type EMG, which belong to a specific category that cuts magnetic motion, harnesses the relative inertial motion between permanent magnets and coils to induce electromagnetic induction and generate current. The effectiveness of this technique depends on the relative linear velocity between coils and the magnet, with areas of high linear velocity, such as the lower limbs or arms[104], being optimal for application.
2.4 Broad Spectrum of Mechanical Energy Harvesters
The quest for sustainable energy solutions has led to the development of various mechanical energy harvesters, each employing different physical principles to convert mechanical movements into electrical energy. The vibration energy harvester leverages the work function difference between two materials to generate electricity (Figure 1D)[105]. This approach, reminiscent of an electrostatic harvester, eliminates the need for external power sources or electrets, enhancing energy autonomy and streamlining the energy conversion process. Figure 1E presents the capacitive harvester, which uses a variable capacitive structure to transform the relative motion between plates into electrical charges[106,107]. This method is crucial for miniaturized systems and can be adapted into a range of designs, such as in-plane gap closing and overlap converters, underscoring its versatility and importance in developing compact energy harvesting solutions. Figure 1F illustrates a mechanically assisted thermoelectric generator that utilizes the Seebeck effect to convert thermal energy into electrical energy. This effect is based on the principle that a voltage is generated across a thermoelectric material-comprising p- and n-type semiconductors connected in series-when there is a temperature differential. The mechanical energy could be converted to generate or sustain the temperature difference. Such a generator allows for converting thermal energy, which could originate from industrial waste heat or biological sources, into valuable electrical power[108-111]. Distinct from piezoelectric or triboelectric generators, which depend on mechanical deformation or contact-induced charge generation, the thermoelectric generator capitalizes on thermal gradients. This offers an alternative energy conversion pathway in scenarios where heat is abundantly available, presenting a versatile solution for sustainable energy capture[112]. Hydrodynamic harvester harnesses kinetic energy from fluid flows (Figure 1G)[113], which is effective in converting mechanical energy from environmental water flows into electrical energy, providing a viable option for energizing devices in water-rich environments. Figure 1H showcases Boland’s electret generator prototype to directly convert mechanical energy into electricity[106,114]. This generator employs an electret, a material known for its ability to maintain a quasi-permanent electric charge. Specifically, Boland's prototype capitalizes on the relative rotational motion between two components: the upper plate and the stator, to generate electrical power.
2.5 Advances with Soft MEGs
Transitioning to soft MEGs heralds a significant advancement in the domain of mechanical energy harvesting. These devices exploit the giant magnetoelastic effect inherent in soft magnetic elastomers, converting biomechanical energy directly into electricity. This pioneering approach effectively surmounts the limitations associated with traditional EMGs by delivering enhanced energy conversion efficiency and ensuring a harmonious integration with human biomechanical processes, thereby addressing the issue of mechanical moduli mismatch between the generators and human tissues. Soft MEGs feature a dual-layer design comprising magnetic induction and magnetomechanical coupling layers[15-17,115]. This configuration allows MEGs to efficiently convert both mechanical and magnetic fluctuations into electrical energy, ensuring compatibility with the delicate mechanics of human tissue. Figure 1I and 1J illustrates the operational principle of the MEG device, where mechanical deformation alters the material's magnetic properties[20,24,115,116]. The integrated layers synergistically transform mechanical and magnetic variations into electrical output, minimizing stress and rendering the MEG suitable for soft interfacing applications with the human body. Figure 1K and 1L showcases the dynamic behavior of a magnetoelastic sphere generator under external force applied during bicycle vibration energy harvesting. This interaction showcases how contact with a glass ball induces a change in the material's magnetic alignment and flux density, attributed to the reorientation of magnetic dipoles from the elastomer's resting to its compressed state under stress[24,117]. This allows for the conversion of mechanical pressure into electrical signals, enhancing sensor technologies that demand precise force detection and differentiation for improved control.
Soft MEGs distinguish themselves through their unparalleled flexibility, high energy conversion efficacy, and compatibility with human biomechanics, heralding a paradigm shift in renewable energy systems, notably in wearable and implantable healthcare devices. By offering a viable and sustainable alternative for battery-powered devices, soft MEGs exemplify the potential for transformative impact on bioelectronics, enabling continuous, battery-free operation of critical healthcare devices, thereby reshaping patient care and quality of life. By paving the way for the development of sustainable and autonomous solutions in bioelectronics, soft MEGs signify a stride in wearables and medical implants.
3 TRADITIONAL ELECTROMAGNETIC DEVICES
Traditional electromagnetic devices encompass essential components like coils, inductors, and MEGs, including materials such as metal, Terfenol-D, Galfenol, and metglas (Figure 2). These devices are pivotal for actuation, energy conversion, or efficient power transfer, yet face challenges in flexibility and integration with wearable and soft electronics due to their rigidity.
|
Figure 2. Comparative Overview of Traditional Electromagnetic Devices and Soft Systems. On the left (highlighted in yellow), the utilization of traditional rigid electromagnetic devices, including Terfenol-D and Galfenol in transportation actuators. Reproduced from Ref.[123] with permission from ASME; and single-cell capture. Reproduced from Ref.[124] with permission from Wiley; metglas transformers in biomedical sensors. Reproduced from Ref.[125] with permission from MDPI, under a Creative Commons CC BY license; and medical implants. Reproduced from Ref.[127] with permission from Springer Nature, under a Creative Commons CC BY license; coils and inductors in transcranial magnetic stimulation (TMS) and wireless power. Reproduced from Ref.[122] with permission from MDPI, under a Creative Commons CC BY license. On the right (highlighted in green), applications of soft magnetic elastomers, magnetorheological elastomers, and soft magnetic textiles. Reproduced from Ref.[15,18,130] with permission from Springer Nature. Images of soft actuators, targeted therapy delivery, adaptive damping vibration control in enhancing optical coherence tomography (OCT) imaging, wearable health monitoring, electricity supply. Reproduced from Ref.[18,131,132,146,147,158], respectively with permission from Wiley, Elsevier, Optica, and Springer Nature.
3.1 Metal-based Electromagnetic Components: Coils and Inductors
Coils and inductors are pivotal components in many electromagnetic applications, owing to their high magnetic permeability and excellent corrosion resistance[118]. These characteristics make them indispensable for generating precise and stable magnetic fields. Consequently, they find widespread use in electronic circuits and medical technologies, notably in transcranial magnetic stimulation (TMS) systems that utilizes controlled magnetic pulses for neural stimulation[119-121], benefiting from the stability and precision offered by Ni-based components. Additionally, their durability and high efficiency render them ideal for wireless power transfer systems that depend on robust and stable magnetic coupling to transmit energy effectively without the need for physical connections[118,122]. However, their relative rigidity complicates integration into wearable devices, which necessitate materials that can endure repeated mechanical stress.
3.2 Rigid MEGs
Rigid MEGs generally utilize magnetostrictive materials and amorphous metal alloys to convert magnetic energy into mechanical energy and vice versa. These devices leverage the unique properties of materials such as Terfenol-D, Galfenol, and metglas, facilitating actuation and efficient energy conversion across diverse applications.
3.2.1 Magnetostrictive Materials: Terfenol-D and Galfenol
Terfenol-D and Galfenol stand as pivotal magnetostrictive materials, known for their ability to undergo significant dimensional changes when subjected to magnetic fields[123,124]. Their high magnetostrictive coefficients renders them invaluable for applications that demand precise control over movement, such as in vibration damping systems and precision positioning devices utilized within the automotive and aerospace sectors. In precision actuation, where exact and controlled movements are crucial, the sensitivity of Terfenol-D and Galfenol to magnetic fields facilitates micro-scale manipulations. This is advantageous in medical technologies, like actuators designed for the delicate task of single cell capture without inflicting damage[124].
Despite these advantages, their integration into mechanical energy harvesting systems is hindered by several factors. The intrinsic rigidity of these materials leads to devices that lack the compliance necessary for seamless integration with the low-modulus and dynamic nature of soft- electronics and biological systems. The high mechanical stress required to induce deformation necessitates robust, often sizeable magnetic field generation systems, contradicting the trend towards miniaturization in contemporary electronics. Furthermore, their dependence on rare earth elements detracts from sustainability and cost-effectiveness. Therefore, while Terfenol-D and Galfenol find potential applications requiring precise actuation, such as in specialized biomedical devices, their adoption in mechanical energy harvesting, where flexibility and gentle interaction with human tissues are critical, remains limited.
3.2.2 Amorphous Metal Alloys for Energy Efficiency: Metglas Transformer
Metglas, an amorphous metal alloy, is distinguished by its non-crystalline structure, endowing it with exceptional magnetic properties such as low hysteresis loss[125]. This property is crucial in transformer cores to ensure energy-efficient magnetic field induction and reversal, minimizing power loss. Additionally, the high electrical resistance of metglas reduces eddy current losses, enhancing its effectiveness in transformers and other power conversion devices. Its high magnetic permeability and low coercivity make it suitable for precise current and magnetic fields sensors in biomedical applications[125]. These current sensors are integral to medical devices, where accurate detection of bioactivity is essential for administering precise, life-saving treatments and stimulations[126,127] ,while the magnetic field sensors are utilized for applications requiring accurate motion detection[128,129], such as in advanced prosthetics and robotic surgical instruments. Its high energy efficiency is also evident in inductive charging systems for biomedical implants, where its low energy dissipation during charging cycles ensures safe and effective power transfer from the primary to the secondary coil with minimal loss for high-frequency electronics. This minimizes the risk of excessive heat generation, crucial for the longevity of the implant and the safety of surrounding tissues[127]. Meanwhile, its low coercivity is advantageous in electromagnetic shielding, protecting sensitive electronic components from interference, thereby preserving signal integrity and device reliability.
However, despite these advantages, metglas faces limitations in flexible and wearable integration due to its brittleness and the complex manufacturing process it requires. Additionally, the amorphous structure, beneficial for static applications, presents limitations in direct conversion of mechanical motion into electrical energy needed in harvesting ambient or human movement. The lack of a crystalline structure and the random arrangement results in an absence of predictable deformation and stress distribution patterns that are critical for efficient energy conversion through piezoelectric effects or magnetostriction, diminishing their capability to convert mechanical energy from ambient movements or human activity into electrical output. Therefore, while metglas stands as a cornerstone in enhancing efficiency in energy conversion and transfer applications, its limitations in mechanical energy harvesting highlight the necessity for alternative materials better suited to dynamic energy conversion and the demands of wearable technology.
4 SOFT MAGNETIC SYSTEMS
Soft magnetic systems combine the flexibility of elastomeric matrices with the dynamic response of magnetic particles to excel in converting mechanical energy into electrical energy (Figure 2)[15,18,130]. These systems, in the fusion of soft materials with magnetic functionalities, are designed to adapt to mechanical stimuli, making them ideal for various sustainable energy harvesting applications, from health monitoring to energy-efficient damping, and for pioneering wearable technologies that offer enhanced comfort and functionality.
4.1 Magnetorheological Elastomers
Magnetorheological elastomers (MREs) consist of non-ferrous magnetic particles, such as carbonyl iron, dispersed in a polymer matrix like silicone or polyurethane rubber[130-133]. This combination allows the material to form chain-like structures when exposed to magnetic fields, altering the elastomer's properties. This adaptability provides a range of stiffness and damping capabilities that can be precisely adjusted with magnetic stimuli. MREs combine the reactive nature of magnetic particles with the flexibility of elastomeric compounds to undergo dramatic changes in mechanical properties like stiffness, damping, and viscoelasticity under magnetic influence[131]. These changes are immediate, making MREs adaptable for diverse applications from adaptive systems to soft robotics[134,135]. Unlike soft magnetic elastomers that are designed for fast magnetic response, MREs are optimized for significant and adjustable changes in mechanical properties. This distinction underpins their utility in various domains, including vibration damping in optical coherence tomography (OCT) for clearer imaging[132], adaptive damping in vehicles for improved comfort, and customizable support in prosthetics and orthotics for enhanced patient care. Additionally, MREs offer nuanced haptic feedback in virtual reality[136-139] and robotic surgery[140-142], improving user experience and precision. The manufacturing process of MREs, which can produce both isotropic and anisotropic configurations, benefits from soft materials engineering like 3D printing[143-145], which allows for the creation of MREs with complex shapes and tailored properties, meeting specific needs across various fields.
4.2 Polymer-based Soft MEGs
Soft MEG signify a notable advancement in energy harvesting technologies, leveraging the unique combination of soft magnetic particles embedded within a flexible elastomer matrix[22,146-148]. The integration not only imbues these materials with mechanical compliance but also magnetic activity, enabling rapid magnetization changes. This dynamic responsiveness is pivotal for applications requiring immediate adaptation to magnetic stimuli, allowing for converting mechanical vibrations, movements, or pressures into electrical energy, making them suited for diverse applications. From powering small electronic devices and sensors[22,146,147] to contributing to the energy efficiency of large systems, such as wind energy[23] and wave vibration energy harvesting[24], soft MEG hold the promise of harnessing energy from the environment, reducing reliance on traditional power sources. Meanwhile, their vibration damping capabilities are essential in environments and medical devices where operational noise should be minimized to avoid disruptions or adverse effects on patient care and sensitive experiments[49,149]. By converting vibrational energy into electrical energy, soft MEG not only reduce noise but also contribute to energy efficiency. The sensitivity of soft MEG to magnetic fields makes them ideal for creating sensors that can convert mechanical stress into electrical signals for health monitoring[15,49,115,150], structural integrity assessments[49,151,152], and environmental sensing[49,116]. This capability enables the development of non-invasive, continuous monitoring systems that offer precise insights into various parameters, from physiological health metrics to structural conditions, with remarkable accuracy. Moreover, the application of soft MEG in targeted therapy delivery[20,150,153] showcases their potential in medical advancements. By utilizing their magnetic field responsiveness, these generators can be directed to specific areas within the body, allowing for the localized generation of electrical energy. This approach can be harnessed to stimulate tissues[154] or power implantable medical devices[15,115], providing a new avenue for treatment modalities. Transitioning to soft MEG represents a significant leap forward from traditional rigid MEG energy harvesting. Compared with rigid MEG, their capability to transform mechanical energy into electricity in sensitive and demanding applications opens up new possibilities for sustainable mechanical energy harvesting for improved healthcare outcomes through advanced medical devices and treatments.
4.3 Textiles-based MEGs
Soft magnetic textiles represent a significant advancement in fabric technology, transitioning from conventional clothing to multifunctional self-powered bioelectronics that integrate with energy harvesting, health monitoring, and various technological applications[16-18,155]. Merging the comfort and utility of traditional textiles with the advanced functionality of magnetic responsiveness, soft magnetic textiles are generally created through two primary methods: weaving magnetoelastic fibers into fabrics and yarns[18,156] ,or affixing thin, soft magnetic patches onto fibers[16,17]. This integration maintains textiles' inherent lightness, softness, and breathability while introducing the capability to generate electricity[157], signifying a significant leap towards self-sustaining electronic systems centered on human activity and well-being. Constructed from one-dimensional soft fibers that exhibit a notable magnetoelastic effect[18], these textiles engage magnetic dipoles under mechanical stress, offering a magnetomechanical coupling that surpasses that of traditional metal alloys. By combining soft magnetic particles, such as carbonyl iron, with polymers like silicone or polyurethane rubber, these textiles achieve a mechanical compatibility with human skin.
Soft magnetic textiles are distinguished by their humidity resistance[22,156], an essential feature for wearable devices in active or aquatic environments, ensuring durability and comfort without compromising performance. They operate through a dual-phase mechanism that converts mechanical stress into magnetic changes and then transforms these magnetic alterations into electrical energy[17,18,49,158]. This process, independent of external magnetic fields, can generate a short-circuit current density of ~0.6mA·cm−2[18], establishing a strong foundation for energy harvesting. In wearable health monitoring, these textiles are revolutionizing the way devices track cardiovascular metrics by converting arterial pulse waves to electrical signals, a function invaluable during intense physical activity[17,18,49,159-161]. Further integration with mobile applications facilitates seamless health data collection and analysis, heralding a future where wearable technology is a cornerstone of preventive medicine and daily health management.
Beyond health monitoring, soft magnetic textiles act as a portable and renewable electricity source. By harvesting energy from human movement, they can power small electronic devices[16,17,159,162], promoting sustainability and independence from traditional power sources. This soft magnetic electronic textile is vital for the evolution of self-sufficient wearable technologies. Additionally, the generated magnetic fields have therapeutic benefits, offering non-invasive methods for pain relief and tissue healing[156,159,160], potentially leading to medical garments that not only monitor health but also actively enhance physical well-being. Moreover, in a time when electronic interference is a concern, these textiles provide electromagnetic shielding[159,163], protecting both the wearer and sensitive electronic devices, an increasingly relevant feature in both personal and professional realms. Soft magnetic textiles are redefining wearable technology by combining traditional fabric comfort with the pioneering capabilities of material science. Through their roles in wearable health monitoring, electricity generation, therapeutic magnetic field generation, and electromagnetic shielding, these textiles pave the way towards a future where our clothing keeps us connected, powered, and healthy.
5 PRINCIPLES OF SOFT MAGNETOELASTICITY
Soft magnetoelasticity merges magnetism's physical laws with the mechanical properties of soft materials, enabling energy harvesting. Compared to traditional materials like Terfenol-D and Galfenol, soft magnetoelastic systems offer several advantages: improved magnetomechanical coupling for efficient energy conversion, lower Young's modulus for flexibility, operation without external magnetic fields, and durable magnetic properties under mechanical stress (Table 1)[15,18,115]. By optimizing particle size, porosity, and matrix flexibility, these systems efficiently convert mechanical motion into energy, paving the way for advancements in wearable technology and soft robotics. A comparative summary of magnetic properties of soft magnetoelastic elastomers versus traditional rigid magnetoelastic systems is provided in Table 2.
Table 1. Performance Characteristics of Various Magnetoelastic Generators
System |
Energy Harvesting Efficiency (W m-2) |
Mechanical Modulus (kPa) |
Stretchability |
Sensing Rangea |
Magnetomechanical Coupling Factor (T Pa-1) |
Fe3O4 soft MEG |
~0.38 |
303.06 |
107.27% |
High (III) |
1.35×10-9 |
SrFe12O19 soft MEG |
~2.97 |
834.48 |
36.53% |
High (II) |
1.06×10-8 |
NdFeB soft MEG |
20.17 |
692.23 |
189.22% |
High (I) |
7.19×10-8 |
Terfenol-D |
~3.82 |
1.2˟108 |
No |
Low (IV) |
1.36×10-8 |
Galfenol |
~10.80 |
7.6˟107 |
No |
Low (V) |
3.85×10-8 |
Notes: aThe order (I-V) is from the highest to the lowest sensing range.
Table 2. Comparative Analysis of Soft Magnetoelastic Systems Versus Traditional Electromagnetic Devices
Parameter |
Description |
Application |
Analysis |
Comparison |
Wavy chain |
Adjustments of magnetic dipoles in response to forces, explaining soft magnetoelasticity |
Used to understand energy conversion in magnetoelastic systems |
Magnetomechanical coupling factor (d33) revealing efficiency: higher values mean better conversion of mechanical to magnetic energy |
More adaptable to mechanical stimuli than traditional rigid materials like Terfenol-D or Galfenol |
Magnetostriction (λ) |
Magnetization change inducing dimensional changes |
Central to converting mechanical to magnetic/electrical forms in soft magnetoelastic systems |
Large λ values indicating a strong magnetoelastic response, crucial for effective energy harvesting. |
More pronounced effect at lower stress levels compared to traditional materials, which require higher stresses |
Magnetic flux density (B) |
B decrease owing to deformation under stress |
Efficient at converting mechanical to magnetic/electrical energy |
A higher d33 resulting in a greater decrease in B, signaling higher energy conversion efficiency |
Higher sensitivity to stress-induced magnetic property changes than traditional systems |
Coercivity (Hc) |
Response of B to applied uniaxial stress varies |
Useful in actuators requiring precise deformation control |
Lower Hc allows for more significant magnetoelastic effects under less stress |
Lower Hc in soft MEG systems |
Output power (Pout) |
Optimal load resistance for maximum voltage output and efficient energy conversion |
Key in applications where mechanical-to-electrical energy conversion is essential |
Peak output power occurs at resonance frequency; lower internal resistance improves efficiency |
Traditional materials may not optimize resonance and internal resistance as effectively due to rigidity |
Size and porosity (Ө) |
Size and porosity influencing magnetoelastic response and energy conversion efficiency |
Material design optimization in magnetoelastic systems |
Generally, smaller size enhances magnetic sensitivity. However, this trend reverses below a certain size threshold due to superparamagnetism. Lower porosity often results in stronger magnetic responses by increasing the density of magnets. An optimal porosity is necessary to balance magnetic performance with mechanical properties like flexibilitya |
Traditional devices may not exhibit such sensitivity to size and porosity due to less flexible structures |
Output voltage (V) |
Voltage output in response to deformation |
Vital for devices experiencing quick and diverse movements. |
The output voltage is primarily driven by mechanical strain, which induces changes in magnetic flux through the realignment of magnetic dipoles. Operating near the generator's resonance frequency maximizes voltage by enhancing energy transfer efficiency. The coil geometric design and the alignment of magnetic particles optimize magnetic responsiveness for high voltage outputs. |
Traditional rigid magnets might produce more abrupt voltage changes with higher initial current but less sustainability |
Frequency (f) |
Frequency-dependent response |
Effective conversion at certain frequencies, likely at resonance |
Devices more effectively convert mechanical energy into electrical energy at specific frequencies |
Traditional systems may not have such a pronounced frequency-dependent response due to less dynamic material properties |
Notes: aMicromagnets might generally offer advantages over nanomagnets due to reduced thermal fluctuation effects and potentially more stable magnetic properties.
5.1 Wavy Chain Model
Central to understanding the enhanced performance of soft magnetoelastic materials is the wavy chain model, which elucidates how mechanical stress influences their magnetic properties, thereby optimizing energy conversion[15,18,115]. In this model, the alignment and distribution of magnetic particles within a soft matrix are conceptualized as a series of wavy chains. When mechanical stress is applied, these chains undergo deformation, leading to a significant alteration in the material's magnetic flux density. Figure 3A shows the giant magnetoelastic effect, where mechanical stress on magnetostrictive materials leads to a significant realignment of magnetic domains[15]. The wavy-chain model adeptly describes these orientation adjustments of magnetic dipoles in response to mechanical forces, distinguishing it from traditional magnetoelastic effects observed in alloys such as Terfenol-D and Galfenol[15]. In magnetoelasticity, the magnetomechanical coupling factor (d33) indicates the conversion efficiency between magnetic and mechanical energy. A simplified equation relates the change in elastic energy density (ΔE) to magnetization (M) and magnetic field strength (H) as[164]:
$$ \Delta E=\frac{1}{2}{d}_{33}MH\left(1\right)$$
where ΔE represents the change in elastic energy density due to a magnetic field that is applied, with d33 measuring the strain change induced by a magnetic field or the alteration in magnetization resulting from applied stress. Permeability (μ), linking magnetic field (H) to magnetic susceptibility (χ) and magnetic flux density (B), indicating magnetization response to an applied magnetic field, further elucidate the material's behavior under stress[165]:
$$ {B}={\mu }{H}\left(2\right)$$
$$ {M}={\chi }{H}+{{d}}_{33}{\sigma }\left(3\right)$$
|
Figure 3. Overview of Soft Magnetoelasticity Featured Properties and Energy Conversion Mechanism. A: Wavy-chain model for stress-induced effects: A wavy-chain arrangement with varying horizontal and vertical spacings in relaxed and compressed states. B: Magnetoelastic response to stress: Simulations of magnetic flux density across an elastomer under 0 and 300kPa pressures. C: Material-specific magnetic behavior: Variations in magnetic flux density in response to uniaxial stress in different magnetic materials, NdFeB, SrFe12O19, and Fe3O4. D: Magnetoelastic behavior: Comparison of magnetoelastic behaviors across systems under compressive stress, showing relative flux density alterations (ΔB/B0). E: Magnetoelastic effect: Comparative experiments highlighting the substantial magnetoelastic effect in soft magnetoelastic generators. F: Magnetic flux and magnetization: M, magnetization versus applied magnetic field curves for hard and soft magnetic materials.G: Energy harvesting efficiency: Dependency of output current and voltage on the load resistance for dual-layer soft magnetoelastic generators. H: Magnetic particle size effect: Variations in relative magnetic flux density in soft magnetoelastic generators containing nano- and micro-magnets. I: Porosity effect: Absolute flux density changes in soft systems with varying porosity levels. J: Stress-porosity effects: Magnetic flux density under stress, comparing layers with porosities of 0.477 and 0, at 0, 139, and 278kPa. K: Voltage response to bending: Typical open-circuit voltage response curve upon bending deformation. L: Frequency dependent outputs. A, C-E, G-K, Reproduced from Ref.[15] with permission from Nature. B, Reproduced from Ref.[16] with permission from American Chemical Society. L, Reproduced from Ref.[156] with permission from Elsevier Inc.
The equations illustrate the intricate relationship between magnetic and mechanical properties, highlighting the linear initial response in soft elastomers. However, the real complexity emerges when considering non-linear effects and the interaction among various parameters[166]. Further optimization of the model views nanomagnets as magnetic dipoles aligned in a wavy chain structure after magnetization, characterized by vertical (h) and horizontal (l) distances between particles[18]. When compressive stress is applied, this wavy chain structure transforms, altering dipole-dipole interactions and the demagnetizing field, resulting in a reduction of magnetic flux density. This change is supposed to be reversible; removing the stress restores the original magnetic flux density and the wavy chain structure. The model quantitatively relates magnetic field variations to mechanical stress using the equation[18]:
$$ \frac{{H}_{1\perp }}{{H}_{0\perp }}\approx \frac{\frac{1}{\chi }-\frac{{d}_{33}}{2a{\lambda }^{1.5}+1}+\frac{{r}^{3}}{3{\lambda }^{3}{ℎ}^{3}}\left(0.3006-f\left(\frac{l}{ℎ{\lambda }^{1.5}}\right)\right)}{\frac{1}{\chi }-\frac{{d}_{33}}{2a+1}+\frac{{r}^{3}}{3{ℎ}^{3}}\left(0.3006-f\left(\frac{l}{ℎ}\right)\right)}\left(4\right)$$
where H1⊥ and H0⊥ depict the vertical magnetic fields in the presence and absence of mechanical pressure, respectively. The equation incorporates variables such as r for nanomagnet radius, χ for remnant magnetization, a for the wavy chain’s aspect ratio, l and ℎ for horizontal and vertical distances between dipoles, and k for the effect of nonideal factors like neighboring chain interactions, with λ indicating the principal stretch linking mechanical deformation to magnetic response. This equation correlates the magnetic field variation to applied stress, showcasing how mechanical compression alters the magnetic flux density-a cornerstone for understanding the principles behind energy conversion in these systems. Empirical evidence supports the model, particularly with parameters like r=2.5μm, l=14.85μm, ℎ=13.5μm, a=105, χ=14.99, and d33=3.8, aligning closely with experimental data for compressive stress up to 450kPa[18]. This agreement demonstrates the model's precision and the significant magnetic field change (~18mT) induced by mechanical stress in soft magnetic fibers (Equation 4).
This conceptual framework, often referred to as the wavy chain model, provides an insightful perspective on how mechanical stress impacts magnetic properties in soft elastomers. The alteration in magnetic flux density induced by mechanical stress, as described by this model, enables the generation of a substantial electromotive force, even under low levels of stress. This property is advantageous for applications in energy-harvesting wearables and implantable medical devices, where efficient energy conversion is paramount for device functionality and longevity.
The biocompatible and mechanically compliant nature of soft magnetoelastic materials plays a crucial role in their interaction with human tissues. Unlike their rigid counterparts, these materials can mimic the flexibility and elasticity of biological tissues, ensuring seamless integration and minimizing the risk of irritation or adverse reactions. This compatibility not only enhances the comfort and wearability of bioelectronic devices but also maximizes the efficiency of energy harvesting from biomechanical movements, addressing two critical aspects of bioelectronics design: energy conversion efficiency and tissue compatibility. It underscores the potential of utilizing mechanical-magnetic energy conversion with high efficiency in soft MEGs for applications in thermal management, optical modulation, and energy generation.
5.2 Magnetostriction (λ) for Energy Harvesting in Soft Magnetoelasticity
Magnetostriction in ferromagnetic materials, pivotal for mechanical energy harvesting in soft magnetoelastic systems, emerges from the principle that a change in magnetization induces dimensional changes to optimize the system's energy[167]. This phenomenon, central to transforming mechanical energy into magnetic and electrical forms, progresses through several key mechanisms: internal domain rotation and magnetic domain wall movement under external fields, modify material dimensions and enable the conversion of mechanical stress into magnetic energy[168,169].
Stemming from demagnetization energy considerations, materials elongate along their magnetization direction to reduce energy losses, influencing the efficiency of energy conversion based on the material’s geometrical configuration. Meanwhile, itinerant electron model reveals the behavior of magnetic 3d electrons, whose energy states are shaped by the crystal's symmetry[170], shedding light on how spontaneous magnetization aligns with crystal axes, a factor critical for optimizing energy harvesting. Furthermore, for rare earth metals-based soft magnetoelastic materials, the unique anisotropy of 4f electron orbitals lead to substantial lattice distortions and enhanced magnetoelastic effects, boosting energy harvesting capabilities[169]. These theoretical perspectives provide a comprehensive view of the mechanisms driving magnetostriction, crucial for the development of soft magnetoelastic materials aimed at mechanical energy harvesting. By detailing the interplay between mechanical deformation and magnetic field variations, these insights inform the design and optimization of devices that convert mechanical movements into usable electrical energy, highlighting the potential of soft magnetoelasticity in sustainable energy solutions.
5.3 Pressure-Induced Reduction of Magnetic Flux Density (B)
Soft magnetoelastic systems, due to their inherent flexibility, undergo more substantial mechanical deformation under stress compared to stiffer materials. This leads to a more significant realignment of magnetic domains and a pronounced decrease in magnetic flux density (B)[16,18,20], making these materials effective at converting mechanical energy into magnetic or electrical energy. This attribute renders them highly suitable for sensors and energy harvesting devices. Figure 3B showcases simulations that model the magnetoelastic response of an elastomer to varying pressures with COMSOL Multiphysics[16]. These simulations show the decrease in magnetic flux density with applied mechanical pressure, highlighting the material's ability to convert mechanical disturbances into magnetic variations. This core phenomenon of the magnetoelastic effect facilitates a coupling between magnetic and elastic properties, where mechanical stress prompts a realignment of magnetic domains, reducing net magnetization and magnetic flux density. The efficiency of this interaction is quantified by the magnetomechanical coupling factor (d33), measuring the strength of interaction between mechanical and magnetic states. To quantitatively describe this behavior, an empirical model considers the initial magnetic induction without stress (B0), the impact of stress (σ), and the material's porosity (θ). The proposed equation to capture this relationship is:
$$ B\left(\sigma ,\theta \right)={B}_{0}\left(1-\alpha \theta \right){e}^{-\left({k}_{0}+{k}_{1}\theta \right)\sigma }\left(5\right)$$
where B(σ, θ) represents the magnetic induction at a specific stress level (σ) and porosity (θ); B0 is the baseline magnetic induction without stress, in a non-porous material; α indicates the reduction in B0 due to porosity. k0 is the stress decay constant for a non-porous material. k1 represents the change in the stress decay rate per unit porosity. This model suggests that as stress (σ) increases, magnetic induction (B) decreases. The empirical constants α, k0, and k1 would be determined through model fitting to experimental data, allowing a comprehensive understanding of how mechanical stress and porosity affect the magnetic properties of magnetoelastic materials.
5.4 Contrasting Coercivity (Hc) in Magnetoelastic Materials Under Stress
Figure 3C illustrates the magnetic flux density in relation to applied uniaxial stress across different magnetic materials, including NdFeB, SrFe12O19, and Fe3O4, showcasing the diversity in magnetoelastic responses[15]. NdFeB, a typical hard magnetic material with high coercivity and remanence, exhibits a significant decrease in magnetic flux density with increasing stress[15,115,171]. Mechanical stress leads to a considerable realignment of magnetic domains, impacting its overall magnetization. With lower coercivity than NdFeB, SrFe12O19’s magnetoelastic effect is less pronounced, reflected by a more gradual decrease in magnetic flux density under stress. The magnetic flux density of Fe3O4 is stable under mechanical forces. NdFeB’s characteristics enable it to maintain strong magnetization up to a specific stress threshold, making it suitable for soft actuators where precise deformation control is essential[172-174]. NdFeB-based magnetoelastic elastomers allows for creating components with varied magnetic domain alignments. Magnetic fields-assisted 4D printing anisotropic magnetoelastic elastomers with predetermined magnetic domains, enabling them to undergo specific deformations, such as rolling, twisting, and folding, under a magnetic field, highlighting the potential for complex movements and functionalities in soft magnetic systems[172].
A comparative analysis of magnetoelastic responses, measured by the change in magnetic flux density (ΔB/B0), across different materials under varying stress levels was shown in Figure 3D[15]. For traditional materials such as Galfenol and Terfenol-D, located in the high-stress region of the graph, there's a noted substantial magnetoelastic effect. However, this effect is observed at stress levels beyond what is typical for human biomechanical movements. These materials, while exhibiting significant changes in magnetic flux density, necessitate large amounts of stress to manifest notable magnetoelastic behavior. Conversely, the soft MEG system within the low-stress region, demonstrates a significant magnetoelastic effect at much lower stress levels, aligning with the range of human motion. Soft MEG, with their pronounced magnetoelastic effects at lower stress levels, are efficient at converting biomechanical energy into magnetic energy. The ability of soft MEG to operate effectively under the stress levels associated with human movement highlights their potential for being integrated into devices designed to capture energy from everyday activities for power generation.
Figure 3E contrasts the electrical current output over time between a traditional rigid magnet and a soft MEG[15]. Soft MEG displays a smoother current curve with a more gradual peak, suggesting a sustained current generation over a longer period despite a lower initial current, which is advantageous for prolonged energy harvesting applications, like wearable devices that require continuous adaptation to human motion. In soft magnetoelastic systems, the selection between soft and hard magnetic particles is essential. Hard magnetic materials exhibit a wide hysteresis loop, signaling high coercivity and remanence (Figure 3F). Magnetic elastomers, comprised of metal alloys or ceramic materials such as NdFeB, SmCo, and alnico, showcase significant magnetic hardness. This attribute enables them to sustain large magnetic field even after the removal of the external magnetizing force, attributed to their large remanence (Br) and coercivity (Hc). Soft magnetic materials, on the other hand, show a narrow hysteresis loop, reflecting their low coercivity and remanence. This property allows them to easily magnetize and demagnetize with minor external magnetic field changes, fitting for energy harvesting requiring frequent magnetic field alterations.
5.5. Output Power
Figure 3G highlights the electrical performance of a dual-layer soft MEG in relation to varying load resistances[15]. The voltage profile shows an initial increase against resistance, peaking at an intermediate value before declining at higher resistances, indicative of optimal load resistance for maximum voltage output. Output power, derived from the product of current and voltage, can be calculated to find the optimal power transfer at the point where the resistance of the load matches the internal resistance of the generator. The equation for output power in an MEG is[52]:
$$ {P}_{\text{out}}=\frac{m{\xi }_{st}{Y}_{0}^{2}{\left(\frac{\omega }{{\omega }_{n}}\right)}^{3}}{{\left[1-{\left(\frac{\omega }{{\omega }_{n}}\right)}^{2}\right]}^{2}+{\left(2{\xi }_{st}\frac{\omega }{{\omega }_{n}}\right)}^{2}}\left(6\right)$$
where Y0 : vibration amplitude;
m, the mass of the vibrating system or seismic mass;
ξst: the structural damping factor of the transducer;
ω: the angular frequency of the vibration;
ωn: the natural or resonance angular frequency of the system.
The equation implies that maximum power output occurs when the system is operating at or near the resonance frequency (ω is close to ωn), but actual output will also be influenced by the damping factor. Analyzing voltage and current trends against load resistance demonstrates the MEG's internal magnetic properties, such as resistance and inductance, which are vital for transforming mechanical energy into electricity efficiently. The point of maximum power transfer, where the voltage peaks, is where the MEG's internal magnetic characteristics best match the external load. This analysis underscores the potential of MEG in energy conversion applications, where efficient mechanical energy harvesting is essential.
5.6 Size and Porosity Effects
Exploration of nanoparticle size on magnetomechanical coupling is important to underscore the size effect in magnetoelastic efficiency under stress[15]. Figure 3H shows the efficiency of magnetomechanical coupling in systems utilizing micromagnets and nanomagnets under different levels of uniaxial stress. It reveals that nanomagnets exhibit a more significant decrease in normalized magnetic field strength with increasing stress, indicating their heightened sensitivity to stress-induced magnetic property changes. This sensitivity is pivotal for developing efficient magnetoelastic systems. This observed behavior aligns with basic magnetic principles in materials, where transition metals and certain lanthanides display significant magnetic moments due to unpaired electrons in their d or f orbitals, aligning to form magnetic domains. Nanoscale magnetic particles can manifest single-domain behavior, yielding a net magnetic moment. Nonetheless, excessively small particles may experience superparamagnetism, with thermal fluctuations randomizing magnetic dipole alignment. Surface treatment of magnetic particles is crucial to prevent agglomeration and enhance magnetization efficiency. Approaches such as silane coupling agents or core-shell structuring can improve particle dispersion and functionality[175-179]. For example, NdFeB particles coated with Au or Ag achieve both robust ferromagnetic properties and high electrical conductivity[177-179], suitable for specific magnetoelastic applications.
Figure 3I and 3J further show the effects of porosity (θ) on the magnetic flux density (B) of magnetoelastic materials under uniaxial stress[15]. Materials with varying porosity levels (0, 0.187, and 0.477) respond differently to stress, with lower porosity resulting in higher flux density (Equation 5). This suggests that materials with minimal porosity exhibit stronger magnetic responses when stressed, highlighting the critical role of material design and porosity control in enhancing magnetoelastic system performance. Understanding the interplay between magnetomechanical coupling efficiency and particle size, along with the impact of porosity on magnetic response, is fundamental for advancing soft magnetoelastic generators. These insights facilitate the development of applications ranging from soft robotics to energy harvesting, where precise material property control is essential.
5.7 Output Voltage
Figure 3K shows the voltage output from a soft MEG subjected to bending deformation, highlighting the open-circuit voltage response over time without any load.[15] The voltage curve features spikes corresponding to moments of rapid bending-induced changes in magnetic flux linkage through the MEG's coils, with peaks aligning with maximal deformation, altering the coil's magnetic field. In accordance with Faraday's law of induction, the output voltage (E) is directly proportional to the magnetic flux rate change through the coil, described by the equation:
$$ E\left(V\right)=-N\frac{\Delta \Phi }{\Delta t}\left(7\right)$$
where N is the coil's turn count, ΔΦ denotes the magnetic flux change in webers, and ΔΦ/Δt is the flux change duration in seconds. The voltage fluctuations observed in Figure 3K indicate the MEG's responsiveness to deformation, vital for devices experiencing quick and diverse movements, whose efficiency in converting mechanical deformation to electrical energy underscores soft magnetoelastic systems' potential for powering self-sufficient sensors and energy harvesting devices.
5.8 Frequency Dependence
Figure 3L depicts the output voltage of a soft MEG over time, showcasing how the device responds to different frequencies of applied mechanical stress[156,180]. The amplitude of voltage oscillations varies with the frequency, displaying a frequency-dependent response characteristic. This behavior underscores the MEG capability to convert mechanical energy more effectively into electrical energy at certain frequencies, likely aligning with resonant frequency. At resonance, the mechanical input harmonizes with the magnetoelastic material's natural oscillating frequency, enhancing energy transfer and, consequently, the voltage output. The output voltage's frequency-dependent nature for MEG can be encapsulated by the equation:
$$ V\left(t\right)={V}_{0}\mathit{sin}\left(2\pi ft+\varphi \right)\times \frac{1}{\sqrt{{\left(1-{\left(\frac{f}{{f}_{n}}\right)}^{2}\right)}^{2}+{\left(2\zeta \frac{f}{{f}_{n}}\right)}^{2}}}\left(8\right)$$
where V(t) is the instantaneous voltage at time t, V0 is the maximum voltage amplitude, f is the applied mechanical stress frequency, t is time, and ϕ is the phase difference, fn is the system's natural or resonance frequency, and ζ is the damping ratio. The denominator signifies the damped harmonic oscillator's transfer function, illustrating how the voltage response amplitude varies with the applied stress frequency relative to the system's natural frequency and damping. This equation emphasizes the MEG's nuanced frequency response, pivotal for optimizing energy conversion in applications requiring precise control over material properties and system dynamics.
6 WEARABLE AND IMPLANTABLE SOFT MAGNETOELASTIC BIOELECTRONICS
The integration of soft magnetoelastic materials into the realm of bioelectronics heralds a new era of healthcare, where the seamless convergence of technology and biology can enhance patient care and outcomes. The unique properties of these materials including their high energy conversion efficiency and exceptional compatibility with human tissues, are being leveraged in various healthcare applications, notably in wearable health monitoring devices and implantable medical technologies.
6.1 Wearable Soft Magnetoelastic Textiles
Wearable soft magnetoelastic textiles represent a groundbreaking integration of magnetoelastic materials with textile engineering, transforming the landscape of health monitoring and personal bioelectronics. These textiles, through their dynamic response to mechanical forces, facilitate electrical signal generation via electromagnetic induction. Notably, the design involves embedding micromagnets within a porous polymer matrix (Figure 4A), optimizing the conversion of mechanical energy into electrical energy efficiently[16]. Figure 4B showcases the aesthetic and functional variety of soft magnetoelastic textiles' woven patterns, emphasizing how textile design influences both the visual appeal and electrical performance of wearables[18]. The checkerboard pattern presented may symbolize an array of magnetoelastic elements within the fabric. Each element is poised to enhance the power generation of the textile upon experiencing mechanical forces like stretching, bending, or pressure. The structure of soft magnetoelastic textiles encompasses a conductive yarn layer for electrical signal transmission and energy harvesting, a soft MEG layer made from magnetostrictive material for converting mechanical deformation into magnetic flux changes, and a textile substrate providing structural support and wearability (Figure 4C). An equation based on the Neo-Hookean model for hyperelastic materials, models mechanical stress as a function of deformation,
$$ s=G\left(\lambda -\frac{1}{{\lambda }^{2}}\right)\left(9\right)$$
where s, compressive stress; λ, deformation; G, the shear modulus of the material.
|
Figure 4. Wearable and Implantable Soft Magnetoelastic Bioelectronics. A: Energy conversion mechanism: Diagram of the soft magnetoelastic generator containing micromagnets and a polymer matrix. Reproduced from Ref.[16] with permission from American Chemical Society. B: Mechanical flexibility and energy harvesting capability: Weaving patterns for the wearable soft magnetoelastic textile. Reproduced from Ref.[18] with permission from Springer Nature. C: Textile MEG design: Structure of the textile MEG featuring a magnetoelastic film, an embedded coil, and a fabric base. D: Self-powering capabilities: Scheme of soft magnetoelastic generator as a sustainable energy source for wearable electronics. Reproduced from Ref.[15] with permission from Springer Nature. E: Motion outputs: Current outputs from bodily movements like pulse, finger and knee bending, running and jumping. Reproduced from Ref.[115] with permission from American Chemical Society. F: Wearable health monitoring: Image of the wearable soft magnetoelastic generator to power a standard thermometer. Reproduced from Ref.[15] with permission from Springer Nature. G: Mechanical sensitivity and dynamic response: Sensing signals as a function of varied pressure and frequencies. H: Weight-sensitivity and adaptability for force monitoring: Weight-dependent electrical responses. G and H reproduced from Ref.[17] with permission from Cell Press. I: Implantable energy harvesting: Illustration of the implanted soft magnetoelastic generator for ultrasound energy capture. Reproduced from Ref.[15] with permission from Springer Nature.
The magnetoelastic layer's mechanical property changes under various strain levels and their impact on the generated magnetic field could be predicted by this equation[18] (Equation 9). In magnetoelastic applications, this equation could be used to calculate the stress in a magnetic material that exhibits elastic properties. When these materials are deformed by external forces, their magnetic properties may change-a phenomenon that is exploited in the design of smart materials and systems. Experimental findings and theoretical confirmation show that compressive stress around 100kPa is sufficient to induce a considerable variation in magnetic field, reaching up to ~18mT in the one-dimensional soft MEG textiles[18]. This integration considers the mechanical stress (s) from the Neo-Hookean model, area change (A) due to deformation, and magnetic flux density change (B) as a result of the magnetoelasticity, leading to a specialized equation:
$$ \epsilon =-N\frac{d}{dt}\left({B}_{0}\left(1-\mu s\right)\cdot A\left(\lambda \right)\right)\left(10\right)$$
where N is the coil's turn count; A is the coil area; B0 represents the initial magnetic flux density; μ is the magnetoelastic coupling coefficient; and A(λ) denotes the coil deformed area as a function of stretch ratio λ. This comprehensive design approach allows for the optimization of soft magnetoelastic textiles for various applications, including energy harvesting from body movements and sensing mechanical forces for health monitoring. By considering mechanical-to-electrical energy conversion, soft MEG textiles can be designed to not only offer comfort and conform to the body but also actively engage in energy conversion and data collection, marking a step towards the realization of smart textiles that blend functionality with fashion.
6.2 Wearable Soft Magnetoelastic Elastomers
Wearable soft magnetoelastic elastomers stand at the forefront of health monitoring, offering a versatile technology for precise tracking of physiological signals across diverse biomechanical activities[181-183]. These flexible sensors are adept at detecting varying pressure levels generated by a wide range of movements, owing to their pronounced magnetoelastic effect. Engineered for resilience, they accommodate bending, stretching, and twisting[15,16,115,180], ensuring comfort and continuous, reliable health data collection when worn on different body parts. The design of these advanced sensors capitalizes on the magnetoelastic materials' sensitivity, transforming mechanical deformations from daily activities into electrical signals that reflect various bodily movements. This capability enables the sensors to span a broad sensing range, essential for comprehensive body monitoring and capturing subtle physiological changes.
At the core of wearable soft magnetoelastic elastomers is the fusion of magnetoelastic material sensitivity with the durability needed for everyday wear. Aimed at creating a user-friendly interface, these sensors integrate with the wearer, acting as an extension that translates the body's movements into electrical data[2,150,180]. This integration marks a significant step towards personalized healthcare, enabling ongoing monitoring outside clinical settings and providing individuals with immediate insights into their health. The integration of a soft MEG into wearable electronics exemplifies its dual functionality as both a sustainable energy source and a sensor for various health metrics (Figure 4D)[15]. This dual role is essential for the advancement of self-sustained wearable healthcare devices, capable of powering devices such as electrocardiograms (ECG) and thermometers, while also detecting physiological signals like sweat and pulse. Figure 4E showcases the MEG's sensitivity to physiological movements and signals[115], illustrating its electrical output in response to different bodily activities, including wrist pulses, knee movements, finger bending, and dynamic actions such as running and jumping. This demonstrates the MEG’s ability to capture a wide range of mechanical energies, converting them into electrical signals for health monitoring and energy harvesting from human movements. A practical application of the MEG is highlighted, where it is affixed to a person's arm to power a thermometer, showcasing its applicability in non-invasive health monitoring (Figure 4F)[15]. Figure 4G shows the operational sensitivity of the MEG, showing its current output as a function of applied pressure at various frequencies, indicating its effectiveness in transducing mechanical stimuli into electrical signals across a spectrum of pressures and deformation rates, valuable for continuous health monitoring[17]. Figure 4H illustrates the MEG's response to varying weights[17], demonstrating its fine-tuned sensitivity to pressure changes, which could be useful for detecting subtle physiological changes or for applications requiring precise pressure sensing.
6.3 Implantable Soft Magnetoelastic Elastomers
Figure 4I depicts an implantable soft MEG designed to convert ultrasound energy into electricity, utilizing the magnetoelastic effect within a biocompatible package suitable for subdermal placement[15]. This approach enables the powering of biomedical devices such as pacemakers or biosensors from ultrasound, bypassing the need for battery replacements or external power sources[183-185]. The operation of this MEG is based on ultrasound emitted from an external source that induce mechanical vibrations in the magnetoelastic material upon penetration through the skin. These vibrations result in changes in magnetic flux density, which, in turn, generate an electromotive force across the device's coils by Faraday's induction law (Equation 7). The deformation of the magnetoelastic layer under ultrasound influence modifies the magnetic field experienced by the coils, facilitating electrical energy generation.
Key considerations for this system include ensuring biocompatibility to avoid immune responses, achieving efficient conversion of ultrasound mechanical energy to electrical energy, ensuring durability against constant ultrasound exposure, and integrating effective energy storage solutions[186-188]. Relevant parameters for this technology's success include the ultrasound intensity (I), which impacts the energy available for conversion, and the magnetoelastic material's mechanical properties like shear modulus (G) and Young's modulus (E), affecting deformation response. The magnetoelastic layer's dimensions also play a crucial role, determining the material volume available for energy conversion. A promising avenue for providing a continuous power source for internal medical devices, exploiting the widespread use of ultrasound in medical environments and the energy-harvesting potential of magnetoelastic materials.
7 DIVERSE APPLICATIONS
This section underscores the substantial promise of MEG across a spectrum of applications. From internal medical devices to wearable energy harvesters and sensors, MEG are portrayed as pivotal components in the future of integrated electronics for everyday life and healthcare management (Figure 5).
7.1 Sweat-Resistant Sensor
MEG is a robust, efficient sensor for wearable health monitoring technologies in sweat-prone conditions, whose integration potential into wearable textiles or direct skin application, offering a dependable energy and data source for health monitoring without the hassle of frequent maintenance or moisture-induced damage concerns[15,22,47,189]. Figure 5A showcases a MEG engineered for optimal performance in various moisture levels[22], demonstrating its durability through electrical output comparisons in both dry and wet conditions. This validation underscores MEG’s resilience, a crucial feature for wearable health devices exposed to sweat from physical activities. The MEG remains consistent electrical current output when shifting from dry to wet environments, highlighting its unaffected power conversion capabilities by moisture. This resilience is vital for uninterrupted physiological signal monitoring during activities that induce sweating. Unlike traditional devices that may need protective encapsulation against sweat-potentially compromising sensitivity and functionality-the MEG maintains its performance, making it an ideal self-powered, sweat-resistant biosensor.
|
Figure 5. Soft Magnetoelastic Generators for Diverse Applications. A: Moisture-resilient performance: Magnetoelastic generators in a moist environment. Reproduced from Ref.[22] with permission from Elsevier. B: Tactile sensing: Electrical signal reconstruction from a 16-pixel artificial skin array. C: Tactile signal mapping: Voltage profiles corresponding to tactile inputs from orthogonal channels. D: Magnetic flux density mapping: Visualization of magnetic flux density under a 5-newton force, with stress on a single pixel and dual-channel sensory activation. E: Stress-sensitivity and spatial differentiation: Magnetic flux density changes against shear stress at the center and adjacent areas of a pixel. B-E, reproduced from Ref.[117] with permission from Science. (F,G) Cardiac monitoring: A magnetoelastic sensor on heart, and generated current with heartbeats. Reproduced from Ref.[115] with permission from Springer Nature. Partially created from Biorender.com. H: Muscle activity monitoring: Electrical output from a magnetoelastic patch on the biceps during muscle contraction and relaxation. Reproduced from Ref.[156] with permission from Cell Press. I, the contraction phase of the bicep during tension; II, the relaxation phase during extension. I: Wearable energy harvesting: Diagram of potential biomechanical energy sources on the human body. J: Mechanical stimulation responses: Measurements of three mechanical stimulation types on soft magnetoelastic textile. K: Respiratory monitoring: Integration of textile MEG into medical scrubs for respiratory tracking, capturing distinct respiratory patterns including normal and rapid breathing, and coughing. I-K. Reproduced from Ref.[17] with permission from Cell Press.
7.2 Tactile Feedback
Soft MEG array can also be utilized as a tactile feedback system for sensory applications like artificial skin[49,190-192]. This system demonstrates the ability to detect mechanical stress and convert it into electrical signals, showcasing the potential for intricate tactile feedback applications. Figure 5B depicts a 16-pixel MEG array mimicking artificial skin, capable of reconstructing signals from mechanical interactions[117]. Each pixel generates a voltage response under pressure, enabling high-precision tactile sensing crucial for developing responsive prosthetics and improving human-machine interactions by accurately replicating touch sensations. Figure 5C extends this functionality, illustrating the MEG’s capacity to create detailed tactile maps[117]. This is demonstrated through unique voltage patterns generated by 'writing' letters on the artificial skin, a feature vital for nuanced input user interfaces, such as touchscreens capable of recognizing handwriting or textures. The visualization of magnetic flux density changes in response to force on a single pixel helps elucidate how mechanical stress at varying intensities and locations influences the magnetic field and the MEG's electrical output (Figure 5D)[117]. This dual-channel sensory activation showcases the MEG's ability to distinguish subtle stress differences. Figure 5E presents the relationship between shear stress and magnetic flux density, highlighting the system's differentiated response to central versus edge-applied forces on a pixel.[117] This includes mean values and deviations, showcasing the MEG's sensitivity to spatial stress gradients, essential for detailed pressure profiling in touch-sensitive devices or robotics. This tactile feedback is underpinned by the magnetoelastic effect, wherein mechanical deformation alters a material's magnetic properties. Within the MEG array, the magnetoelastic film in each pixel undergoes stress-induced magnetic flux density changes. These changes are converted into electrical signals, enabling the MEG to capture not only force magnitude but also its direction and distribution, making it suitable for tactile feedback applications requiring comprehensive sensory information.
7.3 In Vivo Cardiac Monitoring
A magnetoelastic sensor was developed for cardiac function monitoring (Figure 5F), suggesting its implantation near the heart to directly measure mechanical movements associated with cardiac phases of expansion and relaxation[115]. The sensor generates an electrical current that rhythmically varies, mirroring the heart's contractions and relaxations (Figure 5G)[115]. This pattern demonstrates the sensor's capability to transduce the heart's mechanical motions into an electrical signal for medical diagnostics. This process involves correlating the cardiac tissue's mechanical stress with changes in magnetic flux density, a typically nonlinear relationship necessitating calibration for precise electrical signal translation. The ability of soft MEG to distinguish between systolic and diastolic heart phases, offering diagnostic potential for heart conditions and post-surgery cardiac tissue health monitoring (Figure 5G). Designing this sensor entails addressing biocompatibility, tissue irritation minimization, and signal stability over time. It must balance sensitivity for detecting heart movements with the durability needed for continuous operation within the body’s dynamic environment. Overcoming these challenges could position the magnetoelastic sensor as a vital tool for in vivo heart health monitoring, providing healthcare providers with real-time data and potentially alerting patients to abnormal cardiac events.
7.4 Wearable Muscle Energy Harvesting
Soft MEG can also be utilized as versatile tools for biomechanical energy harvesting and muscle activity sensing[115,193-195]. Figure 5H showcases the application of MEG in monitoring bicep muscle dynamics during flexion and extension[156]. The electrical output variations align with the muscle's contraction cycles, highlighting its potential in sports Science for performance optimization and medical rehabilitation for tracking recovery. Figure 5I outlines the potential biomechanical energy sources across the human body[17], highlighting the potential of harvesting energy from natural bodily motions, and the integration of wearable MEG to power devices through daily activities. The relationship between applied mechanical energy (e.g., muscle contractions) and the MEG's electrical output, potentially described by the equation:
$$ {P}_{\text{out}}=\xi \cdot A\cdot \left(\frac{dB}{dt}\right)\cdot V\left(11\right)$$
where Pout is the power output, ξ represents a conversion efficiency factor, A is the MEG's area, and V is the volume of the MEG undergoing deformation. This equation reveals that the MEG's power output is directly proportional to the magnetic field's rate of change, which is influenced by the applied mechanical energy, such as muscle movements. Considering the cost of harvest and total cost of harvesting provides insights into the energy efficiency of these devices in practical applications. These metrics, comparing metabolic cost to electrical power output, offer a way to assess the feasibility of incorporating MEG into wearable technology from an energy economy perspective.
7.5 Smart MEG Textile for Non-Invasive Respiratory Monitoring
Integrating MEG into smart textiles facilitates seamless physiological parameter monitoring without interfering with daily routines, offering a non-intrusive medical tool[17,162,196]. Figure 5J depicts the adaptability of textile MEG to various mechanical stresses-compression, bending, and twisting[17]. The voltage and current outputs for each type of mechanical stimulation underscore the MEG's capability to convert diverse mechanical energies into electrical signals, pivotal for developing smart textiles that respond to the wearer's movements and environment. Furthermore, a MEG textile integrated into medical scrubs was developed to monitor respiratory patterns effectively (Figure 5K)[17]. It captures changes in the magnetic field resulting from chest expansion and contraction during various respiratory activities, including coughing, rapid breathing, and normal breathing. The MEG gathers data on respiratory patterns, which can be transmitted to a dedicated application for instant feedback and visualization, proving beneficial for patients with respiratory conditions by enabling real-time monitoring and early detection of potential problems. The MEG operates by transforming the mechanical energy from body movements into electrical signals. The electrical current generated during different respiratory activities reveals distinct patterns corresponding to each breathing type, showcasing the MEG's sensitivity to varying mechanical stresses caused by respiratory movements.
A hypothetical equation that might describe the relationship between respiratory movements' mechanical stress (σ), the change in magnetic flux density (ΔB), and the induced current (I) is:
$$ I=C\cdot \frac{d\left(\Delta B\right)}{dt}\left(12\right)$$
where I represents the induced current, C is a proportionality constant related to the MEG's design and materials, and d(ΔB)/dt denotes the change rate in magnetic flux density due to mechanical deformation from breathing. The constant C, determined through experimentation, would indicate the MEG’s efficiency in converting biomechanical stress into electrical signals. Challenges for this application include maintaining consistent skin contact, minimizing signal interference from external magnetic fields, and ensuring the MEG's durability through regular use and laundering of the medical scrubs. This approach to respiratory monitoring via smart MEG textiles promises for real-time health monitoring, offering potential improvements in patient care and health management.
8 CONCLUSION AND OUTLOOKS
Soft magnetoelastic materials are pioneering advancements in personal healthcare by enabling continuous, non-invasive monitoring and management of health conditions. These materials convert mechanical energy from individual activities into a sustainable power source for both implantable and wearable medical bioelectronics, heralding a new era of proactive and personalized healthcare (Figure 6). Advances in soft MEG, including giant magnetoelastic layers and liquid-metal patterns, promise to amplify soft MEG electrical output. These developments could power wearable bioelectronics via body movements and generate implantable power sources without encapsulation, expanding bioelectronics applications. The future of MEG technology is geared towards creating devices that are not just effective but also safe, comfortable, and convenient for all users.
|
Figure 6. Perspectives and Challenges in Soft Magnetoelasticity for Mechanical Energy Conversion. The potential advancements and current obstacles for, A and B: wearable magnetoelastic generators and C and D: implantable magnetoelastic generators. Images created from Biorender.com.
8.1 Soft Magnetoelastic Wearables for Continuous and Adaptive Health Monitoring
Incorporating soft magnetoelastic materials into wearable technologies marks the beginning of a new era in persistent health surveillance (Figure 6A). These materials enable devices to continuously monitor health metrics, facilitating a proactive healthcare model that dynamically adjusts therapeutic protocols based on real-time health data. This ensures treatments are timely and personalized, catering to individual's evolving health needs. Wearable devices utilizing soft magnetoelasticity provide real-time health data, empowering users with the ability to modulate therapy on demand. These devices can instantly initiate therapeutic actions, optimizing treatment outcomes. Rehabilitation wristbands offering tactile feedback to guide exercises and smart textiles that correct posture throughout the day exemplify the potential for enhancing recovery and promoting musculoskeletal health.
Soft magnetoelastic wearables transcend traditional monitoring methods by providing non-invasive detection and acting as guardians of well-being, continuously conveying vital health information, and alerting users to potential health issues. Their application in pain management through smart patches underscores the depth of personalized care achievable with magnetoelastic technology. Wearable MEG shows great promise in integrating advanced health management into everyday life. By merging continuous monitoring with intelligent analytics, these devices become proactive health management tools, potentially reducing dependency on traditional healthcare services. As wearables increasingly connect with telehealth systems, they promise to redefine healthcare delivery, making comprehensive health management an integral part of daily living.
8.2 Potential Challenges in Wearable MEG
As MEG integrate more into our daily routines, the design of MEG places a premium on ergonomics and comfort[197-199]. Devices are being tailored to the human body, using flexible, breathable materials and slim profiles to improve the user experience and support regular use by being minimally invasive in everyday activities (Figure 6B). Efforts are underway to identify bio-compatible, non-irritating materials to ensure they can be worn continuously without causing any skin allergic reactions or irritation, even for the most sensitive skin types. Meanwhile, compliance and safety of the developed non-toxic soft MEG material are at the forefront of development, with devices undergoing rigorous testing and certification processes to meet regulatory standards and assure users of their dependability and safety. This push towards developing new sustainable composites aims at making MEG wearable and promoting inclusivity.
Enhancing signal clarity is also a focal point. Advances in noise cancellation technology are being made to eliminate environmental interference, ensuring the integrity of data through sophisticated algorithms and materials that mitigate electromagnetic disruptions, which is essential for the accuracy of health monitoring and diagnostics. Ensuring reliability across various environmental conditions is another critical goal. Research aims to make MEG water-resistant, temperature stable, and durable against physical stresses[200], guaranteeing their functionality in diverse settings, from the gym to rainy conditions. Durability considerations are paramount, with MEG being constructed from wear-resistant materials and incorporating self-healing or easily repairable components to withstand regular use. This approach not only enhances device longevity but also contributes to sustainability by reducing electronic waste. Furthermore, improving power management is a key strategy to prolong MEG operation. Innovations in energy-efficient components and the enhancement of energy harvesting techniques are crucial for extending device usability, ensuring MEG can provide continuous service without frequent recharging. User interfaces for MEG are evolving towards greater intuitiveness and personalization, adapting to user interactions to be accessible to a wide audience, including those less tech-savvy[49,201]. The development focuses on age-agnostic, user-friendly interfaces that employ visual, tactile, and auditory feedback, making the devices approachable for a broad demographic. Addressing these key areas, MEG are poised to become an integral part of daily life, transforming personal health monitoring with their capabilities.
8.3 Wireless Implantable energy Harvesting
Implanted MEG are set to transform medical devices by leveraging energy from bodily movements to wirelessly power internal devices[15,202,203]. This advancement eliminates the need for battery replacements through surgical procedures, minimizing invasive procedure risks. Developments in magnetoelastic materials and designs enhance energy conversion efficiency, leading to smaller, more potent, and reliable devices. The use of these materials for targeted drug delivery and neural stimulation demonstrates their impact on treatment methods to achieve precise control for dosing and responsiveness to immediate body needs, reducing side effects and enhancing therapeutic efficacy (Figure 6C)[20,147,204,205]. The advent of magnetically controlled wearable robots introduces a novel dimension to therapeutic devices[146,174,187]. Leveraging magnetic fields' ability to penetrate various media, these robots navigate the body with remarkable precision to deliver targeted therapies in deep tissue. Additionally, magnetically actuated neural interfaces in implanted MEG present new treatments for neurological conditions, offering pain relief, rehabilitation, and treatment without drugs by stimulating nerves or brain regions with magnetic fields[127,206-210]. Orthopedic implants benefit from self-repairing magnetoelastic materials that adapt and heal over time in response to body movement, diminishing the need for revision surgeries and improving implant longevity. Magnetic control is pivotal in surgical robotics, facilitating less invasive procedures with improved safety and efficiency. In the vascular domain, MEG contribute to creating smart stents that dynamically support blood vessels, adapting to the vascular system's changing needs to potentially reduce additional surgeries and enhance long-term intervention success[211]. Integrated with sensors, implanted MEG offer continuous monitoring of vital signs and internal functions, facilitating proactive healthcare through timely interventions. The application of 3D printing technology in fabricating magnetoelastic elements introduces solutions to soft robotics. Molding these elements into deformable shapes enables unique actuation and deformation mechanisms activated under magnetic fields, edging close to fully flexible and soft systems suitable for various applications.
8.4 Potential Challenges in Implantable MEG
For in-vivo implants, ensuring long-term biocompatibility is crucial to prevent adverse reactions from the body's immune response. Researchers are focused on developing materials that remain stable and functional over time, resistant to breakdown or corrosion within the body, to avoid inflammation or reduced effectiveness. Optimizing energy harvesting and power delivery is fundamental for the autonomous functionality of implanted MEG. Meanwhile, miniaturization is key for implanted MEG to comfortably fit within the body without sacrificing functionality. Advances in miniaturization technology are enabling the creation of compact devices that maintain optimal performance, packing more functionality into smaller devices while ensuring energy efficiency (Figure 6D). Furthermore, customizability of treatments and the Exploration of biodegradable materials for temporary interventions highlight the personalization potential of implanted MEG. These materials would naturally decompose after fulfilling their therapeutic role, eliminating the need for surgical extraction.
Integrating MEG with bodily functions presents a significant challenge, necessitating devices to be not only unobtrusive but also in sync with the body's systems. This requires a collaborative effort across biology, material Science, and engineering to devise mechanisms that either mimic or support physiological processes. Ensuring that MEG stay fixed in their intended bodily location is vital for consistent performance. This includes devising effective fixation methods to prevent device migration or dislodgement, which could impair function or necessitate further surgical procedures. With implanted MEG collecting and transmitting sensitive health data, securing this information against unauthorized access is imperative. Efforts are underway to implement robust encryption and privacy-preserving measures to safeguard patient data, addressing cybersecurity risks associated with wireless communication. The reliability of wireless therapy provided by in-body MEG is critical for patient confidence and the broader acceptance of these devices. This encompasses ensuring consistent energy harvesting and delivery, precise therapeutic control, and incorporating fail-safe mechanisms to avert malfunctions that could endanger patients.
In summary, the advent of wearable and implantable MEG marks a significant advancement in healthcare. These devices combine magnetoelasticity with magnetic control to usher in a new era of health monitoring systems that are both personalized and attuned to individual health dynamics. Wearable MEG represent large potential in health management, enabling continuous monitoring and on-demand therapeutic interventions as integral parts of daily life. This integration into daily routines promises to improve patient outcomes by facilitating timely medical responses and reducing the reliance on invasive procedures. The development of implanted MEG faces several challenges, including ensuring biocompatibility, achieving seamless integration within the human body, and maintaining functionality without compromising miniaturization. Overcoming these hurdles and evolution towards advanced medical treatments that are both responsive and inherently part of our daily existence highlights the potential of MEG to redefine the future of medical technology.
Acknowledgements
This work was supported from National Natural Science Foundation (Grant 22205254 and 5230130320), Shanghai Science and Technology Innovation Sailing Project (23YF1401700), 2023 Fudan University Medicine-Engineering Integration Project (National Key Project GZ45782065), Fudan University Young Talent Introduction Program (JIH2306031Y), the Initiative Postdoctor Supporting Program, China Postdoctoral Science Foundation (grant No. BX20200209), the National Taipei University of Technology-Penn State Collaborative Seed Grant Program (NTUT-PSU-112-01) and an Alexander von Humboldt research fellowship. The authors thank editors' and reviewers' invaluable comments in improving this work.
Conflicts of Interest
The authors declared no competing financial interests.
Data Availability
Data sharing is not applicable to this review as no datasets were generated or analyzed during the current study.
Copyright Permissions
Copyright © 2025 The Author(s). Published by Innovation Forever Publishing Group Limited. This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.
Author Contribution
Xu S conceptualized the review topic, contributed to writing, and revised the manuscript. Ge M and Tang Y conducted literature research and drafted sections of the manuscript. Wang Y contributed to writing and critically revised the manuscript. Liao Y provided insights into bioelectronic applications. Zhang W and Wang J conceptualized the review topic, supervised the project, and finalized the manuscript.
Abbreviation List
EMG, Electromagnetic generators
FEP, Fluorinated ethylene propylene
Galfenol, GaxFe1-x
ITO, Indium tin oxide
MEGs, Magnetoelastic generators
OCT, Optical coherence tomography
PA, Polyamide
PDMS, Polydimethylsiloxane
PENG, Piezoelectric nanogenerator
PTFE, Polytetrafluoroethylene
PVDF, Polyvinylidene fluoride
TENG, Triboelectric nanogenerator
TMS, Transcranial magnetic stimulation
Terfenol-D, TbxDy1-xFe2
References
[1] Yu Y, Nyein H, Gao W et al. Flexible electrochemical bioelectronics: the rise of in situ bioanalysis. Adv Mater, 2020; 32: 1902083.[DOI]
[2] Gong S, Lu Y, Yin J et al. Materials-driven soft wearable bioelectronics for connected healthcare. Chem Rev, 2024; 124: 455-553.[DOI]
[3] Tan M, Xu Y, Gao Z et al. Recent advances in intelligent wearable medical devices integrating biosensing and drug delivery. Adv Mater, 2022; 34: 2108491.[DOI]
[4] Sunwoo S-H, Ha K-H, Lee S et al. Wearable and implantable soft bioelectronics: Device designs and material strategies. Annu Rev Chem Biomol Eng, 2021; 12: 359-391.[DOI]
[5] Zhang S, Tan R, Zhang K et al. Implantable heterogeneous triboelectric nanogenerator effectively prevents denervation-induced muscle atrophy. Nano Energy, 2024; 125: 109548.[DOI]
[6] Xu S, Farid M, Chen G et al. Reversible metal-ligand coordination for photocontrolled metallopolymer adhesives. Chem, 2024; 10: 1638-1640.[DOI]
[7] Li P, Lee G-H, Kim S et al. From Diagnosis to Treatment: Recent Advances in Patient-Friendly Biosensors and Implantable Devices. ACS Nano, 2021; 15: 1960-2004.[DOI]
[8] Xu M, Liu Y, Yang K et al. Minimally invasive power sources for implantable electronics. Exploration, 2024; 4: 20220106.[DOI]
[9] Yue O, Wang X, Xie L et al. Biomimetic exogenous “tissue batteries” as artificial power sources for implantable bioelectronic devices manufacturing. Adv Sci, 2024: 2307369.[DOI]
[10] Xu S, Chen G, Scott K et al. Soft electrochemical actuators for intraoperative nerve activity monitoring. Matter, 2024; 7: 2795-2797.[DOI]
[11] Townsend N, Kazakiewicz D, Lucy Wright F et al. Epidemiology of cardiovascular disease in Europe. Nat Rev Cardiol, 2022; 19: 133-143.[DOI]
[12] Rana SS, Salauddin M, Sharifuzzaman M et al. Ultrahigh-output triboelectric and electromagnetic hybrid generator for self-powered smart electronics and biomedical applications. Adv Energy Mater, 2022; 12: 2202238.[DOI]
[13] Zhang T, Ding Y, Hu C et al. Self-powered stretchable sensor arrays exhibiting magnetoelasticity for real-time human-machine interaction. Adv Mater, 2023; 35: 2203786.[DOI]
[14] Guo H, Shi Y, Pan F et al. Tough, stretchable dual-network liquid metal-based hydrogel toward high-performance intelligent on-off electromagnetic interference shielding, human motion detection and self-powered application. Nano Energy, 2023; 114: 108678.[DOI]
[15] Zhou Y, Zhao X, Xu J et al. Giant magnetoelastic effect in soft systems for bioelectronics. Nat Mater, 2021; 20: 1670-1676.[DOI]
[16] Chen G, Zhou Y, Fang Y et al. Wearable Ultrahigh Current Power Source Based on Giant Magnetoelastic Effect in Soft Elastomer System. ACS Nano, 2021; 15: 20582-20589.[DOI]
[17] Chen G, Zhao X, Andalib S et al. Discovering giant magnetoelasticity in soft Matter for electronic textiles. Matter, 2021; 4: 3725-3740.[DOI]
[18] Zhao X, Zhou Y, Xu J et al. Soft fibers with magnetoelasticity for wearable electronics. Nat Commun, 2021; 12: 6755.[DOI]
[19] Eem S-H, Jung H-J, Koo J-H. Application of MR elastomers for improving seismic protection of base-isolated structures. IEEE Trans Magn, 2011; 47: 2901-2904.[DOI]
[20] Libanori A, Soto J, Xu J et al. Self-powered programming of fibroblasts into neurons via a scalable magnetoelastic generator array. Adv Mater, 2023; 35: 2206933.[DOI]
[21] Veletic M, Apu EH, Simic M et al. Implants with sensing capabilities. Chem Rev, 2022; 122: 16329-16363.[DOI]
[22] Ock IW, Zhao X, Wan X et al. Boost the voltage of a magnetoelastic generator via tuning the magnetic induction layer resistance. Nano Energy, 2023; 109: 108298.[DOI]
[23] Zhao X, Nashalian A, Ock IW et al. A soft magnetoelastic generator for wind-energy harvesting. Adv Mater, 2022; 34: 2204238.[DOI]
[24] Xu J, Tat T, Zhao X et al. Spherical Magnetoelastic Generator for Multidirectional Vibration Energy Harvesting. ACS Nano, 2023; 17: 3865-3872.[DOI]
[25] Bright A. Contact and Frictional Electrification. Electron Power, 1967; 13: 480.[DOI]
[26] Iversen P, Lacks DJ. A life of its own: The tenuous connection between Thales of Miletus and the study of electrostatic charging. J Electrostat, 2012; 70: 309-311.[DOI]
[27] Xu S, Manshaii F, Xiao X et al. Artificial Intelligence Assisted Nanogenerator Applications. J Mater Chem, 2024; 10.1039/D4TA07127A.
[28] Xu S, Manshaii F, Xiao X et al. Triboelectric Nanogenerators for Self-Powered Neurostimulation. Nano Res, 2024; 17: 8926-8941.[DOI]
[29] Kim W-G, Kim D-W, Tcho I-W et al. Triboelectric Nanogenerator: Structure, Mechanism, and Applications. ACS Nano, 2021; 15: 258-287.[DOI]
[30] Wang S, Lin L, Xie Y et al. Sliding-triboelectric nanogenerators based on in-plane charge-separation mechanism. Nano Lett, 2013; 13: 2226-2233.[DOI]
[31] Conta G, Libanori A, Tat T et al. Triboelectric nanogenerators for therapeutic electrical stimulation. Adv Mater, 2021; 33: 2007502.[DOI]
[32] Henniker J. Triboelectricity in polymers. Nature, 1962; 196: 474-474.[DOI]
[33] Lone SA, Lim KC, Kaswan K et al. Recent advancements for improving the performance of triboelectric nanogenerator devices. Nano Energy, 2022; 99: 107318.[DOI]
[34] Wang C, Guo H, Wang P et al. An advanced strategy to enhance teng output: reducing triboelectric charge decay. Adv Mater, 2023; 35: 2209895.[DOI]
[35] Li J, Che Z, Wan X et al. Biomaterials and bioelectronics for self-powered neurostimulation. Biomaterials, 2023; 304: 122421.[DOI]
[36] Liu Y, Mo J, Fu Q et al. Enhancement of triboelectric charge density by chemical functionalization. Adv Funct Mater, 2020; 30: 2004714.[DOI]
[37] Yu D, Li Z, Xie W et al. Applications of nanogenerator-based wearable devices in orthopedics. Nano Energy, 2022; 103: 107762.[DOI]
[38] Deng W, Zhou Y, Zhao X et al. Ternary Electrification Layered Architecture for High-Performance Triboelectric Nanogenerators. ACS Nano, 2020; 14: 9050-9058.[DOI]
[39] Akram W, Chen Q, Xia G et al. A review of single electrode triboelectric nanogenerators. Nano Energy, 2023; 106: 108043.[DOI]
[40] Khandelwal G, Maria Joseph Raj NP, Kim SJ. Materials beyond conventional triboelectric series for fabrication and applications of triboelectric nanogenerators. Adv Energy Mater, 2021; 11: 2101170.[DOI]
[41] Wang AC, Zhang B, Xu C et al. Unraveling temperature-dependent contact electrification between sliding-mode triboelectric pairs. Adv Funct Mater, 2020; 30: 1909384.[DOI]
[42] Liu D, Zhou L, Wang ZL et al. Triboelectric nanogenerator: from alternating current to direct current. iScience, 2021; 1: 24.[DOI]
[43] Liu Y, Yan W, Han J et al. Aerodynamics-based triboelectric nanogenerator for enhancing multi-operating robustness via mode automatic switching. Adv Funct Mater, 2022; 32: 2202964.[DOI]
[44] Zou Y, Xu J, Chen K et al. Advances in nanostructures for high-performance triboelectric nanogenerators. Adv Mater Technol, 2021; 6: 2000916.[DOI]
[45] Parandeh S, Etemadi N, Kharaziha M et al. Advances in triboelectric nanogenerators for self-powered regenerative medicine. Adv Funct Mater, 2021; 31: 2105169.[DOI]
[46] Jin L, Xiao X, Deng W et al. Manipulating relative permittivity for high-performance wearable triboelectric nanogenerators. Nano Lett, 2020; 20: 6404-6411.[DOI]
[47] Yin J, Kashyap V, Wang S et al. Self-powered eye-computer interaction via a triboelectric nanogenerator. Device, 2024; 2: 100252.[DOI]
[48] Peng Z, Xiao X, Song J et al. Improving relative permittivity and suppressing dielectric loss of triboelectric layers for high-performance wearable electricity generation. ACS Nano, 2022; 16: 20251-20262.[DOI]
[49] Meng K, Xiao X, Wei W et al. Wearable pressure sensors for pulse wave monitoring. Adv Mater, 2022; 34: 2109357.[DOI]
[50] Mahmud MP, Tat T, Xiao X et al. Advances in 4D-printed physiological monitoring sensors. Exploration, 2021; 1: 20210033.[DOI]
[51] Elsanadidy E, Mosa IM, Luo D et al. Advances in Triboelectric Nanogenerators for Self-powered Neuromodulation. Adv Funct Mater, 2023; 33: 2211177.[DOI]
[52] Zhou B, Zhang S, Liu W et al. A Review of Evaluation, Principles, and Technology of Wearable Electromagnetic Harvesters. ACS Appl Electron Mater, 2023; 5: 4035-4050.[DOI]
[53] Park J, Lee Y, Cho S et al. Soft Sensors and Actuators for Wearable Human-Machine Interfaces. Chem Rev, 2024; 124: 1464-1534.[DOI]
[54] Kwon JY, Kim JE, Kim JS et al. Artificial sensory system based on memristive devices. Exploration, 2023; 4: 20220162.[DOI]
[55] Ge X, Hu N, Yan F, Wang Y. Development and applications of electrospun nanofiber-based triboelectric nanogenerators. Nano Energy, 2023; 112: 108444.[DOI]
[56] Su Y, Chen G, Chen C et al. Self-powered respiration monitoring enabled by a triboelectric nanogenerator. Adv Mater, 2021; 33: 2101262.[DOI]
[57] Zhou Z, Weng L, Tat T et al. Smart Insole for Robust Wearable Biomechanical Energy Harvesting in Harsh Environments. ACS Nano, 2020; 14: 14126-14133.[DOI]
[58] Curie J, Curie P. Development of polar electricity by compression in hemihedral crystals with inclined faces. Bull Min, 1880; 3: 90-93.[DOI]
[59] Yuan X, Shi J, Kang Y et al. Piezoelectricity, Pyroelectricity, and Ferroelectricity in Biomaterials and Biomedical Applications. Adv Mater, 2024; 36: 2308726.[DOI]
[60] Meng N, Liu W, Jiang R et al. Fundamentals, advances and perspectives of piezocatalysis: A marriage of solid-state physics and catalytic chemistry. Prog Mater Sci, 2023; 138: 101161.[DOI]
[61] Duan J, Zheng Z, Li Y, Liu T. Enhanced photocatalytic degradation performance of layered Ag-Bi4Ti3O12 plasmon resonance-coupled piezoelectric effect. Mater Res Bull, 2024; 171: 112610.[DOI]
[62] Xu S, Wan X, Manshaii F et al. Advances in Piezoelectric Nanogenerators for Self-Powered Cardiac Care. Nano Trends, 2024; 6: 100042.[DOI]
[63] Yao Z, Deng J, Li L. Piezoelectric performance regulation from 2D materials to devices. Matter, 2024; 7: 855-888.[DOI]
[64] Zhang Y, Mao J, Zheng RK et al. Ferroelectric Polarization-Enhanced Performance of Flexible CuInP2S6 Piezoelectric Nanogenerator for Biomechanical Energy Harvesting and Voice Recognition Applications. Adv Funct Mater, 2023; 33: 2214745.[DOI]
[65] Jin CC, Liu DM, Zhang LX. An emerging family of piezocatalysts: 2D piezoelectric materials. Small, 2023; 19: 2303586.[DOI]
[66] Carlos C, Li J, Zhang Z et al. Strain-Correlated Piezoelectricity in Quasi-Two-Dimensional Zinc Oxide Nanosheets. Nano Lett, 2023; 23: 6148-6155.[DOI]
[67] Choi D, Jo H, Yoon T et al. Transparent, flexible, and highly sensitive piezocomposite capable of harvesting and monitoring kinetic movements of microbubbles in liquid. Adv Funct Mater, 2023; 33: 2307607.[DOI]
[68] Joshi B, Seol J, Samuel E et al. Supersonically sprayed PVDF and ZnO flowers with built-in nanocuboids for wearable piezoelectric nanogenerators. Nano Energy, 2023; 112: 108447.[DOI]
[69] Zhang H, Alanthattil A, Webster RF et al. Robust Switchable Polarization and Coupled Electronic Characteristics of Magnesium-Doped Zinc Oxide. ACS Nano, 2023; 17: 17148-17157.[DOI]
[70] Sood A, Desseigne M, Dev A et al. A comprehensive review on barium titanate nanoparticles as a persuasive piezoelectric material for biomedical applications: prospects and challenges. Small, 2023; 19: 2206401.[DOI]
[71] Wang Y, Li X, Chen Y et al. Pulsed-Laser-Triggered Piezoelectric Photocatalytic CO2 Reduction over Tetragonal BaTiO3 Nanocubes. Adv Mater, 2023; 35: 2305257.[DOI]
[72] Kumar M, Kulkarni ND, Kumari P. Piezoelectric performance enhancement of electrospun functionally graded PVDF/BaTiO3 based flexible nanogenerators. Mater Res Bull, 2024; 174: 112739.[DOI]
[73] Li J, Qu W, Daniels J et al. Lead zirconate titanate ceramics with aligned crystallite grains. Science, 2023; 380: 87-93.[DOI]
[74] Li X, Zhang Z, Peng Z et al. Fast and versatile electrostatic disc microprinting for piezoelectric elements. Nat Commun, 2023; 14: 6488.[DOI]
[75] Wang Y, Zang P, Yang D et al. The fundamentals and applications of piezoelectric materials for tumor therapy: recent advances and outlook. Mater Horiz, 2023; 10: 1140-1184.[DOI]
[76] Podder S, Mondal S, Bhattacharjee S et al. Bi-functional Rb4Ag2BiBr9-PVDF Composite Based Piezoelectric Mechanical Energy Harvester and Light Assisted Self-Charging Power Unit. Nano Energy, 2024; 123: 109380.[DOI]
[77] Chai B, Shi K, Wang Y et al. Modulus-Modulated All-Organic Core-Shell Nanofiber with Remarkable Piezoelectricity for Energy Harvesting and Condition Monitoring. Nano Lett, 2023; 23: 1810-1819.[DOI]
[78] Zong Q, Lv B, Liu C et al. Dendrite-Free and Highly Stable Zn Metal Anode with BaTiO3/P(VDF-TrFE) Coating. ACS Energy Lett, 2023; 8: 2886-2896.[DOI]
[79] Yoon C, Ippili S, Thomas AM et al. Harnessing Flexoelectric and Piezoelectric Effects for Self-Charging Power Systems. ACS Energy Lett, 2023; 8: 4634-4642.[DOI]
[80] Su Y, Liu J, Yang D et al. Electric Field-Assisted Self-Assembly of Diphenylalanine Peptides for High-Performance Energy Conversion. ACS Mater Lett, 2023; 5: 2317-2323.[DOI]
[81] Zhi C, Shi S, Wu H et al. Emerging trends of nanofibrous piezoelectric and triboelectric applications: mechanisms, electroactive materials, and designed architectures. Adv Mater, 2024; 36: 2401264.[DOI]
[82] Newnham R, Skinner D, Cross L. Connectivity and piezoelectric-pyroelectric composites. Mater Res Bull, 1978; 13: 525-536.[DOI]
[83] Kabakov P, Kim T, Cheng Z et al. The Versatility of Piezoelectric Composites. Annu Rev Mater Res, 2023; 53: 165-193.[DOI]
[84] Wang W, Xiang Y, Yu J et al. Development and Prospect of Smart Materials and Structures for Aerospace Sensing Systems and Applications. Sensors, 2023; 23: 1545.[DOI]
[85] Vijayakanth T, Shankar S, Finkelstein-Zuta G et al. Perspectives on recent advancements in energy harvesting, sensing and bio-medical applications of piezoelectric gels. Chem Soc Rev, 2023; 52: 6191-6220.[DOI]
[86] Jiang L, Wu J. Emerging ultrasonic bioelectronics for personalized healthcare. Prog Mater Sci, 2023; 136: 101110.[DOI]
[87] Bhatnagar P, Zaferani SH, Rafiefard N et al. Advancing personalized healthcare and entertainment: Progress in energy harvesting materials and techniques of self-powered wearable devices. Prog Mater Sci, 2023; 139: 101184.[DOI]
[88] Chen S, Zhu P, Mao L et al. Piezocatalytic medicine: An emerging frontier using piezoelectric materials for biomedical applications. Adv Mater, 2023; 35: 2208256.[DOI]
[89] Wang S, Khan AA, Teale S et al. Large piezoelectric response in a Jahn-Teller distorted molecular metal halide. Nat Commun, 2023; 14: 1852.[DOI]
[90] Zhang T, Liu N, Xu J et al. Flexible electronics for cardiovascular healthcare monitoring. Innovation, 2023; 4: 100485.[DOI]
[91] Mahanty B, Ghosh SK, Prasad G et al. Giant Energy Harvesting via Maxwell Displacement Current Enhancement Using Metal Sheet Interspaced Hetero-Layer Structured Piezo-Composite Nanofiber Device. Adv Funct Mater, 2023; 34: 2307723.[DOI]
[92] Yan D, Wang J, Xiang J et al. A flexoelectricity-enabled ultrahigh piezoelectric effect of a polymeric composite foam as a strain-gradient electric generator. Sci Adv, 2023; 9: eadc8845.[DOI]
[93] Liu J, Li S, Zhou S et al. A High-Performance, Biocompatible, and Fully Biodegradable Piezo-Triboelectric Hybrid Nanogenerator Based on PVA/Glycine/PVA Heterostructured Piezoelectric Film. Nano Energy, 2024; 122: 109310.[DOI]
[94] Zhang C. Comparison of Triboelectric Nanogenerator and Electromagnetic Generator. In: Wang ZL, YangnY, Zha J et al (eds). Handbook of Triboelectric Nanogenerators. Springer, Cham, Switzerland. 2023: 1-34.[DOI]
[95] Chen J, Gong S, Gong T et al. Stackable Direct Current Triboelectric-Electromagnetic Hybrid Nanogenerator for Self-Powered Air Purification and Quality Monitoring. Adv Energy Mater, 2023; 13: 2203689.[DOI]
[96] Li G, Wang J, Fu S et al. A Nanogenerator Enabled by a Perfect Combination and Synergetic Utilization of Triboelectrification, Charge Excitation and Electromagnetic Induction to Reach Efficient Energy Conversion. Adv Funct Mater, 2023; 33: 2213893.[DOI]
[97] Zeng Q, Chen A, Zhang X et al. A Dual-Functional Triboelectric Nanogenerator Based on the Comprehensive Integration and Synergetic Utilization of Triboelectrification, Electrostatic Induction, and Electrostatic Discharge to Achieve Alternating Current/Direct Current Convertible Outputs. Adv Mater, 2023; 35: 2208139.[DOI]
[98] Zhang C, Yang S, Dai X et al. Hybridized triboelectric-electromagnetic nanogenerators for efficient harvesting of wave energy for self-powered ocean buoy. Nano Energy, 2024; 128: 109929.[DOI]
[99] Cao X, Zhou H, Zhou Y et al. High Performance Rotary-Structured Triboelectric-Electromagnetic Hybrid Nanogenerator for Ocean Wind Energy Harvesting. Adv Mater Technol, 2023; 8: 2300327.[DOI]
[100] Zhang Y, Liu J, Zhang J et al. A flexible droplet-based triboelectric-electromagnetic hybrid generator for raindrop energy harvesting. Nano Energy, 2024; 121: 109253.[DOI]
[101] Liu Z, Chen X, Xia Z et al. A self-powered electro-coagulation system afforded by flexible electromagnetic flag wind generators for efficient removal of arsenic from water. Nano Energy, 2023; 114: 108648.[DOI]
[102] Alves T, Rodrigues C, Callaty C et al. Airflow-Driven Triboelectric-Electromagnetic Hybridized Nanogenerator for Biomechanical Energy Harvesting. Adv Mater Technol, 2023; 8: 2300689.[DOI]
[103] Skrovanek D, Ludois DC. Electrostatic Wave Energy Conversion: A Review of Devices, Theory, and Hurdles. IEEE Trans Energy Convers, 2024.[DOI]
[104] Vidal JV, Carneiro PM, dos Santos MPS. A complete physical 3D model from first principles of vibrational-powered electromagnetic generators. Appl Energy, 2024; 357: 122387.[DOI]
[105] Varpula A, Laakso SJ, Havia T et al. Harvesting vibrational energy using material work functions. Sci Rep, 2014; 4: 6799.[DOI]
[106] Boisseau S, Despesse G, Seddik BA. Electrostatic conversion for vibration energy harvesting. Small Scale Energy Harves, 2012; 5. 40640.[DOI]
[107] Xu S, Farid M, Chen, J. Multiphasic interfaces enabled aero-elastic capacitive pressure sensor. Matter, 2024; 7: 2351-2354.[DOI]
[108] Zhu Y, Xu W, Ravichandran D et al. A gill-mimicking thermoelectric generator (TEG) for waste heat recovery and self-powering wearable devices. J Mater Chem, 2021; 9: 8514-8526.[DOI]
[109] Ganesh S, Lokesh A, Pranesh N et al. Automated Power Generation Using Biogas and Thermo-Electric Generator by AI Technology. 2022 International Conference on Advances in Computing, Communication and Applied Informatics (ACCAI), Chennai, India. 2022: 1-8.[DOI]
[110] Wang L, Li K, Zhang S et al. Modeling the effects of module size and material property on thermoelectric generator power. ACS Omega, 2020; 5: 29844-29853.[DOI]
[111] Chen W-H, Lee K-T, Chih Y-K et al. Novel renewable double-energy system for activated biochar production and thermoelectric generation from waste heat. Energy Fuels, 2020; 34: 3383-3393.[DOI]
[112] Ren W, Sun Y, Zhao D et al. High-performance wearable thermoelectric generator with self-healing, recycling, and Lego-like reconfiguring capabilities. Sci Adv, 2021; 7: eabe0586.[DOI]
[113] Yu J, Ma E, Ma T. Harvesting energy from low-frequency excitations through alternate contacts between water and two dielectric materials. Sci Rep, 2017; 7: 17145.[DOI]
[114] Boland J, Chao Y-H, Suzuki Y et al. Micro electret power generator. The Sixteenth Annual International Conference on Micro Electro Mechanical Systems, 2003. MEMS-03 Kyoto. IEEE, 2003: 538-541.
[115] Zhao X, Chen G, Zhou Y et al. Giant magnetoelastic effect enabled stretchable sensor for self-powered biomonitoring. ACS Nano, 2022; 16: 6013-6022.[DOI]
[116] Zhou Y, Xiao X, Chen G et al. Self-powered sensing technologies for human Metaverse interfacing. Joule, 2022; 6: 1381-1389.[DOI]
[117] Zhou Y, Zhao X, Xu J et al. A multimodal magnetoelastic artificial skin for underwater haptic sensing. Sci Adv, 2024; 10: eadj8567.[DOI]
[118] Gao C, Huang J, Xiao Y et al. A seamlessly integrated device of micro-supercapacitor and wireless charging with ultrahigh energy density and capacitance. Nat Commun, 2021; 12: 2647.[DOI]
[119] Makaroff SN, Nguyen H, Meng Q et al. Modeling transcranial magnetic stimulation coil with magnetic cores. J Neural Eng, 2023; 20: 016028.[DOI]
[120] Membrilla JAV, Pantoja MF, Puerta APV et al. Design of Transcranial Magnetic Stimulation Coils With Optimized Stimulation Depth. IEEE Access, 2024; 12: 1330-1340.[DOI]
[121] Rissanen IJ, Souza VH, Nieminen JO et al. Advanced Pipeline for Designing Multi-Locus TMS Coils with Current Density Constraints. IEEE Trans Biomed Eng, 2023; 70: 2025-2034.[DOI]
[122] Detka K, Górecki K. Wireless power transfer-A review. Energies, 2022; 15: 7236.[DOI]
[123] Luo Y, Hutapea P. Stress-strain behavior of a smart magnetostrictive actuator for a bone transport device. J Med Devices, 2008; 2: 041002.[DOI]
[124] Khojah R, Xiao Z, Panduranga MK et al. Single-domain multiferroic array-addressable terfenol-D (SMArT) micromagnets for programmable single-cell capture and release. Adv Mater, 2021; 33: 2006651.[DOI]
[125] Bichurin MI, Petrov RV, Leontiev VS et al. Self-biased bidomain LiNbO3/Ni/Metglas magnetoelectric current sensor. Sensors, 2020; 20: 7142.[DOI]
[126] Jiang Z, Liu X, Jin Z et al. Real Time Monitoring of Weak Magnetic Field. 2023 IEEE 6th International Conference on Electronic Information and Communication Technology (ICEICT). IEEE, 2023; 1063-1065.[DOI]
[127] Chen JC, Kan P, Yu Z et al. A wireless millimetric magnetoelectric implant for the endovascular stimulation of peripheral nerves. Nat Biomed Eng, 2022; 6: 706-716.[DOI]
[128] Huong Giang DT, Tam HA, Ngoc Khanh VT et al. Magnetoelectric vortex magnetic field sensors based on the metglas/PZT laminates. Sensors, 2020; 20: 2810.[DOI]
[129] G. Saiz P, Fernández de Luis R, Lasheras A et al. Magnetoelastic resonance sensors: Principles, applications, and perspectives. ACS Sensors, 2022; 7: 1248-1268.[DOI]
[130] Yun G, Tang S-Y, Sun S et al. Liquid metal-filled magnetorheological elastomer with positive piezoconductivity. Nat Commun, 2019; 10: 1300.[DOI]
[131] Bian Y, Liang X, Gao Z. Vibration reduction for a flexible arm using magnetorheological elastomer vibration absorber. Shock Vib, 2018; 2018: 1-13.[DOI]
[132] Kennedy BF, McLaughlin RA, Kennedy KM et al. Optical coherence micro-elastography: mechanical-contrast imaging of tissue microstructure. Biomed Opt Express, 2014; 5: 2113-2124.[DOI]
[133] Moreno-Mateos MA, Hossain M, Steinmann P, Garcia-Gonzalez D. Hybrid magnetorheological elastomers enable versatile soft actuators. NPJ Comput Mater, 2022; 8: 162.[DOI]
[134] Zhu Z, Wang Z, Dai K et al. An adaptive and space-energy efficiency vibration absorber system using a self-sensing and tunable magnetorheological elastomer. Nano Energy, 2023; 117: 108927.[DOI]
[135] Bhat A, Rao VS, Jayalakshmi N. Review of the evolution of magnetorheological fluid-based rehabilitative devices: from the perspective of modelling, sensors and control strategies. IEEE Access, 2023; 11: 88759-88777.[DOI]
[136] Bai H, Li S, Shepherd RF. Elastomeric haptic devices for virtual and augmented reality. Adv Funct Mater, 2021; 31: 2009364.[DOI]
[137] Christie MD, Fredericksen T, Li W. Development of a Magnetorheological Elastomer Actuator for a Mixed Reality Haptic Glove. 2023 IEEE/ASME International Conference on Advanced Intelligent Mechatronics (AIM). IEEE, 2023: 507-510.[DOI]
[138] Zhu M, Sun Z, Zhang Z et al. Haptic-feedback smart glove as a creative human-machine interface (HMI) for virtual/augmented reality applications. Sci Adv, 2020; 6: eaaz8693.[DOI]
[139] Xu S, Liu Y, Lee H et al. Neural interfaces: Bridging the brain to the world beyond healthcare. Exploration, 2024; 4: 20230146.[DOI]
[140] Khalid MA, Saleem MM, Bukhari SA et al. Design and Characterization of Three-Axis High-Range Inductive Tactile Force Sensor Utilizing Magnetorheological Elastomer for Robotic Surgical Applications. IEEE Sensors J, 2022; 23: 247-255.[DOI]
[141] McDonald KJ, Kinnicutt L, Moran AM et al. Modulation of magnetorheological fluid flow in soft robots using electropermanent magnets. IEEE Robot Autom Lett, 2022; 7: 3914-3921.[DOI]
[142] Choi DS, Kim TH, Lee SH et al. Beyond Human Hand: Shape-Adaptive and Reversible Magnetorheological Elastomer-Based Robot Gripper Skin. ACS Appl Mater Interfaces, 2020; 12: 44147-44155.[DOI]
[143] Park J, Choi YT, Flatau AB, Wereley NM. Encapsulations of magnetorheological fluids within 3-D printed elastomeric cellular structures. IEEE Trans Magn, 2021; 58: 1-5.[DOI]
[144] Sun Y, Wang L, Zhu Z et al. A 3D-Printed Ferromagnetic Liquid Crystal Elastomer with Programmed Dual-Anisotropy and Multi-Responsiveness. Adv Mater, 2023; 35: 2302824.[DOI]
[145] Xu S, Ahmed S, Momin M et al. Unleashing the potential of 3D printing soft materials. Device, 2023; 1: 100067.[DOI]
[146] Chung HJ, Parsons AM, Zheng L. Magnetically controlled soft robotics utilizing elastomers and gels in actuation: A review. Adv Intell Syst, 2021; 3: 2000186.[DOI]
[147] Le VH, Rodriguez HL, Lee C et al. A soft-magnet-based drug-delivery module for active locomotive intestinal capsule endoscopy using an electromagnetic actuation system. Sens Actuators A Phys, 2016; 243: 81-89.[DOI]
[148] Pang X, Lee H, Rong J et al. Self-Thermal Management in Filtered Selenium-Terminated MXene Films for Flexible Safe Batteries. Small, 2024; 20: 2309580.[DOI]
[149] Spetzler E, Spetzler B, McCord J. A Magnetoelastic Twist on Magnetic Noise: The Connection with Intrinsic Nonlinearities. Adv Funct Mater, 2023: 34: 2309867.[DOI]
[150] Wang Y, Haick H, Guo S et al. Skin bioelectronics towards long-term, continuous health monitoring. Chem Soc Rev, 2022; 51: 3759-3793.[DOI]
[151] Song H, Jang Y, Lee JP et al. Highly Compressible 3D-Printed Soft Magnetoelastic Sensors for Human-Machine Interfaces. ACS Appl Mater Interfaces, 2023; 15: 59776-59786.[DOI]
[152] Xia N, Jin D, Zhang L. Magnetic Soft Matterials toward Programmable and Multifunctional Miniature Machines. Acc Mater Res, 2024.[DOI]
[153] Abu-Halimah J, Majmudar A, Tian B. Chemical Approaches to Emerging Advancements in Deformable Bioelectronics: Synthesis, Device Concepts, Performance, and Applications. CCS Chem, 2024; 6: 11-26.[DOI]
[154] Li J, Che Z, Wan X et al. Biomaterials and bioelectronics for self-powered neurostimulation. Biomaterials, 2023; 304: 122421.[DOI]
[155] Libanori A, Chen G, Zhao X et al. Smart textiles for personalized healthcare. Nat Electron, 2022; 5: 142-156.[DOI]
[156] Xu J, Tat T, Yin J et al. A textile magnetoelastic patch for self-powered personalized muscle physiotherapy. Matter, 2023; 6: 2235-2247.[DOI]
[157] Wang B, Engay E, Stubbe PR et al. Stiffness control in dual color tomographic volumetric 3D printing. Nat Commun, 2022; 13: 367.[DOI]
[158] Wang R, Du Z, Xia Z et al. Magnetoelectrical Clothing Generator for High-Performance Transduction from Biomechanical Energy to Electricity. Adv Funct Mater, 2022; 32: 2107682.[DOI]
[159] Chen G, Xiao X, Zhao X et al. Electronic textiles for wearable point-of-care systems. Chem Rev, 2021; 122: 3259-3291.[DOI]
[160] Tat T, Chen G, Zhao X et al. Smart textiles for healthcare and sustainability. ACS Nano, 2022; 16: 13301-13313.[DOI]
[161] Xu S, Scott K, Chen J. A bioinspired three-dimensionally architected electronic skin. Device, 2024; 2: 100462.[DOI]
[162] Yin J, Wang S, Di Carlo A et al. Smart textiles for self-powered biomonitoring. Med-X, 2023, 1: 3.[DOI]
[163] Hashemi SA, Ghaffarkhah A, Hosseini E et al. Recent progress on hybrid fibrous electromagnetic shields: key protectors of living species against electromagnetic radiation. Matter, 2022; 5: 3807-3868.[DOI]
[164] Brown Jr WF. Theory of magnetoelastic effects in ferromagnetism. J Appl Phys, 1965; 36: 994-1000.[DOI]
[165] Rinaldi S, Turilli G. Theory of linear magnetoelastic effects. Phys Rev B, 1985; 31: 3051.[DOI]
[166] Dorfmann A, Ogden R. Nonlinear magnetoelastic deformations. Quarterly J Mech Appl Math, 2004; 57: 599-622.[DOI]
[167] Sánchez PA, Stolbov OV, Kantorovich SS, Raikher YL. Modeling the magnetostriction effect in elastomers with magnetically soft and hard particles. Soft Matter, 2019; 15: 7145-7158.[DOI]
[168] Elhajjar R, Law C-T, Pegoretti A. Magnetostrictive polymer composites: Recent advances in materials, structures and properties. Prog Mater Sci, 2018; 97: 204-229.[DOI]
[169] Ebrahimi F, Ahari MF. Mechanics of Magnetostrictive Materials and Structures. CRC Press, 2023.[DOI]
[170] Sander D. Magnetostriction and magnetoelasticity. Handbook Magn Magn Mater, 2020: 1-45.[DOI]
[171] Egorov D, Petrov I, Pyrhönen JJ et al. Hysteresis loss in NdFeB permanent magnets in a permanent magnet synchronous machine. IEEE Trans Ind Electron, 2021; 69: 121-129.[DOI]
[172] Wajahat M, Kim JH, Kim JH et al. 4D Printing of Ultrastretchable Magnetoactive Soft Material Architectures for Soft Actuators. ACS Appl Mater Interfaces, 2023; 15: 59582-59591.[DOI]
[173] Kim Y, Zhao X. Magnetic soft materials and robots. Chem Rev, 2022; 122: 5317-5364.[DOI]
[174] Alapan Y, Karacakol AC, Guzelhan SN et al. Reprogrammable shape morphing of magnetic soft machines. Sci Adv, 2020; 6: eabc6414.[DOI]
[175] Rabbani Y, Shayesteh H, Haghshenas N, Safarzadeh Khosrowshahi M. Investigation of grafting Silane coupling agents on superhydrophobicity of carbonyl iron/SiO2 particles for efficient oil/water mixture and emulsion separation. Sci Rep, 2023; 13: 788.[DOI]
[176] Li S, Zhang J, He J et al. Functional PDMS elastomers: Bulk composites, surface engineering, and precision fabrication. Adv Sci, 2023; 10: 2304506.[DOI]
[177] Uyeda C, Hisayoshi K, Terada K. Separation of gold and other rare materials from an ensemble of heterogeneous particles using a NdFeB magnetic circuit. Sci Rep, 2019; 9: 3971.[DOI]
[178] Lu Y, Yu D, Dong H et al. Dynamic Leakage-Free Liquid Metals. Adv Funct Mater, 2023; 33: 2210961.[DOI]
[179] Lu Y, Yu D, Dong H et al. Magnetically tightened form-stable phase change materials with modular assembly and geometric conformality features. Nat Commun, 2022; 13: 1397.[DOI]
[180] Xu J, Tat T, Zhao X et al. A programmable magnetoelastic sensor array for self-powered human-machine interface. Appl Phys Rev, 2022; 9: 031404.[DOI]
[181] Gong S, Lu Y, Yin J et al. Materials-Driven Soft Wearable Bioelectronics for Connected Healthcare. Chem Rev, 2024; 124: 455-553.[DOI]
[182] Min J, Tu J, Xu C et al. Skin-interfaced wearable sweat sensors for precision medicine. Chem Rev, 2023; 123: 5049-5138.[DOI]
[183] Liu G, Lv Z, Batool S et al. Biocompatible Material-Based Flexible Biosensors: From Materials Design to Wearable/Implantable Devices and Integrated Sensing Systems. Small, 2023; 19: 2207879.[DOI]
[184] Wang S, Cui Q, Abiri P et al. A self-assembled implantable microtubular pacemaker for wireless cardiac electrotherapy. Sci Adv, 2023; 9: eadj0540.[DOI]
[185] Gao M, Yao Y, Yang F et al. Two-dimensional materials for wireless power transfer. Device, 2023; 1: 100022.[DOI]
[186] Luo Y, Abidian MR, Ahn J-H et al. Technology roadmap for flexible sensors. ACS Nano, 2023; 17: 5211-5295.[DOI]
[187] Ebrahimi N, Bi C, Cappelleri DJ et al. Magnetic actuation methods in bio/soft robotics. Adv Funct Mater, 2021; 31: 2005137.[DOI]
[188] Xu S, Han Z, Yuan K et al. Upcycling chlorinated waste plastics. Nat Rev Methods Primers, 2023; 3: 44.[DOI]
[189] Xu S, Xiao X, Chen J. Stretchable Fiber Strain Sensors for Wearable Biomonitoring. Nat Sci Rev, 2024; 11: nwae173.[DOI]
[190] Hellebrekers T, Chang N, Chin K et al. Soft magnetic tactile skin for continuous force and location estimation using neural networks. IEEE Robot Autmot Lett, 2020; 5: 3892-3898.[DOI]
[191] Xie S, Zhang Y, Jin M et al. High sensitivity and wide range soft magnetic tactile sensor based on electromagnetic induction. IEEE Sensors J, 2020; 21: 2757-2766.[DOI]
[192] Roberts P, Zadan M, Majidi C. Soft tactile sensing skins for robotics. Curr Robot Rep, 2021; 2: 343-354.[DOI]
[193] Liu H, Tian H, Li X et al. Shape-programmable, deformation-locking, and self-sensing artificial muscle based on liquid crystal elastomer and low-melting point alloy. Sci Adv, 2022; 8: eabn5722.[DOI]
[194] Li M, Tang Y, Soon RH et al. Miniature coiled artificial muscle for wireless soft medical devices. Sci Adv, 2022; 8: eabm5616.[DOI]
[195] Shin G, Choi Y, Jeon B et al. Soft Electromagnetic Artificial Muscles Using High-Density Liquid-Metal Solenoid Coils and Bistable Stretchable Magnetic Housings. Adv Funct Mater, 2024; 34: 2302895.[DOI]
[196] Zhang X, Ai J, Zou R, Su B. Compressible and Stretchable Magnetoelectric Sensors Based on Liquid Metals for Highly Sensitive, Self-Powered Respiratory Monitoring. ACS Appl Mater Interfaces, 2021; 13: 15727-15737.[DOI]
[197] Yin J, Hinchet R, Shea H, Majidi C. Wearable soft technologies for haptic sensing and feedback. Adv Funct Mater, 2021; 31: 2007428.[DOI]
[198] Heng W, Solomon S, Gao W. Flexible electronics and devices as human-machine interfaces for medical robotics. Adv Mater, 2022; 34: 2107902.[DOI]
[199] Kim H, Kim YS, Mahmood M et al. Fully Integrated, Stretchable, Wireless Skin-Conformal Bioelectronics for Continuous Stress Monitoring in Daily Life. Adv Sci, 2020; 7: 2000810.[DOI]
[200] Wan L, Lu L, Zhu H et al. Tough and Water-Resistant Bioelastomers with Active-Controllable Degradation Rates. ACS Appl Mater Interfaces, 2024; 16: 6356-6366.[DOI]
[201] Zohar O, Khatib M, Omar R et al. Biointerfaced sensors for biodiagnostics. View, 2021; 2: 20200172.[DOI]
[202] Xu S, Momin M, Ahmed S et al. Illuminating the Brain: Advances and Perspectives in Optoelectronics for Neural Activity Monitoring and Modulation. Adv Mater, 2023; 35: 2303267.[DOI]
[203] Jastrzebska-Perfect P, Chowdhury S, Spyropoulos GD et al. Translational neuroelectronics. Adv Funct Mater, 2020; 30: 1909165.[DOI]
[204] Olvera D, Monaghan MG. Electroactive material-based biosensors for detection and drug delivery. Adv Drug Deliv Rev, 2021; 170: 396-424.[DOI]
[205] Zhang Y, Liu F, Zhang Y et al. Self-powered, light-controlled, bioresorbable platforms for programmed drug delivery. PNSA, 2023; 120: e2217734120.[DOI]
[206] Ma B, Xu C, Cui L et al. Magnetic Printing of Liquid Metal for Perceptive Soft Actuators with Embodied Intelligence. ACS Appl Mater Interfaces, 2021; 13: 5574-5582.[DOI]
[207] Sadeghi A, Afshari E, Hashemi M et al. Brainy Biomaterials: Latest advances in smart Biomaterials to develop the next generation of neural interfaces. Curr Opin Biomed Eng, 2023; 25: 100420.[DOI]
[208] Xu S, Manshaii F, Chen J. Is Deep Brain Imaging on the Brink of Transformation with a Bioluminescence Molecule? BMEMat, 2024; 2: e12115.[DOI]
[209] Xu S, Scott K, Manshaii F, Chen J. Heart-brain connection: How can heartbeats shape our minds? Matter, 2024; 7: 1684-1687.[DOI]
[210] Xu S, Xiao X, Manshaii F et al. Injectable fluorescent neural interfaces for cell-specific stimulating and imaging. Nano Lett, 2024; 24: 4703-4716.[DOI]

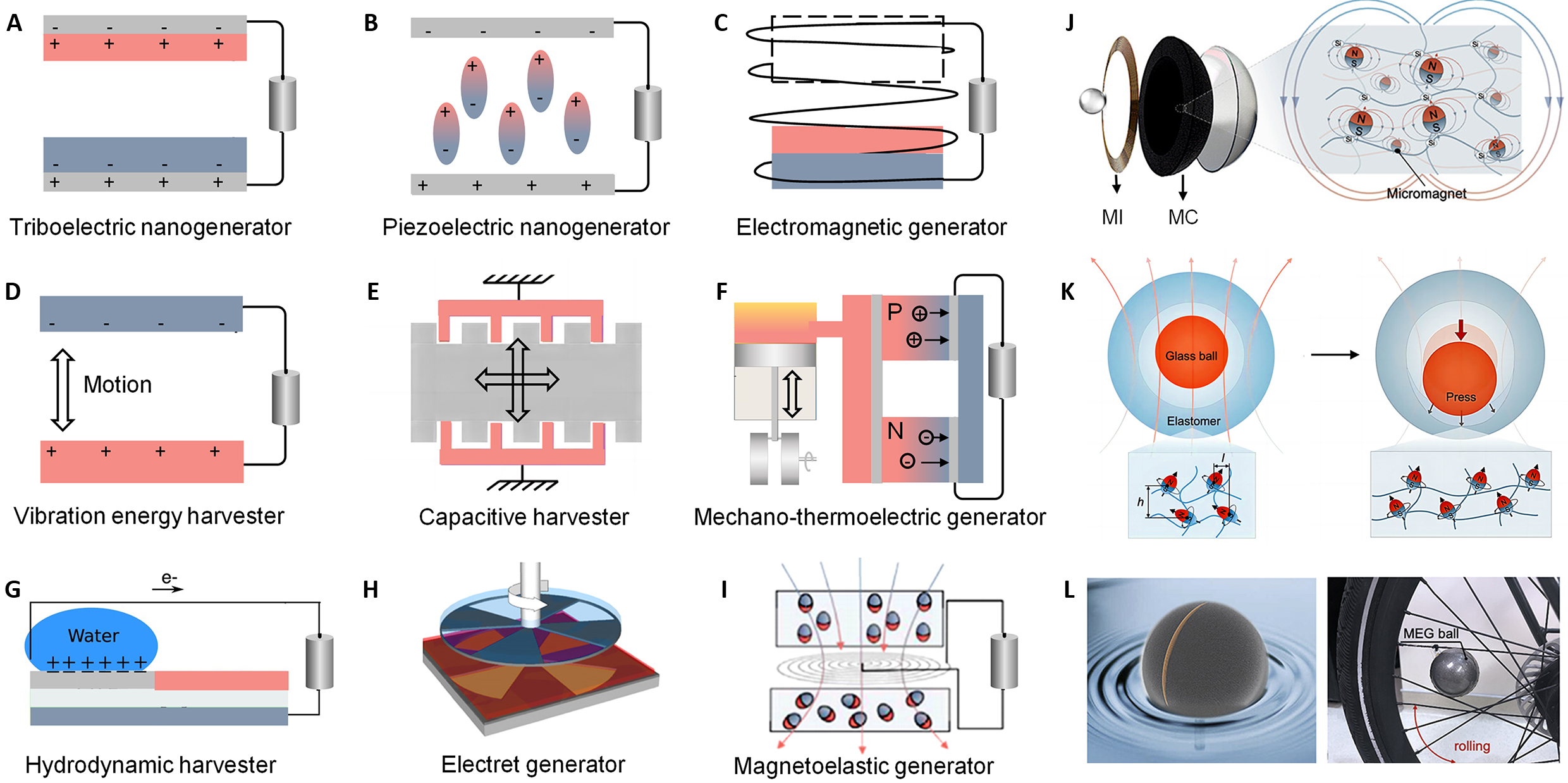
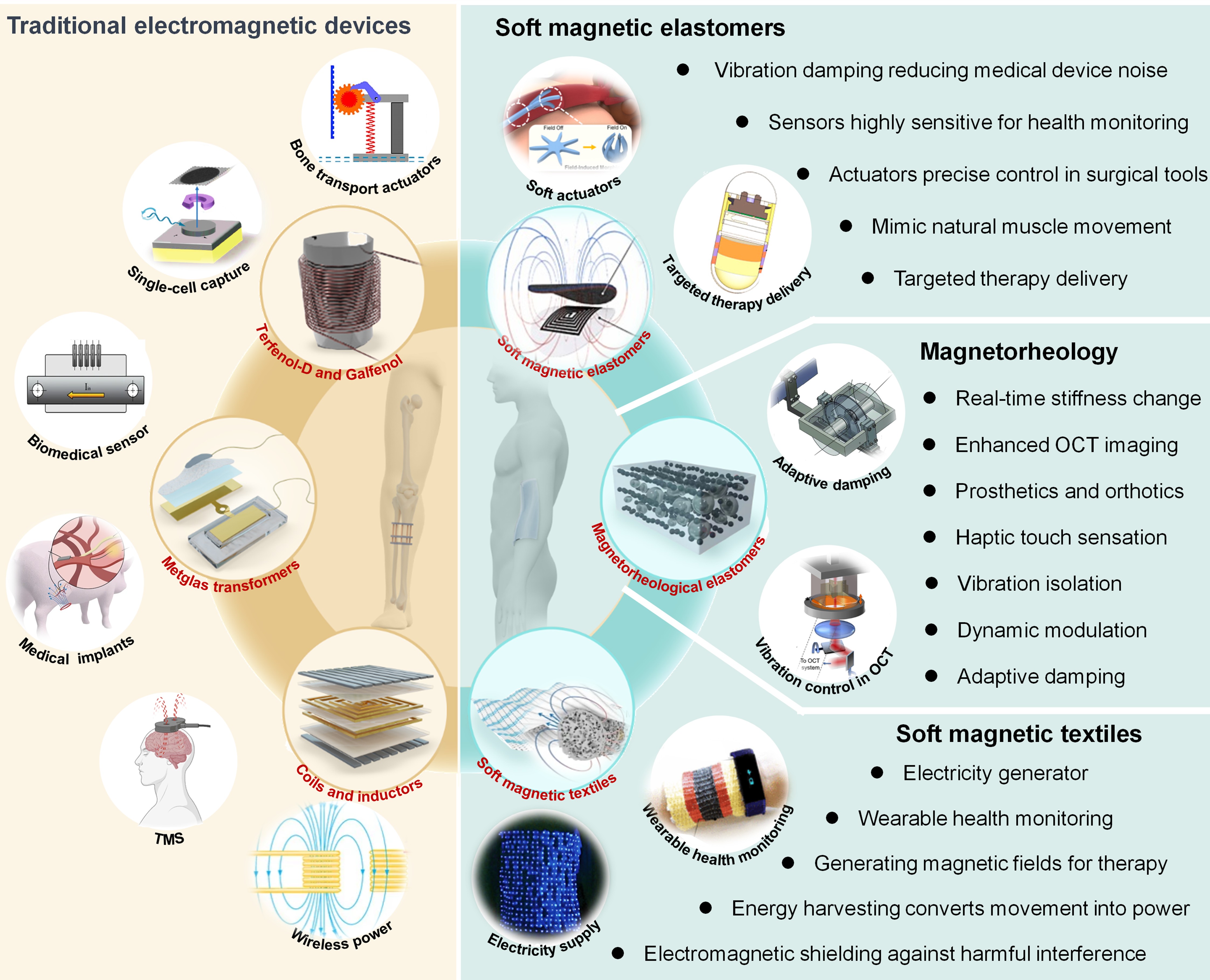

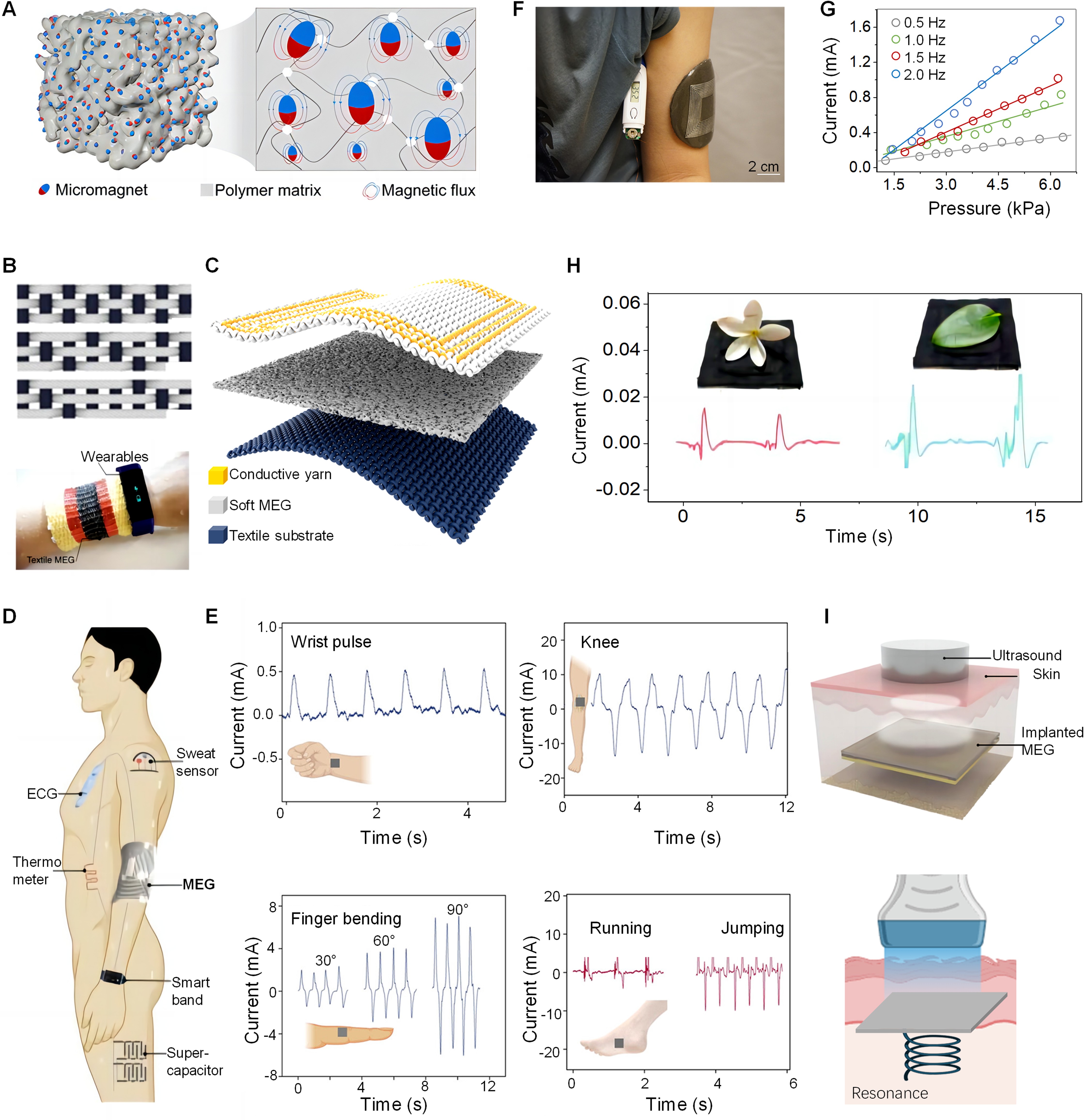
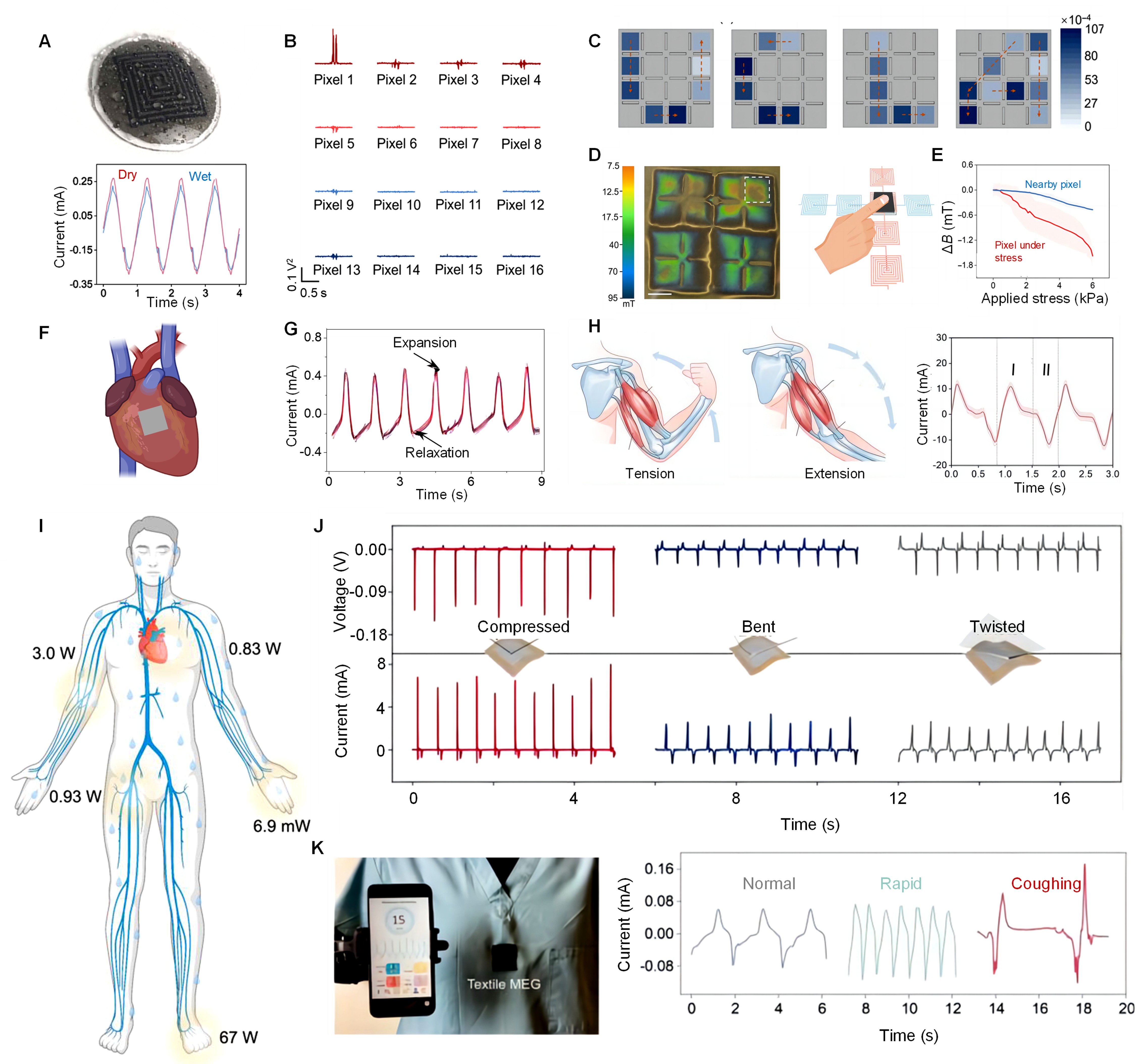
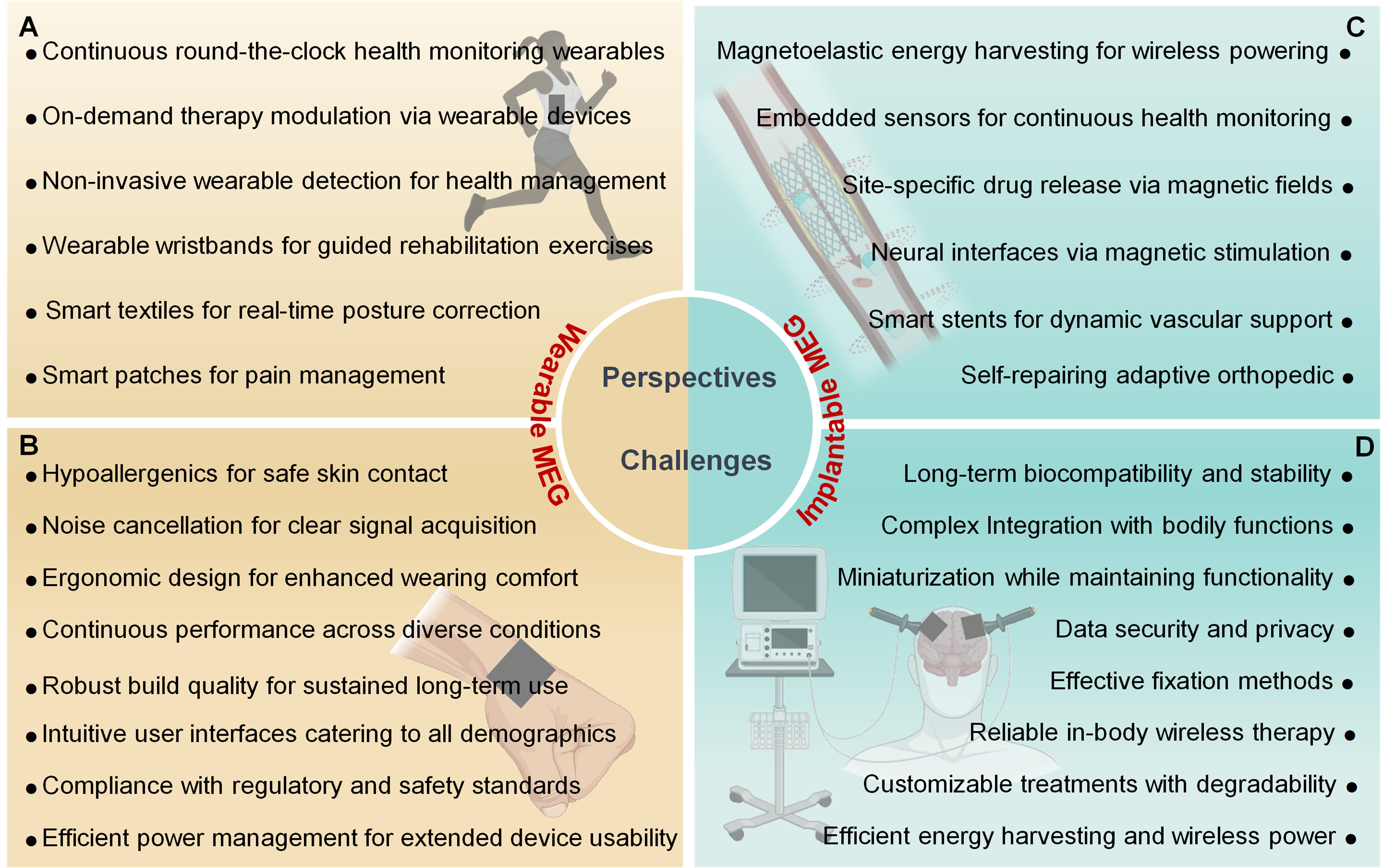




 Copyright ©
Copyright ©