Inflammatory Markers Were Associated with the Comorbidity Patterns
Jiaxin Wu1,2,3#, Ting Fu1,2,3#, Lin Bo4, Shufeng Lei1,2,3*
1Center for Genetic Epidemiology and Genomics, School of Public Health, Suzhou Medical College of Soochow University, Suzhou, Jiangsu Province, China
2Collaborative Innovation Center for Bone and Immunology between Sihong Hospital and Soochow University, Department of Orthopedics, Sihong Hospital, Jiangsu Province, China
3Jiangsu Key Laboratory of Preventive and Translational Medicine for Geriatric Diseases, Soochow University, Suzhou, Jiangsu Province, China
4Department of Rheumatology, The Second Affiliated Hospital of Soochow University, Suzhou, Jiangsu Province, China
#Both authors contributed equally to this work and should be considered co-first authors.
*Correspondence to: Shufeng Lei, PhD, Professor, Director, Center for Genetic Epidemiology and Genomics, School of Public Health, Suzhou Medical College of Soochow University, 199 Renai Road, Suzhou, 215123, Jiangsu Province, China; Email: leisf@suda.edu.cn
DOI: 10.53964/cme.2025006
Abstract
Background: Inflammation is a common risk factor for multiple chronic diseases, but its relationship with multimorbidity that is a coexistence state of multiple medical conditions/diseases is still unknown. This study aimed to investigate the association between multimorbidity and inflammatory markers in a large population-based cohort from the UK Biobank.
Methods: Based on the UK Biobank database, 336,748 eligible participants were included in the final analysis. Data on 14 disease phenotypes (including ICD10 coding and self-report) and 6 inflammatory markers were extracted. We analyzed the correlations of comorbidity patterns and C-reactive protein (CRP), triglyceride (TG), albumin (ALB), alkaline phosphatase (ALP), glycosylated hemoglobin (HbA1c), and apolipoprotein A (APOA1). Nonparametric Spearman correlation analysis and multiple linear regression analysis were used to evaluate the relationship between comorbidity patterns (having 2 or more diseases) and the levels of six inflammatory markers.
Results: The 14 chronic diseases have gender-specific differences in prevalence. All the six biomarkers were significantly associated with comorbidity model, with negatively with ALB and APOA1, and positively TG, CRP, ALP and HbA1c. Logistic regression analyses showed that the associations were significant before and after adjusting for gender, age, smoking, drinking, BMI, and Townsend deprivation index.
Conclusion: This study enhance our understanding of inflammatory pathways in multimorbidity development, clarify the pro-inflammatory biomarker patterns (elevated CRP, TG, ALP, HbA1c and reduced ALB, APOA1) linked to disease clustering, and guide future interventions targeting inflammation modulation or biomarker-driven risk stratification.
Keywords: comorbidity, information indicators, cohort study, correlation, risk factors
1 INTRODUCTION
Multimorbidity is a coexistence state of multiple medical conditions/diseases in an individual[1,2], and is a growing global challenge with substantial effects on individuals, careers and society. The prevalence of multimorbidity is estimated to be up to 67.8% in adults aged 65 years and older by 2035 in the UK[3]. The mechanisms underlying the development of multimorbidity are complex and probably related to aging, heredity, and broader determinants of health, such as socioeconomic deprivation. Inflamm-aging is a highly significant risk factor for morbidity and mortality in older individuals. Inflammation is closely associated with the initiation and progression of age-related diseases such as Alzheimer's disease, cardiovascular disease, type II diabetes, frailty, sarcopenia, osteoporosis, and cancer. Inflammation can be caused by a variety of causes, including blood clots, immune system disorders, cancer, infection, chemical exposure and physical injury[4]. Under normal conditions, inflammation and anti-inflammation are balanced, but this balance is easily disrupted, especially among the elderly. The existence of certain social, psychological, environmental and biological factors is related to the resolution of inflammation, which in turn promotes a low-grade, non-infectious (i.e., "sterile") systemic chronic inflammation state. The chronic inflammatory process produces a large number of free radicals and activates the damage and deterioration process of target cells and organs, leading to a variety of chronic diseases[5]. To date, previous studies have found more than 200 kinds of chronic diseases caused by inflammation, such as cardiovascular disease, cancer, diabetes, chronic kidney disease, nonalcoholic fatty liver disease, autoimmune and neurodegenerative diseases[6-8]. However, the association between chronic inflammation and multimorbidity is still unknown.
C-reactive protein (CRP) concentration in the condition of inflammation, accelerates the release of interleukin-1 (IL-1), interleukin-6 (IL-6), interleukin-8 (IL-8) and tumor necrosis factor-α (TNP-α)[9], and participates in the systemic response of inflammation. CRP is also thought to be involved in endothelial dysfunction, vascular sclerosis, and blood pressure[10], and it affects vascular remodeling and cardiac remodeling development in adverse conditions of medium-pressure overload. Prospective nested case-control studies have shown that it is associated with an increased risk of myocardial infarction, stroke, sudden cardiac death, and peripheral artery disease and is one of the best-known biomarkers of cardiovascular disease[11]. Triglycerides (TG), an important component of blood lipids, are closely related to the occurrence and development of many diseases. Formed by low-density lipoprotein particles increased triglyceride levels are considered to be an independent risk factor for cardiovascular disease[12]. Studies have shown that triglycerides can trigger inflammatory responses and oxidative stress, and the genetic effects of single nucleotide polymorphisms on triglyceride levels are independently associated with the risk of atherosclerotic disease, proving a causal association[13]. High triglyceride levels were associated with an increased risk of type 2 diabetes[14]. Albumin (ALB) is the most abundant protein in the plasma and plays an important role in the transport of free fatty acids, bilirubin and drugs[15]. In normal individuals, liver cells synthesize approximately 15g of albumin per, day to maintain steady-state plasma concentrations (4g/100mL). Decreased albumin synthesis results in low serum albumin levels. Low serum albumin levels are a common characteristic of chronic diseases including cancer and inflammatory diseases. Alkaline phosphatase (ALP) is widely distributed in the serum and on the surface of most cells. It plays an indispensable role in bone metabolism and hepatobiliary diseases. However, emerging evidence suggests that in diseases associated with inflammation, such as hypertension, type 2 diabetes, abnormal cholesterol and other cardiovascular and metabolic diseases, the content or activity of ALP increases[16]. Glycosylated hemoglobin (HbA1c) is the product of the combination of hemoglobin in red blood cells and sugars (glucose) in serum through a non-enzymatic reaction. HbA1c is currently the gold standard for assessment of glycemic control and response to therapy in patients with type 1 (T1D) and type 2 (T2D) diabetes. During the past three decades, a large number of epidemiological studies have shown that high HbA1c is an important risk indicator of diabetic complications[17]. In addition, high levels of HbA1c increase blood viscosity and accelerate atherosclerosis, which in turn leads to cardiovascular events such as coronary heart disease, myocardial infarction and heart failure[18]. Apolipoprotein A1 (APOA1) is one of the main apolipoproteins in High density lipoprotein (HDL). The APOA1 content in patients with cancer and cardiovascular risk was significantly higher than that in healthy individuals. APOA1 is mainly involved in biological processes such as cholesterol transport, endothelial cell apoptosis, platelet activation, vasodilation induction, and anti-oxidation and anti-inflammation[19]. Currently, the correlations between the above inflammation markers and multimorbidity remain unknown.
Motivated by the above situation, this study performed correlation analyses between the above inflammatory indicators and multimorbidity in the large UK Biobank (UKB) cohort and aimed to determine whether these inflammatory markers were correlated with comorbidity patterns and to identify potential risk or protective factors among the markers.
2 MATERIALS AND METHODS
2.1 Data Sources
Based on the data downloaded from UKB database, a total of 502,422 participants were included in this study. In this study, 14 common diseases/conditions and 6 inflammatory markers were selected. The 14 selected diseases/conditions were listed in the English Longitudinal Study of Ageing wave 7 (14-year follow-up) and are considered to be major diseases in middle-aged and older people and the main cause of long-term illness in people aged 65 years and over in other national surveys. It mainly includes hypertension, type 2 diabetes, cancer, chronic obstructive pulmonary disease, heart disease, stroke, mental health problems (mainly stress and anxiety), arthritis, asthma, high cholesterol, cataracts, Parkinson's disease, hip fractures and depression (see https://www.elsa-project.ac.uk/data-and-docu for details).
Data fields from the UKB data were selected for this section: (1) age (field 21,003: age of the participant at the time of assessment), (2) smoking (field 22,506), (3) drink frequency (field 1,558), (4) ethnic group (field 21,000: Ethnic background), (5) International Classification of Diseases, Tenth Revision (ICD-10) code (Field 41,270: Diagnosis -- ICD10), (6) sex (field: 31), (7) Townsend Deprivation Index (TDI) (field 189: Townsend deprivation index at recruitment), (8) Body mass index (BMI) (field 21001) and (9) non-cancer disease coding, self-reported disease (field: 20,002). The patients with ≥2 above 2 diseases/conditions were defined as comorbidity pattern.
Tables S1 and S2 list the details of the six chronic inflammatory indicators and 14 diseases/conditions involved in this study, and the data were obtained from the test data of the blood samples of the study subjects. A detailed description of all the relevant data fields can be found in a UKB: https://biobank.ndph.ox.ac.uk/showcase/.
2.2 Statistical Analysis
The correlations between comorbidity patterns and C-reactive protein (CRP), triglyceride (TG), albumin (ALB), alkaline phosphatase (ALP), glycosylated hemoglobin (HbA1c), apolipoprotein A1 (ApoA1) were analyzed. After excluding the missing and extreme values (Defined as values >3 SDs from the mean), 336,748 participants were included in the final analysis. Nonparametric Spearman correlation analysis and multiple logistic regression analysis were used to evaluate the correlation between comorbidity patterns (having 2 or more diseases) and the levels of six inflammatory markers (ALB, ALP, CRP, TG, ApoA1, HbA1c). All analyses were performed using R 4.4.0 with a statistical significance threshold of P<0.05.
3 RESULTS
3.1 Differences in Disease Prevalence and Baseline Characteristics
The baseline characteristics of the study subjects are shown in Table 1. Age, gender, smoking, different drinking frequency, BMI, TDI and six inflammatory markers were significantly different between the non-Multimorbidity pattern and Multimorbidity pattern (P<0.05). Table 2 shows the prevalence of 14 chronic diseases in participants of different genders. In terms of gender, the prevalence of chronic diseases in males was in the descending order of hypertension (26.9%), asthma (15.1%), heart problems (12.4%), depression (11.2%), high cholesterol (10.7%), arthritis (8.0%), cataract (7.0%), mental problems (6.4%), cancer (4.4%), type 2 diabetes (4.3%), chronic obstructive pulmonary disease (3.6) %), hip fracture (1.4%), stroke (1.2%), Parkinson's disease (0.5%); The prevalence of chronic diseases in women, in order, Hypertension (35.7%), heart problems (22.8%), high cholesterol (17.0%), asthma (13.1%), depression (11.2%), type 2 diabetes (7.3%), cataract (6.1%), arthritis (5.9%), cancer (5.0%), chronic obstructive pulmonary disease (4.9%), mental problems (4.0%), stroke (2.2%), Parkinson's disease (1.0%), hip fracture (0.9%). Based on the chi-square test, the prevalence rates of heart problems, high cholesterol, type 2 diabetes, chronic obstructive pulmonary disease, stroke and Parkinson's disease were higher in women than in men (P<0.001), while the prevalence rates of asthma, depression, arthritis, mental health problems and hip fractures were higher in men than in women.
Table 1. The Basic Characteristics of the Study Subjects Stratified by the Multimorbidity State
|
Overall (n=336,748) |
Multimorbidity Pattern |
|||
No (n=227,256) |
Yes (n=109,492) |
P |
SMD |
||
Age, mean (SD) |
56.90 (8.00) |
55.26 (7.98) |
60.31 (6.91) |
<0.001 |
0.99 |
Gender, n (%) |
|
|
|
0.005 |
0.021 |
Male |
156,622 (46.5) |
100,120 (44.1) |
56,502 (51.6) |
|
|
Female |
180,726 (53.5) |
127,136 (55.9) |
52,990 (48.4) |
|
|
Smoke, n (%) |
|
|
|
<0.001 |
0.276 |
Prefer not to answer |
1,163 (0.3) |
585 (0.3) |
578 (0.5) |
|
|
No |
183,035 (54.4) |
133,055 (58.5) |
49,980 (45.6) |
|
|
Yes, almost all days |
118,383 (35.2) |
72,784 (32.0) |
45,599 (41.6) |
|
|
Only occasionally |
34,167 (10.1) |
20,832 (9.2) |
13,335 (12.2) |
|
|
Drink frequency, n (%) |
|
|
|
<0.001 |
0.481 |
Prefer not to answer |
229 (0.1) |
95 (0.0) |
134 (0.1) |
|
|
Daily or almost daily |
70,778 (21.1) |
47,458 (20.9) |
23,320 ( 21.3) |
|
|
3 to 4 times a month |
81,149 (24.1) |
58,323 (25.7) |
22,826 ( 20.8) |
|
|
1 to 2 times a week |
89,291 (26.5) |
62,585 (27.5) |
26,706 ( 24.4) |
|
|
1 to 3 times a month |
37,417 (11.1) |
25,430 (11.2) |
11,987 ( 10.9) |
|
|
Special occasions only |
35,682 (10.6) |
21,283 ( 9.4) |
14,399 ( 13.2) |
|
|
Never |
22,202 (6.6) |
12,082 ( 5.3) |
10,120 (9.2) |
|
|
BMI (Kg/m2) |
27.45 (4.75) |
26.72 (4.33) |
28.96 (5.22) |
<0.001 |
0.608 |
TDI |
-1.56 (2.93) |
-1.74 (2.82) |
-1.20 (3.12) |
<0.001 |
0.272 |
TG (mmol/L) |
1.76 (1.02) |
1.67 (0.97) |
1.94 (1.09) |
<0.001 |
|
CRP (mg/L) |
2.60 (4.36) |
2.27 (3.88) |
3.29 (5.13) |
<0.001 |
|
ALB (g/L) |
45.23 (2.60) |
45.36 (2.55) |
44.96 (2.68) |
<0.001 |
|
ALP (g/L) |
83.68 (26.27) |
81.77 (25.26) |
87.63 (27.83) |
<0.001 |
|
ApoA1 (g/L) |
1.54 (0.27) |
1.56 (0.27) |
1.51 (0.27) |
<0.001 |
|
HbA1c (mmol/mol) |
35.96 (6.47) |
34.88 (4.77) |
38.21 (8.60) |
<0.001 |
|
Notes: Mean(SD): Mean (standard deviation); SMD: Standardized mean difference; BMI: Body mass index; TDI: Townsend deprivation index; TG: Triglyceride; CRP: C-reaction protein; ALB: Albumi; ALP: Alkaline phosphatase; APOA1:Apolipoprotein A1; HbA1c: Glycosylated hemoglobin.
Table 2. Prevalence of 14 Chronic Diseases Among Participants Stratified by Gender
Diseases/conditions |
Female |
Male |
P |
||
N (%) |
Position |
N (%) |
Position |
||
Hypertension |
59,647 (26.9) |
1 |
67,192 (35.7) |
1 |
<0.001 |
Type 2 diabetes |
9,511 (4.3) |
10 |
13,753 (7.3) |
6 |
<0.001 |
Cancer |
9,837 (4.4) |
9 |
9,502 (5.0) |
9 |
<0.001 |
COPD |
7,879 (3.6) |
11 |
9,334 (4.9) |
10 |
<0.001 |
Heart problem |
27,440 (12.4) |
3 |
42,822 (22.8) |
2 |
<0.001 |
Stroke |
2,631 (1.2) |
13 |
4,072 (2.2) |
12 |
<0.001 |
Psychiatric problems |
14,142 (6.4) |
8 |
7,586 (4.0) |
11 |
<0.001 |
Arthritis |
17,631 (8.0) |
6 |
11,081 (5.9) |
8 |
<0.001 |
Asthma |
33,238 (15.0) |
2 |
24,625 (13.1) |
4 |
<0.001 |
High cholesterol |
23,689 (10.7) |
5 |
31,966 (17.0) |
3 |
<0.001 |
Cataract |
15,406 (7.0) |
7 |
11,402 (6.1) |
7 |
<0.001 |
Parkinson’s disease |
1,207 (0.5) |
14 |
1,882 (1.0) |
13 |
<0.001 |
Hip fracture |
3,167 (1.4) |
12 |
1,780 (0.9) |
14 |
<0.001 |
Depression |
24,756 (11.2) |
4 |
14,097 (7.5) |
5 |
<0.001 |
3.2 Association Analysis of Inflammatory Markers with Disease Patterns
First, we tested the effect of different inflammatory factors on single diseases, as shown in Table S3, after correcting for age, sex, smoking, drinking, BMI, and TDI, the effect of inflammatory factors on single diseases remains significant. Table 3 shows the correlations between six inflammatory biomarkers and comorbidity patterns. As we expected all the six biomarkers were significantly associated with comorbidity pattern, with negatively with ALB and ApoA1, and positively TG, CRP, ALP and HbA1c. Logistic regression analyses (Table 4) showed that the associations were significant before and after adjusting for gender, age, smoking, drinking, BMI, and TDI. Then, we conducted comprehensive sensitivity analyses to investigate specific comorbidity patterns: (1) the cardiovascular-metabolic group (participants with ≥2 of hypertension, type 2 diabetes, high cholesterol, or heart problems) and (2) the psycho-metabolic group (participants with ≥1 metabolic disease plus ≥1 psychiatric condition). Our results demonstrated significant associations between all six inflammatory markers and both comorbidity subgroups (Table S4).
Table 3. Correlation Analysis between Morbidity Pattern and Six Inflammation Indicators
|
TG |
CRP |
ALB |
ALP |
ApoA1 |
HbA1c |
||||||
r |
P |
r |
P |
r |
P |
r |
P |
r |
P |
r |
P |
|
Morbidity patterns |
0.141 |
<0.001 |
0.165 |
<0.001 |
-0.069 |
<0.001 |
0.113 |
<0.001 |
-0.091 |
<0.001 |
0.233 |
<0.001 |
Notes: TG: Triglyceride; CRP: C-reaction protein; ALB: Albumin; ALP: Alkaline phosphatase; APOA1: Apolipoprotein A1; HbA1c: Glycosylated hemoglobin.
Table 4. Logistic Analysis between Inflammatory Markers and Morbidity Pattern
|
Model 1 |
Model 2 |
||
OR (95%Cl) |
P |
OR (95%Cl) |
P |
|
TG |
1.159 (1.150,1.168) |
<2E-16 |
1.044 (1.035,1.053) |
<2E-16 |
CRP |
1.027 (1.025,1.029) |
<2E-16 |
1.014 (1.012,1.016) |
<2E-16 |
ALB |
0.965 (0.962,0.968) |
<2E-16 |
1.010 (1.007,1.014) |
<2E-16 |
ALP |
1.005 (1.005,1.006) |
<2E-16 |
1.002 (1.001,1.002) |
5.59E-10 |
ApoA1 |
0.793 (0.769,0.816) |
<2E-16 |
0.847 (0.817,0.878) |
<2E-16 |
HbA1c |
1.088 (1.087,1.090) |
<2E-16 |
1.051 (1.050,1.053) |
<2E-16 |
Notes: Model 1: unadjusted covariates. Model 2: adjusted for age, sex, smoking, drinking, BMI, and TDI.
3.3 Regression Model Robustness Validation
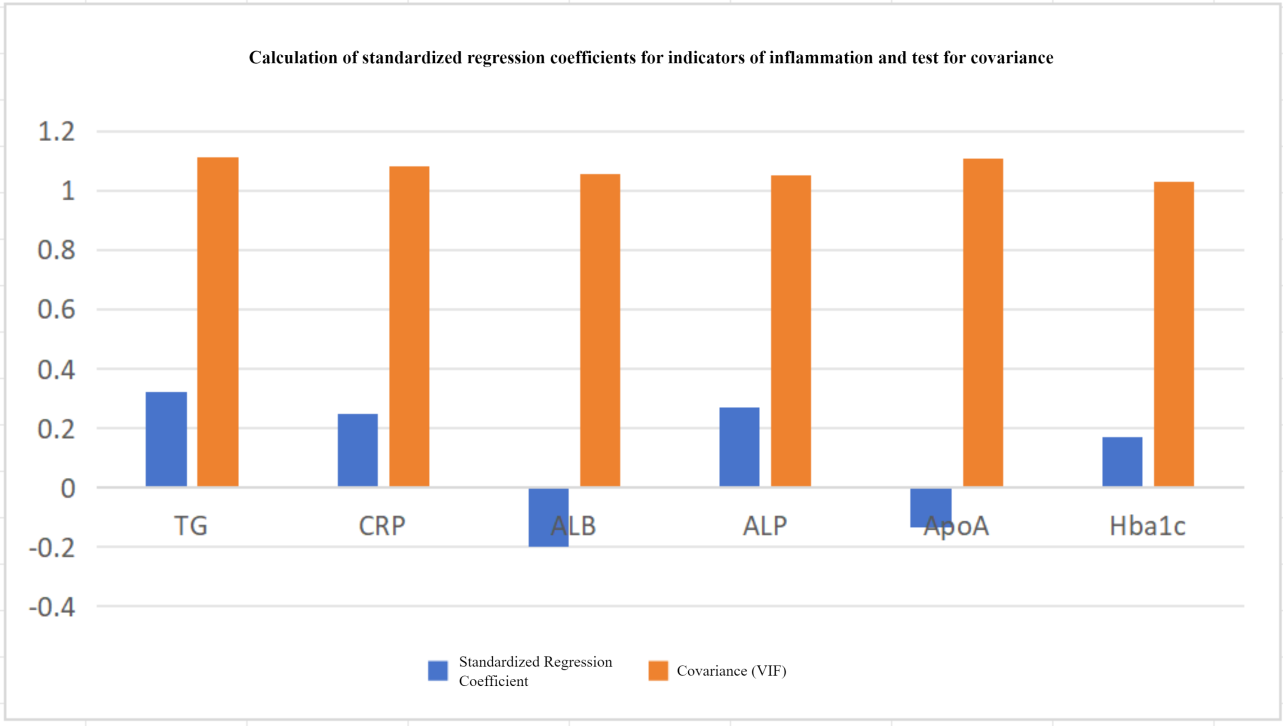
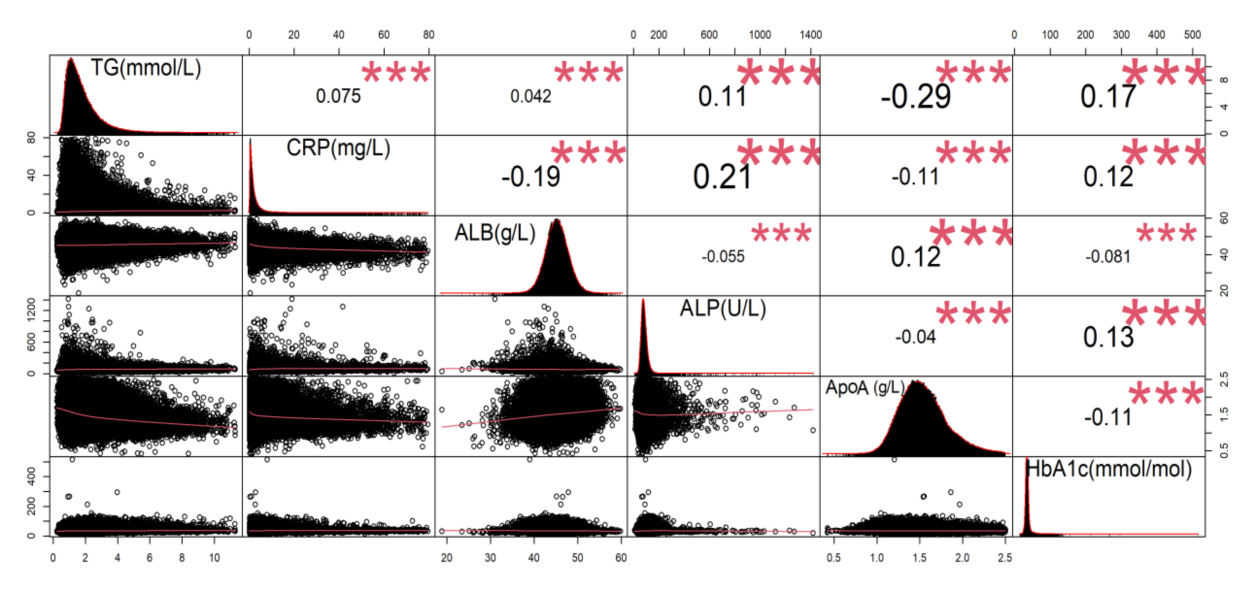
In order to verify the reliability of the regression model, we performed correlation analysis and covariance tests on the independent variables. Correlation analyses showed that the correlation coefficients between the independent variables were much less than 1, which suggested that there is no high correlation between the six inflammation indicators (Figure 1). Then, we carried out the covariance test and the Variance Inflation Factor (VIF) values were all around 1, much less than 10, suggested that the effect of covariance between independent variables can be excluded. And we calculated standard regression coefficients for inflammation indicators, in which TG had the largest standard regression coefficient (0.321), which indicates that the effect of TG on the coexistence of multiple diseases is the largest, followed by ALP (as shown in Figure 2).
|
Figure 1. Correlation Coefficients for the Six Indicators of Inflammation Markers.
|
Figure 2. VIF Analysis of Variance Expansion Factor for Six Indicators of Inflammation.
4 DISCUSSION
This study represented the first effort of investigating the relationship between inflammation and comorbidity patterns based on 14 kinds of diseases/conditions. Spearman rank correlation analysis and multiple logistic regression analysis were used to analyze the correlation between comorbidity patterns and inflammatory markers. In general, the prevalence of hypertension was different between men and women, and the prevalence of hypertension was the highest among the diseases/conditions in both men and women. Five inflammatory markers (namely triglyceride, C-reactive protein, albumin, alkaline phosphatase and apolipoprotein A) were the risk factors for the comorbidity pattern.
A few previous studies have checked the link between chronic inflammation and a variety of diseases, most studies mainly focus on the relationship between inflammation and single disease. For example, Friedman et al. found that IL-6 and CRP were positively associated with individual chronic diseases including asthma, arthritis, hypertension, diabetes, neurological disorders, cancer, stroke, and heart problems[20].
IL-6 accelerates atherosclerosis by promoting hepatic production of CRP and fibrinogen. It also induces the expression of endothelial cell adhesion molecules (VCAM-1/ICAM-1) and promotes monocyte infiltration, thus affecting cardiovascular diseases[21]. IL-6 can affect diabetes by inhibiting adipocyte insulin receptor substrate-1 (IRS-1) phosphorylation, promoting hepatic gluconeogenesis (via STAT3-SOCS3 pathway), and impairing skeletal muscle glucose uptake[22]. CRP and the associated risk of dementia and further research indicated that the monomeric form of CRP can infiltrate the BBB/be released from damaged micro-vessels to access the brain[23].
While ApoA1, as one of the main types of apolipo-proteins, is involved in lipid metabolism and is associated with the development of chronic diseases, such as diabetes, heart problems and stroke[24-27]. ApoA1 is the major apolipoprotein of HDL and is known to have anti-atherogenic effects by reversing cholesterol transport, preventing endothelial dysfunction, and inhibiting oxidative stress[28]. ApoA1 directly interacts with ATP-binding cassette transporter A1 (ABCA1) and ATP-binding cassette transporter G1 (ABCG1) to promote cholesterol efflux[29]. ApoA1 interacts directly with ABCA1 and ABCG1 to promote cholesterol efflux. In addition, ApoA1 exerts anti-inflammatory effects through cholesterol efflux or other mechanisms, and its circulating levels are significantly reduced during the acute phase response to inflammation[30]. At the same time, ApoA1 protects against liver injury by inhibiting macrophage death via the TLR4-NF-κB pathway[31].
Alkaline phosphatase is more used to evaluate liver-related diseases[32,33]. Albumin is the most abundant plasma protein and is widely used as an indicator of malnutrition[34], which in turn is associated with cardiovascular disease[35]. Low albumin levels associated with poor recovery after acute pathological, and known pathology of acute inflammation can reduce serum albumin level[36], is associated with shortened life in the elderly. Hypoalbuminemia in adults is defined as serum albumin levels below 3.5g/dL, but levels below 2.5g/dL generally reflect liver and kidney function, correlate with liver and kidney disease, and are considered clinically significant[37]. In addition, a UKB-based cross-sectional analysis of cardiometabolic multiple diseases showed that the subgroup with higher triglycerides was at risk for diabetes[38]. In a study of type 2 diabetes patients coexisting with multiple diseases, it was shown that there was a certain association between multiple diseases and HbA1c[39,40].
Of course, this study also has some limitations. First, we only drew the correlation between the six inflammatory indicators and the comorbidity pattern as a whole, and did not specifically analyze each disease with the six inflammatory indicators in turn, which may cause some influence on the results. Second, while we found statistically significant associations between all six inflammatory markers and comorbidity patterns, the clinical significance of some associations may be limited. At the same time, we divided the individuals with one disease and those without the disease into the same group as the control group, and such groups may cause some errors. Therefore, more studies, such as genome-wide association studies using cohort data on comorbidity patterns and genetic analysis of genome-wide association study data on inflammatory markers and comorbidities, are needed to explore causal associations or genetic overlap between them.
5 CONCLUSION
Through cross-sectional analysis using data from a large population-based cohort study in UKB, this study found that comorbidity patterns with ≥2 diseases/conditions were associated with six inflammatory markers, among which TG, ALB, ALP, CRP and HbA1c were risk factors (OR>1), while APOA1 was a protective factor (OR<1). As the conclusion is based on cross-sectional studies only, it may be affected by other unknown confounding factors. In the future, more specific analyses are needed to verify the relationship between inflammatory markers and comorbidity patterns.
Acknowledgements
We thank all the studies for making the summary association statistics data publicly available.
Conflicts of Interest
The authors declared no conflict of interest.
Ethics Statement
UK Biobank data has approval from the North West - Haydock Research Ethics Committee (REC reference: 21/NW/0157). This research has been conducted with the UK Biobank Resource under Project 76875.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files. Data are available in https://biobank.ndph.ox.ac.uk/showcase/.
Copyright Permissions
Copyright © 2025 The Author(s). Published by Innovation Forever Publishing Group Limited. This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.
Lei S and Fu T conceived the design of the study; Fu T, Wu J and Bo L cleared up the datasets and mainly performed the data analyses; Lei S and Fu T drafted and revised the manuscript, all authors approved the manuscript and provided relevant suggestions.
Abbreviation List
ABCA1, ATP-binding cassette transporter A1
ABCG1, ATP-binding cassette transporter G1
ALP, Alkaline phosphatase
ApoA1, Apolipoprotein A
BMI, Body mass index
HbA1c, Glycosylated hemoglobin
TDI, Townsend Deprivation Index
UKB, UK Biobank
References
[1] Mino-Leon D, Reyes-Morales H, Doubova SV et al. Multimorbidity Patterns in Older Adults: An Approach to the Complex Interrelationships Among Chronic Diseases. Arch Med Res, 2017; 48: 121-127.[DOI]
[2] Piotrowicz K, Pac A, Skalska A et al. Patterns of multimorbidity in 4588 older adults: implications for a nongeriatrician specialist. Pol Arch Intern Med, 2021; 131.[DOI]
[3] Kingston A, Robinson L, Booth H et al. Projections of multi-morbidity in the older population in England to 2035: estimates from the Population Ageing and Care Simulation (PACSim) model. Age Ageing, 2018; 47: 374-380.[DOI]
[4] Roe K. An inflammation classification system using cytokine parameters. Scand J Immunol, 2021; 93: e12970.[DOI]
[5] Furman D, Campisi J, Verdin E et al. Chronic inflammation in the etiology of disease across the life span. Nat Med, 2019; 25: 1822-1832.[DOI]
[6] Heidland A, Klassen A, Rutkowski P et al. The contribution of Rudolf Virchow to the concept of inflammation: what is still of importance? J Nephrol, 2006; 19 Suppl 10: S102-109.
[7] Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet (London, England), 2018; 392: 1736-1788.[DOI]
[8] Aggarwal BB, Shishodia S, Sandur SK et al. Inflammation and cancer: how hot is the link? Biochem Pharmacol, 2006; 72: 1605-1621.[DOI]
[9] Black S, Kushner I, Samols D. C-reactive Protein. J Biol Chem, 2004; 279: 48487-48490.[DOI]
[10] Hage FG. C-reactive protein and hypertension. J Hum Hypertens, 2014; 28: 410-415.[DOI]
[11] Ridker PM, Rifai N, Rose L et al. Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first cardiovascular events. New Engl J Med, 2002; 347: 1557-1565.[DOI]
[12] Miller M, Stone NJ, Ballantyne C et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation, 2011; 123: 2292-2333.[DOI]
[13] Tada H, Kawashiri MA. Genetic Variations, Triglycerides, and Atherosclerotic Disease. J Atheroscler Thromb, 2019; 26: 128-131.[DOI]
[14] Zhou T, Liu X, Liu Y et al. Meta-analytic evaluation for the spatio-temporal patterns of the associations between common risk factors and type 2 diabetes in mainland China. Medicine, 2019; 98: e15581.[DOI]
[15] Chojkier M. Inhibition of albumin synthesis in chronic diseases: molecular mechanisms. J Clin Gastroenterol, 2005; 39: S143-146.[DOI]
[16] Kim JH, Lee HS, Park HM et al. Serum alkaline phosphatase level is positively associated with metabolic syndrome: A nationwide population-based study. Clin Chim Acta, 2020; 500: 189-194.[DOI]
[17] Martinez M, Santamarina J, Pavesi A et al. Glycemic variability and cardiovascular disease in patients with type 2 diabetes. BMJ Open Diab Res Ca, 2021; 9.[DOI]
[18] Hoffmann AP, Honigberg MC. Glycated Hemoglobin as an Integrator of Cardiovascular Risk in Individuals Without Diabetes: Lessons from Recent Epidemiologic Studies. Curr Atheroscler Rep, 2022; 24: 435-442.[DOI]
[19] Cho SMJ, Lee H, Shim JS et al. Association of Snoring with Prediabetes and Type 2 Diabetes Mellitus: The Cardiovascular and Metabolic Diseases Etiology Research Center Cohort. Diabetes Metab J, 2020; 44: 687-698.[DOI]
[20] Friedman EM, Christ SL, Mroczek DK. Inflammation Partially Mediates the Association of Multimorbidity and Functional Limitations in a National Sample of Middle-Aged and Older Adults: The MIDUS Study. J Aging Health, 2015; 27: 843-863.[DOI]
[21] Ridker PM, Rane M. Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ Res, 2021; 128: 1728-1746.[DOI]
[22] Akbari M, Hassan-Zadeh V. IL-6 signalling pathways and the development of type 2 diabetes. Inflammopharmacology, 2018; 26: 685-698.[DOI]
[23] Cooper J, Pastorello Y, Slevin M. A meta-analysis investigating the relationship between inflammation in autoimmune disease, elevated CRP, and the risk of dementia. Front Immunol, 2023; 14: 1087571.[DOI]
[24] Casillas-Muñoz F, Valle Y, Muñoz-Valle JF et al. APOA1 and APOB polymorphisms and apolipoprotein concentrations as biomarkers of risk in acute coronary syndrome: Relationship with lipid-lowering therapy effectiveness. Med Clin, 2018; 151: 1-7.[DOI]
[25] Siegel RD, Cupples A, Schaefer EJ et al. Lipoproteins, apolipoproteins, and low-density lipoprotein size among diabetics in the Framingham offspring study. Metabolism, 1996; 45: 1267-1272.[DOI]
[26] As S, Sahukar S, Murthy J et al. A study of serum apolipoprotein A1, apolipoprotein B and lipid profile in stroke. J Clin Diagn Res, 2013; 7: 1303-1306.[DOI]
[27] Sniderman AD, Thanassoulis G, Glavinovic T et al. Apolipoprotein B Particles and Cardiovascular Disease: A Narrative Review. JAMA Cardiol, 2019; 4: 1287-1295.[DOI]
[28] Assmann G, Nofer JR. Atheroprotective effects of high-density lipoproteins. Annu Rev Med, 2003; 54: 321-341.[DOI]
[29] Linsel-Nitschke P, Tall AR. HDL as a target in the treatment of atherosclerotic cardiovascular disease. Nat Rev Drug Discov, 2005; 4: 193-205.[DOI]
[30] Rosenson RS, Brewer HB Jr, Ansell B et al. Translation of high-density lipoprotein function into clinical practice: current prospects and future challenges. Circulation, 2013; 128: 1256-1267.[DOI]
[31] Chen RX, Jiang WJ, Liu SC et al. Apolipoprotein A-1 protected hepatic ischaemia-reperfusion injury through suppressing macrophage pyroptosis via TLR4-NF-κB pathway. Liver Int, 2023; 43: 234-248.[DOI]
[32] Villén N, Guisado-Clavero M, Fernández-Bertolín S et al. Multimorbidity patterns, polypharmacy and their association with liver and kidney abnormalities in people over 65 years of age: a longitudinal study. BMC Geriatr, 2020; 20: 206.[DOI]
[33] Us Altay D, Kaya Y, Mataraci Değirmenci D et al. Non-alcoholic fatty liver disease: The importance of physical activity and nutrition education-A randomized controlled study. J Gastroen Hepatol, 2024; 39: 2723-2734.[DOI]
[34] Cabrerizo S, Cuadras D, Gomez-Busto F et al. Serum albumin and health in older people: Review and meta analysis. Maturitas, 2015; 81: 17-27.[DOI]
[35] Huang T, An Z, Huang Z et al. Serum albumin and cardiovascular disease: a Mendelian randomization study. BMC Cardiovasc Disor, 2024; 24: 196.[DOI]
[36] Soeters PB, Wolfe RR, Shenkin A. Hypoalbuminemia: Pathogenesis and Clinical Significance. Jpen-Parenter Enter, 2019; 43: 181-193.[DOI]
[37] Gatta A, Verardo A, Bolognesi M. Hypoalbuminemia. Intern Emerg Med, 2012; 7 Suppl 3: S193-199.[DOI]
[38] Mulugeta A, Hyppönen E, Ala-Korpela M et al. Cross-sectional metabolic subgroups and 10-year follow-up of cardiometabolic multimorbidity in the UK Biobank. Sci Rep, 2022; 12: 8590.[DOI]
[39] Chiang JI, Jani BD, Mair FS et al. Associations between multimorbidity, all-cause mortality and glycaemia in people with type 2 diabetes: A systematic review. PLoS One, 2018; 13: e0209585.[DOI]
[40] Tee C, Xu H, Fu X et al. Longitudinal HbA1c trajectory modelling reveals the association of HbA1c and risk of hospitalization for heart failure for patients with type 2 diabetes mellitus. PLoS One, 2023; 18: e0275610.[DOI]





 Copyright ©
Copyright ©