mRNA Technology in Modern Medicine: Review and Future Prospects
Heslley Silva1,2*
1State University of Minas Gerais, Belo Horizonte, Minas Gerais, Brazil
2University Center of Formiga, Formiga, Minas Gerais, Brazil
*Correspondence to: Heslley Silva, PhD, Professor, State University of Minas Gerais, Av. São Paulo Rod MG 040 URB), 3996 - Vila Rosário, Ibirité - MG, 32412-190, Brazil; E-mail: heslley@uniformg.edu.br
DOI: 10.53964/cme.2025001
Abstract
Messenger RNA (mRNA) technology has revolutionized modern medicine, particularly in developing vaccines and gene therapies. While its prominence soared during the COVID-19 pandemic, its foundation was built on decades of meticulous research. This review explores the historical evolution of mRNA technology, its stabilization and delivery breakthroughs, and its applications in combating infectious diseases, cancer, and genetic disorders. The study utilized a systematic search of peer-reviewed articles from leading databases such as PubMed and Scopus, focusing on advancements and clinical applications. Future potential in treating chronic diseases, enhancing immunotherapy, and addressing public health emergencies is also discussed, emphasizing the need for sustained research and innovation to harness its transformative capabilities fully.
Keywords: mRNA, vaccines, COVID-19, chronic diseases, cancer, infectious diseases, public health
1 INTRODUCTION
Messenger RNA (mRNA) technology has emerged as an innovative tool for developing vaccines and gene therapies. Although its practical application has gained visibility recently, the principles of this technology have been established after decades of intensive research. Initially, technical difficulties related to the instability of mRNA and its rapid degradation in the body hampered its progress. However, over time, significant advances have been made, allowing mRNA to become a viable and effective platform for creating vaccines and other therapies.
The effectiveness of mRNA vaccines during the COVID-19 pandemic has demonstrated their potential not only in terms of speed of development but also efficacy and safety. The vaccines developed by Pfizer-BioNTech and Moderna, both based on mRNA technology, were the first to be authorized and widely distributed, showing efficacy of over 90% in preventing symptomatic SARS-CoV-2 infections[1-3]. This unprecedented success has accelerated the global recognition of mRNA as a versatile and powerful platform for immunization.
mRNA technology offers unparalleled flexibility, allowing rapid adaptations in response to new viral variants and different pathogens. This feature was crucial during the recent pandemic when new variants of the virus emerged[4], and vaccines had to be quickly adjusted to maintain their effectiveness[5-7]. The speed with which these modifications can be made is a significant advantage over traditional vaccines, which often require years of development and production.
Initial resistance to the use of mRNA vaccines, often based on myths and misinformation[8], especially through the internet and social networks[9,10], has gradually been overtaken by the accumulated scientific evidence. Studies have shown that mRNA vaccines do not alter human DNA and are safe, with generally mild and temporary side effects[11-13]. The widespread adoption of these vaccines has saved millions of lives and continues to be a crucial tool in controlling the COVID-19 pandemic in many countries[14-16].
In addition to its application against COVID-19, mRNA technology is being explored in several other medical areas. Research is underway into the development of vaccines against other infectious diseases, such as influenza, Zika and cytomegalovirus, as well as personalized therapies against cancer and rare genetic diseases[5,17-19]. The potential of mRNA to revolutionize the treatment of a wide range of diseases is vast, and continued investment in research and development is essential to exploit its capabilities fully. This review aims to briefly present the history and development of this technology, as well as its current and future potential uses in the fight against the main forms of disease.
2 HISTORY AND EVOLUTION OF mRNA TECHNOLOGY
2.1 History and Evolution
Using mRNA to induce an immune response is not new[20,21]. A crucial breakthrough occurred in 1989 when it was demonstrated that direct injection of RNA and DNA in vitro could express proteins in mouse tissues, laying the foundations for future applications of mRNA[22-24].
However, significant challenges, such as mRNA instability and adverse immune response, have slowed progress[25]. mRNA is naturally unstable due to rapid degradation by ribonucleases present in the body, as well as potentially inducing unwanted immune responses[26].
The modification of mRNA was one of these advances. The introduction of modified nucleosides, such as pseudouridine, improved the stability of the mRNA and reduced its immunogenicity, making it more suitable for therapeutic use[27]. In parallel with the chemical modifications, developing efficient delivery systems was essential[28,29]. Liposomes and Lipid nanoparticles (LNPs) were developed to protect the mRNA from degradation and facilitate its entry into the target cells[30-33].
The revolution in the use of mRNA as a vaccine was accelerated by the COVID-19 pandemic. The Pfizer-BioNTech and Moderna vaccines were the first to use mRNA to induce immunity against SARS-CoV-2[1,3,34]. These successes were a testimony to the decades of research that resolved the initial technical limitations[22].
The successful application of mRNA in vaccines against COVID-19 has not only demonstrated its efficacy but has also paved the way for new research and development in various areas of medicine[35-37]. Currently, mRNA is being explored to treat various infectious diseases, cancers and rare genetic diseases. mRNA technology offers hope for treating rare genetic diseases[38-40]. Research indicates that mRNA could be a powerful tool for therapeutic cancer vaccines, making it possible to produce personalized vaccines that train the immune system to attack specific cancer cells[41].
The pioneering work of Katalin Karikó and Drew Weissman on the modification of mRNA not only transformed modern biotechnology but also received worldwide recognition with the award of the Nobel Prize for Medicine[42]. His research has addressed and overcome critical challenges of mRNA stability and immunogenicity, using modified nucleosides such as pseudouridine to increase mRNA stability and reduce adverse immune responses[43]. This breakthrough has been essential in enabling mRNA-based vaccines and gene therapies, which have demonstrated unprecedented efficacy, particularly during the COVID-19 pandemic with the vaccines from Pfizer-BioNTech and Moderna[44,45].
2.2 Using mRNA Technology
2.2.1 Vaccines Against COVID-19
In the first year of the COVID-19 pandemic, the scientific community and the general population faced a scenario of great difficulty and uncertainty[46]. The rapid spread of the virus, combined with the initial lack of knowledge about its characteristics and modes of transmission, has generated an atmosphere of fear and misinformation[47]. Scientists were under intense pressure to develop effective treatments and vaccines in a short space of time, while health systems around the world were overwhelmed by the number of critically ill patients in need of intensive care[48,49]. International collaboration was crucial, but it also revealed the disparities between countries in terms of resources and response capacities[50,51].
The lack of effective treatment at the start of the pandemic has made the situation even worse[52]. The social isolation measures implemented to contain the spread of the virus have proved extremely challenging to maintain. Many countries faced resistance from the population, economic difficulties and logistical problems[53]. Social distancing, while necessary, has had a significant impact on people’s mental health and well-being, as well as exacerbating pre-existing social and economic inequalities[54]. Mitigation strategies, such as wearing masks and hand hygiene, were insufficient to completely contain transmission, highlighting the urgent need for a vaccine[55].
The first tests of the mRNA vaccine against COVID-19 were conducted with remarkable speed and scientific precision. The first vaccine to be tested was from Pfizer-BioNTech (BNT162b2), whose development began shortly after the SARS-CoV-2 genetic sequence became available in January 2020. Clinical trials began in March 2020, covering several phases that evaluated the safety, immunogenicity and efficacy of the vaccine. Phase I of these trials was conducted in the United States and Germany, involving a small number of healthy volunteers to verify the safety and initial immune response of the vaccine[1,37].
As the preliminary results proved promising, phases II and III were quickly implemented in several countries, including the United States, Germany, Turkey, South Africa, Brazil and Argentina. These phases involved significantly more participants and aimed to determine the vaccine’s effectiveness in preventing COVID-19 infection. In November 2020, data from the phase III studies indicated that the vaccine was approximately 95% effective in preventing COVID-19, leading to emergency use authorization by the FDA in the United States in December 2020[56,57].
The first countries to apply the vaccine were the United States and the United Kingdom, followed by several other European countries and worldwide[58,59]. The speed of distribution and administration of the vaccine was facilitated by international cooperation and the existing health infrastructure, allowing for broad vaccination coverage in a short time[60].
The speed with which these vaccines have been developed, tested and distributed is a testament to the flexibility and potential of mRNA technology. Traditionally, vaccine development can take years or even decades[61,62]. However, with the mRNA platform, the genetic code of the SARS-CoV-2 virus was sequenced and incorporated into a functional vaccine in a matter of weeks[63]. This agility is due to the nature of mRNA technology, which allows rapid adaptation to new pathogens simply by changing the mRNA sequence[64,65]. Unfortunately, this virtue of mRNA vaccines is commonly used to falsely defame them on the internet and networks, where many argue that their speed is a sign of their unreliability compared to other immunizers[66,67].
2.2.2 Mechanism of Action
mRNA vaccines work by introducing into the body a mRNA sequence that codes for a specific protein of the pathogen. In the case of COVID-19 vaccines, the mRNA sequence encodes the spike (S) protein of SARS-CoV-2, the virus that causes the disease. When the mRNA is administered, it is encapsulated in LNPs to protect the fragile molecule and facilitate its entry into the host’s cells[68-70].
Once inside the cells, the mRNA is translated into the cytoplasm by the host cell’s ribosomal machinery, producing the spike protein[71]. This protein is then processed and presented on the cell surface, where it is recognized by the immune system as a foreign molecule[17]. The presentation of the spike protein stimulates the adaptive immune response, inducing the production of specific neutralizing antibodies against SARS-CoV-2 and activating helper and cytotoxic T cells[72]. These defense mechanisms are essential to confer immunity to the individual, preparing the body to fight the real virus in future exposures[5,13].
The effectiveness of mRNA vaccines lies in their ability to induce a robust and memory immune response[73]. The production of neutralizing antibodies prevents the virus from entering cells, while T cells help to destroy infected cells. This dual mechanism of action is crucial for protection against viral infections, providing a comprehensive and long-lasting defense against the pathogen[5,68].
Although the efficacy of mRNA vaccines is widely acknowledged, there remain aspects that require further understanding, such as the duration of the immune response and the detailed mechanisms of T-cell activation. Future research is essential to elucidate how mRNA technology can be optimized to provide even more robust and long-lasting protection, particularly against emerging viral variants. Additionally, it is crucial to investigate the potential long-term effects of these vaccines and to develop strategies to mitigate any possible adverse reactions.
This diagram illustrates the step-by-step process of mRNA vaccine development, starting from the discovery phase in the laboratory to its applications in real-world epidemiology. Key phases include RNA sequencing, computational modeling of mRNA, large-scale manufacturing, and the deployment of vaccines in public health interventions. It highlights the role of mRNA vaccines in combating infectious diseases and shaping modern epidemiological practices.
This diagram was generated using advanced AI tools designed to create professional and academic-quality visuals. The AI, powered by OpenAI’s DALL·E model, synthesizes text-based inputs into detailed and accurate graphical representations.
2.2.3 Simplified Steps in the Mechanism of Operation of mRNA Vaccines (Figure 1)
(1) Vaccine administration: The mRNA vaccine is injected into the muscle. The mRNA is encapsulated in LNPs for protection and transportation.
(2) Cell entry: The LNPs facilitate the entry of the mRNA into the muscle cells. The mRNA is released into the cell’s cytoplasm.
(3) mRNA translation: In the ribosomes, the mRNA is translated into SARS-CoV-2 spike proteins. Spike proteins are produced by the normal cellular machinery.
(4) Spike Protein Presentation: The spike proteins are processed and displayed on the surface of the cells. The cells present the spike protein to T and B lymphocytes.
(5) Adaptive Immune Response: B lymphocytes produce specific neutralizing antibodies against the spike protein. T lymphocytes are activated and help destroy infected cells.
(6) Immune Memory: The production of T and B memory cells is induced. The body is prepared to respond quickly to future SARS-CoV-2 infections.
|
Figure 1. Schematic Diagram of mRNA Vaccine Development Process and Epidemiological Applications.
2.2.4 Safety of mRNA Technology for Vaccines
Despite unfounded criticism of the safety of mRNA vaccines, extensive studies have shown them to be safe and effective. Modification of mRNA nucleosides reduces innate immunogenicity and increases stability, minimizing adverse reactions[17,74,75]. It should be borne in mind that the absence of live or attenuated viral components in mRNA vaccines eliminates the risk of infection from the vaccine itself.
The safety of mRNA vaccines has been rigorously evaluated in several clinical trials (Figure 1). Studies have shown that mRNA vaccines against COVID-19 are well tolerated, with generally mild to moderate side effects such as pain at the injection site, fatigue and headache[1,3,76]. These adverse effects are transient and resolved within a few days. In addition, continuous surveillance after the vaccines have been distributed to the public reinforces the safety of these vaccines, with serious adverse events being extremely rare and closely monitored[77,78].
Another important aspect of the safety of mRNA vaccines is the absence of any risk of mRNA integration into the human genome. The mRNA used in vaccines does not enter the cell nucleus and, therefore, does not interact with human DNA[79]. Instead, it is naturally degraded after the translation of the spike protein, eliminating concerns of genetic alterations[68].
Finally, mRNA technology enables a rapid response to new viral variants. The ability to quickly modify the mRNA sequence to match new virus strains demonstrates the flexibility and safety of this technology, enabling quick and effective adjustments without compromising the safety of the final product (Figure 1)[13,80]. This advance not only reinforces the continued effectiveness of vaccines, but also ensures that established safety processes are maintained in new iterations of the vaccine.
2.2.5 Continuous Monitoring and Adaptation
The ability to continuously monitor the efficacy of vaccines and quickly adapt them to new variants is an essential feature of mRNA vaccines. This continuous monitoring, together with the agility to adapt, allows mRNA vaccines to maintain their effectiveness in an evolving pandemic scenario. When a new variant emerges, scientists can sequence its genome and quickly insert the new mRNA sequence that codes for the virus’s altered spike protein into the existing vaccine platform. This allows new versions of the vaccine to be developed and produced in a matter of weeks, as opposed to the months or years needed to update traditional vaccines based on proteins or inactivated viruses[81,82].
A practical example of this capacity is the rapid response to the Delta and Omicron variants of SARS-CoV-2. Studies have shown that mRNA vaccines after being modified to include the mutations present in these variants, have maintained robust efficacy. The administration of booster doses has proved particularly effective, significantly increasing the levels of neutralizing antibodies and providing additional protection against these more transmissible variants[83-85].
Furthermore, the flexibility of mRNA vaccines allows them to be adjusted not only to new variants of the same virus but also to different pathogens. This technology is already being exploited to rapidly create vaccines against other emerging viruses and even for non-viral diseases, demonstrating its potential to transform the global response to future outbreaks[5,86].
Continuous monitoring also involves collecting and analyzing data post-distribution of the vaccines, allowing for the rapid identification of any rare adverse effects and the ongoing assessment of vaccine efficacy. This feedback loop is crucial to ensuring that mRNA vaccines remain safe and effective, adjusting as necessary to meet the ever-changing challenges presented by new variants and different populations[78,87].
2.3 Today’s and Tomorrow’s Challenges
Although mRNA technology has shown immense potential, there are challenges to be faced. The distribution and storage of mRNA vaccines, which require extremely low temperatures, is a significant logistical obstacle[88-90]. However, research is underway to develop mRNA formulations that are more stable at higher temperatures, which could broaden access to these vaccines globally[91,92].
Large-scale production of mRNA vaccines faces challenges related to the availability and cost of the inputs required for mRNA synthesis, including nucleotides, enzymes and LNPs. Global production capacity is limited, and growing demand can lead to shortages of essential supplies[93-95]. Immunogenicity is also a significant challenge. Although the mRNA can be modified to reduce the innate immune response and increase stability, there is still a risk of adverse immune reactions, especially in repeated administrations, which can limit the effectiveness of long-term treatments[96,97].
Another critical challenge is the effective delivery of the mRNA to the target cells. The development of safe and efficient delivery systems that can protect the mRNA from degradation before it reaches the cellular target is essential. LNPs are currently the main technology used, but improvements are still needed to increase specificity and reduce side effects (Figure 2)[32,33].
|
Figure 2. Schematic Diagram of mRNA Applications in Gene Therapy.
The regulation and approval of new vaccines and mRNA therapies can also be a lengthy and complex process. Regulatory agencies require extensive safety and efficacy data, which can delay the availability of new vaccines to the public[94,98]. Finally, public acceptance and hesitancy towards new mRNA technologies pose additional challenges. Effective education and communication about the benefits and risks associated with mRNA vaccines are essential to ensure public confidence[99-102].
3 POTENTIAL USES AND FUTURE PROSPECTS
This diagram showcases the applications of mRNA technology in gene therapy, highlighting key processes such as RNA synthesis, delivery mechanisms using LNPs, cellular translation into therapeutic proteins, and clinical outcomes. It demonstrates how mRNA-based treatments target genetic disorders like cystic fibrosis and muscular dystrophy, offering promising advancements in precision medicine.
The diagram was generated using OpenAI’s DALL·E model, an advanced artificial intelligence tool that transforms descriptive text into detailed and visually accurate diagrams.
3.1 Chronic Diseases
Initial research indicates that mRNA technology can be used to treat chronic diseases. For example, studies are exploring the use of mRNA to induce the expression of therapeutic proteins in cardiovascular and metabolic diseases (Table 1)[103,104]. A promising example is the use of mRNA for cardiac regeneration after myocardial infarctions, where mRNA encoding growth factors is used to promote tissue repair[105,106].
Table 1. This Table Summarizes the Main Chronic Diseases Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues Being Explored to Treat these Diseases
Chronic Illness |
Laboratories/Research Groups |
Treatment Path |
Myocardial Infarction |
Moderna, BioNTech |
Use of mRNA encoding growth factors to promote cardiac tissue regeneration. |
Neurodegenerative diseases (Alzheimer) |
Moderna, Pfizer, BioNTech |
Induction of the expression of proteins that combat the formation of beta-amyloid plaques. |
Cystic Fibrosis |
Moderna, Vertex Pharmaceuticals |
Correction of specific genetic mutations using mRNA to encode functional proteins. |
Asthma |
AstraZeneca, Moderna, Ethris |
Development of inhalable drugs that deliver therapeutic molecules directly to the lungs. |
Chronic Obstructive Pulmonary Disease (COPD) |
AstraZeneca, Moderna, Ethris |
Development of inhalable drugs that deliver therapeutic molecules directly to the lungs. |
Rheumatoid arthritis |
Pfizer, BioNTech |
Coding of immunomodulatory proteins by mRNA to regulate the immune system and reduce inflammation. |
Systemic Lupus Erythematosus |
Pfizer, BioNTech |
Coding of immunomodulatory proteins by mRNA to regulate the immune system and reduce inflammation. |
Hemophilia |
BioMarin Pharmaceutical, Moderna |
Induction of the production of deficient coagulation factors using mRNA. |
Additionally, mRNA therapy is being investigated for the treatment of neurodegenerative diseases, such as Alzheimer’s, by inducing the expression of proteins that combat the formation of beta-amyloid plaques[107,108]. Another application is in cystic fibrosis, where mRNA can be used to correct specific genetic mutations, offering new hope for the treatment of this debilitating condition[109,110]. These approaches offer new perspectives for the treatment of conditions that currently have limited options for effective therapy.
mRNA technology offers promising new possibilities in the treatment of chronic diseases of the respiratory system, such as asthma[111] and COPD[112]. Using mRNA, it is possible to develop inhaled drugs that deliver therapeutic molecules directly to the lungs, ensuring a localized and targeted approach that minimizes systemic side effects and increases treatment efficacy (Figure 2). Furthermore, these drugs allow for continuous production of therapeutic proteins within the lungs, providing sustained therapeutic effects and potentially improving the management of both acute and chronic conditions.
The partnership between companies such as AstraZeneca, Moderna and Ethris has been key to advancing the application of mRNA therapies for respiratory diseases (Table 1)[113,114]. These collaborations focus on the development of stabilized, non-immunogenic mRNAs, which are chemically modified to evade the immune system and deliver the mRNA directly to the alveolar cells. Once inside the cells, the mRNA instructs the production of specific proteins that can replace, inhibit or increase proteins involved in respiratory diseases, addressing disease mechanisms that other drugs cannot treat (Figure 2).
mRNA technology is also being explored for the treatment of autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus. Recent studies indicate that mRNA can be used to encode immunomodulatory proteins that regulate the immune system and reduce the chronic inflammation characteristic of these diseases[115,116]. For example, the administration of mRNA that codes for anti-inflammatory interleukins could help decrease autoimmune activity, offering a new therapeutic approach for patients who do not respond to conventional treatments[117].
Another promising area is the use of mRNA to treat hematological disorders such as hemophilia[118]. mRNA therapy can be used to induce the production of coagulation factors that are deficient in patients with hemophilia A or B[119,120]. This approach has the potential to provide sustained correction of coagulation factor levels, significantly reducing the risk of bleeding and improving patients’ quality of life.
3.2 Rare Diseases
mRNA technology, widely known for its application in COVID-19 vaccines, is showing great potential in the treatment of rare diseases (Table 2). This innovative approach is being explored to treat various genetic and metabolic diseases, offering new hope for conditions that often lack effective therapies[38,40].
A relevant example is the use of mRNA to treat argininosuccinic aciduria, a rare genetic disease that affects protein metabolism and leads to the accumulation of ammonia in the blood[121,122]. Researchers from UCL and King’s College London, in collaboration with the Moderna laboratory, have shown that mRNA therapy can correct glutathione levels and significantly reduce the symptoms of the disease in murine models[123]. This treatment showed that treated mice survived significantly longer compared to untreated mice, highlighting the future potential of this technology in humans[124].
Table 2. This Table Summarizes the Main Rare Diseases that are Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues that are Being Explored for the Treatment of these Diseases
Rare Disease |
Laboratories/Research Groups |
Treatment Path |
Argininosuccinic aciduria |
UCL, King’s College London, Moderna |
Use of mRNA to correct glutathione levels and reduce symptoms. |
Propionic and methylmalonic acidoses |
Moderna |
mRNA therapies to correct organic acid metabolism. |
Fragile X Syndrome |
Catholic University of the Sacred Heart |
Replacement of the deficient protein caused by the mutation in the FMR1 gene. |
Lipoprotein lipase deficiency (LPLD) |
Moderna |
mRNA to increase lipoprotein lipase (LPL) activity, reducing triglycerides. |
Glycogen storage disease type 1B |
Telethon Institute of Genetics and Moderna |
mRNA to produce the enzyme glucose-6-phosphate translocase. |
Crigler-Najjar Syndrome |
Moderna |
mRNA to correct the deficiency of the UGT1A1 enzyme, reducing bilirubin. |
Cystic fibrosis |
Moderna, Vertex Pharmaceuticals |
Provision of genetic instructions to correct the mutation in the CFTR gene. |
Acute hepatic porphyria (AHP) |
Alnylam Pharmaceuticals |
Supplying the enzyme needed for heme synthesis. |
Niemann-Pick disease type C |
UCL, Innovation News Network |
mRNA to provide the functional enzyme needed for lipid metabolism. |
Duchenne muscular dystrophy (DMD) |
Moderna. |
Genetic instructions for dystrophin production. |
The mRNA technology is being tested to treat other rare metabolic diseases, such as propionic and methylmalonic acidoses[125,126]. These conditions affect the metabolism of organic acids and can lead to serious complications from birth. New clinical trials are underway to evaluate the effectiveness of these therapies in humans, with promising preliminary results[127,128].
Another important application of mRNA technology is in the treatment of fragile X syndrome, the most common hereditary cause of intellectual disability[129]. Researchers of the Institute of Genomic Medicine of the Catholic University of the Sacred Heart, in Rome, are exploring the use of mRNA to replace the deficient protein caused by the mutation in the FMR1 gene[130,131]. In vitro studies have shown that mRNA therapy can induce the production of the necessary protein, offering a potential route to reversing the neurological damage associated with the disease[132].
LPL deficiency, a genetic disease that causes extremely high levels of triglycerides in the blood, leading to recurrent pancreatitis and other health problems, is also being tackled with mRNA technology[133]. The Moderna laboratory is developing an mRNA therapy to correct this enzyme deficiency[134]. Pre-clinical studies in animal models have shown that the administration of mRNA can increase the activity of LPL, reducing triglyceride levels and preventing episodes of pancreatitis.
Glycogen storage disease type 1B is one such condition for which a mRNA treatment is being sought[135]. Essa doença genética impede a produção da enzima glicose-6-fosfato translocase, levando ao acúmulo de glicogênio e gordura no fígado e nos rins[136]. Researchers from the Telethon Institute of Genetics and Medicine[137], in collaboration with the Moderna laboratory, are developing an mRNA-based therapy for this condition, with initial studies in animal models showing promising results.
Another promising application is in the treatment of Crigler-Najjar syndrome, a rare liver disease characterized by a deficiency of the enzyme UGT1A1, which results in high levels of bilirubin in the blood[138,139]. Moderna lab is exploring the use of mRNA to correct this enzyme deficiency, with pre-clinical studies suggesting that this therapy can effectively reduce bilirubin levels and improve liver function in animal models[140].
Cystic fibrosis, a genetic disease that causes the production of thick, sticky mucus in the lungs, is also a target for mRNA therapies[109]. The approach aims to provide the genetic instructions needed to correct the mutation in the CFTR gene responsible for the disease[141]. Researchers are evaluating the effectiveness of this therapy in initial clinical studies, and the results so far indicate a significant improvement in the lung function of treated patients[21,142].
Another rare disease that can be treated with mRNA is AHP, a group of genetic disorders that affect the production of heme, a vital component of hemoglobin[143]. AHP can cause severe episodes of abdominal pain, neurological problems and other debilitating symptoms[144]. Alnylam Pharmaceuticals is investigating an mRNA approach to provide the deficient enzyme needed for heme synthesis[145,146]. Initial studies suggest that this therapy can reduce the frequency and severity of attacks in patients with AHP.
Niemann-Pick type C disease, a genetic disorder that impedes the body’s ability to metabolize cholesterol and other lipids within cells, is also being targeted by mRNA research[147]. This disease can lead to a series of neurological and liver problems[148]. Researchers are developing mRNA therapies that could provide the functional enzyme needed for proper lipid metabolism, potentially slowing or reversing the progression of the disease[149].
DMD, a genetic disease that causes progressive muscle degeneration, is another rare condition that could benefit from mRNA technology[150]. DMD is caused by mutations in the gene that codes for dystrophin, a protein essential for muscle integrity[151]. mRNA therapies are being developed to provide genetic instructions for the production of dystrophin, offering hope of treatment for DMD patients[152]. Pre-clinical studies in animal models showed an improvement in muscle function after treatment with mRNA[153].
These examples demonstrate how mRNA technology is expanding its frontiers beyond vaccines, promising to revolutionize the treatment of several rare diseases. With continued research and technological advances, we are likely to see a significant increase in the application of mRNA in personalized therapies for conditions that currently lack effective treatment options. Notably, the viability of these treatments will revolutionize a field marked by the dilemma of resources and ethics involving very high costs and a small number of patients who are reached by the procedures that exist today.
3.3 Cancer
mRNA technology is being widely exploited for the treatment of various types of cancer, offering new therapeutic prospects (Table 3)[154,155]. One notable example is the development of personalized cancer vaccines, which use mRNA to encode neoantigens specific to each patient’s tumor, stimulating a targeted and effective immune response[156]. These personalized vaccines have shown promising results in clinical trials for melanoma[157,158] and lung cancer[159,160].
Table 3. This Table Summarizes the Main Types of Cancer Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues Being Explored for the Treatment of these Cancers
Cancer Type |
Laboratories/Research Groups |
Treatment Path |
Melanoma |
Moderna, BioNTech |
Personalized mRNA vaccines that encode neoantigens specific to the patient’s tumor. |
Lung Cancer |
Moderna, BioNTech |
Personalized mRNA vaccines that encode neoantigens specific to the patient’s tumor. |
Leukemia |
Novartis, Kite Pharma |
Production of CAR-T cells modified to express chimeric antigen receptors. |
Lymphomas |
Novartis, Kite Pharma |
Production of CAR-T cells modified to express chimeric antigen receptors. |
Breast Cancer |
Moderna, Pfizer, BioNTech |
Gene therapy with mRNA to suppress oncogenes or restore the function of suppressor genes. |
Ovarian Cancer |
Moderna, Pfizer, BioNTech |
Gene therapy with mRNA to suppress oncogenes or restore the function of suppressor genes. |
Glioblastoma |
Florida University |
mRNA vaccine extracted from patient’s own tumor cells to create a personalized vaccine. |
Pancreatic Cancer |
Memorial Sloan Kettering Cancer Center |
BNT122 mRNA vaccine (RO7198457) to induce immune response against tumor cells. |
Head and Neck Cancer |
Moderna, BioNTech |
Personalized mRNA vaccines that induce an immune response against abnormal proteins. |
Prostate Cancer |
Moderna, BioNTech |
mRNA vaccines to treat metastatic prostate cancer by targeting specific antigens. |
Gastrointestinal Cancer |
Moderna, BioNTech |
mRNA vaccines to treat gastrointestinal cancers by targeting specific antigens. |
The mRNA technology is also being used to produce CAR-T cells, which are T cells modified to express chimeric antigen receptors targeting cancer cells[161]. This approach has the potential to treat leukemia[162] and lymphomas[163] with improved specificity and effectiveness.
Another significant advance is the application of mRNA to deliver gene therapies that suppress oncogenes or restore the function of tumor suppressor genes[164,165]. Studies are investigating the delivery of mRNA encoding corrected mutant p53 proteins, with the aim of improving the response to treatment in cancers such as breast and ovarian cancer[166-168].
A significant example is glioblastoma, an aggressive type of brain tumor[169]. Researchers have developed an mRNA vaccine that triggered a strong immune response against glioblastomas in initial clinical studies[170]. This vaccine uses mRNA extracted from patient’s own tumor cells to create a personalized vaccine, which has been able to reprogram the immune system to attack tumor cells, showing promising results in both animal models and humans[171,172].
Another important advance is in the treatment of pancreatic cancer[173]. Researchers at Memorial Sloan Kettering Cancer Center have developed an mRNA vaccine that has shown potential for treating this particularly deadly cancer[174,175]. The BNT122 vaccine, also known as RO7198457, is being tested in clinical trials and has demonstrated the ability to induce a significant immune response against pancreatic tumor cells, offering new hope for patients with this condition[165].
mRNA technology is also being applied in the development of vaccines against head and neck cancer[176]. Initial studies of personalized mRNA vaccines have shown positive responses in patients with advanced head and neck cancer, with some patients experiencing a complete reduction in tumors[177]. Essas vacinas são projetadas para induzir uma resposta imunológica direcionada contra proteínas anormais produzidas pelas células cancerosas, promovendo a destruição específica dessas células[178].
New perspectives include the treatment of prostate cancers[179] and gastrointestinal[180,181]. In recent clinical studies, mRNA vaccines have been developed to treat metastatic prostate cancers by targeting specific antigens present in cancer cells[182]. These advances are underpinned by the growing understanding of how mRNA can be optimized for greater stability and efficient translation within target cells, as well as the development of new delivery systems that ensure mRNA precisely targets tumors[183]. Collaboration between research institutions and biotechnology companies has been crucial in moving these innovative therapies into clinical development, with the potential to significantly transform cancer treatment (Table 3)[19].
3.4 Viral Diseases
mRNA technology has demonstrated enormous potential for developing vaccines against several viral diseases in addition to COVID-19 (Table 4). For example, research is underway to develop mRNA vaccines against the flu virus[184]. Seasonal flu vaccines have variable effectiveness and need to be reformulated annually; however, mRNA vaccines can be quickly adapted to match each season’s predominant flu strains, offering a faster, more accurate response[185,186].
Table 4. Summary of the Main Types of Viral Diseases Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues Being Explored to Treat these Diseases
Viral Disease |
Laboratories/Research Groups |
Treatment Path |
Flu |
Moderna, Pfizer |
Rapidly adapted mRNA vaccines to match the predominant seasonal strains. |
Rabies |
Moderna |
mRNA vaccines that induce a robust immune response. |
Zika |
Moderna |
mRNA vaccines that offer long-lasting protection against the virus. |
HIV |
Moderna, BioNTech |
mRNA vaccines that can be adjusted to combat different variants of the virus. |
Respiratory Syncytial Virus (RSV) |
Moderna, Pfizer |
mRNA vaccines that induce robust immune responses. |
Cytomegalovirus (CMV) |
Moderna, Pfizer |
mRNA vaccines that induce robust immune responses. |
Yellow Fever |
Pfizer |
mRNA vaccines adapted to produce specific viral proteins. |
Dengue fever |
Pfizer |
mRNA vaccines to create an efficient immune response against the four serotypes. |
Ebola |
Moderna |
mRNA vaccines that induce a strong immune response. |
Chikungunya |
Pfizer |
mRNA vaccines that encode viral proteins to stimulate the immune system. |
Smallpox |
Moderna |
mRNA vaccines to offer protection against related pathogens. |
Hepatitis B |
Moderna, Pfizer |
mRNA vaccines to provide long-term protection against the virus. |
Another promising example is the development of mRNA vaccines against the rabies virus, probably the most lethal of viral diseases[187]. Preliminary studies have shown that these vaccines can induce a robust immune response and protect against infections in animal models[188,189]. mRNA technology is also being explored for the Zika virus, with research indicating that mRNA vaccines may provide long-lasting protection against this virus that causes serious birth defects in newborns[190,191].
mRNA vaccines against the human immunodeficiency virus (HIV) are similarly being investigated. HIV is a particularly difficult challenge due to its high mutation rate, but the flexibility of mRNA technology allows for the creation of vaccines that can be quickly tweaked to combat different variants of the virus[192,193].
mRNA technology is also being applied to other viral diseases, such as RSV and CMV. Recent research demonstrates that mRNA vaccines can induce robust immune responses against RSV, a virus that causes serious respiratory infections in children and old people[194,195]. Similarly, mRNA vaccines against CMV, which can cause serious complications in newborns and immunocompromised people, are in development and show promising results in preclinical studies[196,197].
There are also other relevant examples of viral diseases that have promising treatments being researched, particularly for diseases that currently receive relatively little investment because they are common in poorly developed countries and are typical of countries with fewer resources:
Researchers are exploring mRNA vaccines for yellow fever[198], a mosquito-borne virus that can cause high fever, jaundice and bleeding[199]. mRNA technology can rapidly adapt to produce specific viral proteins that stimulate a robust immune response, offering a new approach to control yellow fever outbreaks[200].
Dengue, another mosquito-borne disease, is a viral infection that can cause fever, intense muscle pain and, in severe cases, bleeding[201]. mRNA vaccines are being developed to create an efficient immune response against the four serotypes of the dengue virus, which could significantly improve the prevention of the disease in endemic areas[202,203].
The Ebola virus, known for causing severe hemorrhagic fevers with high mortality rates, is another target for mRNA vaccines[20]. Initial studies have shown that mRNA vaccines can induce a strong immune response and protect against Ebola infections in animal models, offering hope for the control of future outbreaks[204].
Chikungunya, transmitted by mosquitoes, causes high fever and severe joint pain that can last for months[205]. Researchers are developing mRNA vaccines that encode viral proteins to stimulate the immune system to effectively fight the chikungunya virus, potentially reducing the disease burden in affected areas[206].
With the resurgence of concerns about smallpox and related viruses such as monkeypox, mRNA technology is being investigated to develop vaccines that can offer effective protection against these pathogens[207]. The flexibility of mRNA vaccines allows for a rapid response to possible bioterrorism threats or new natural outbreaks[208].
Although vaccines for hepatitis B already exist, mRNA technology offers a new and potentially more effective approach[209]. Researchers are working on mRNA vaccines that could provide long-lasting protection against the hepatitis B virus, with the aim of reducing the incidence of chronic infections and associated liver cancer[210].
The versatility and speed of the development of mRNA vaccines make this technology a powerful tool in combating various viral diseases, providing new hope for preventing future pandemics and controlling endemic diseases (Table 4).
3.5 Bacterial Diseases
mRNA technology also holds significant potential for combating bacterial diseases, especially in light of growing bacterial resistance to antibiotics (Table 5). The flexibility of the mRNA platform allows rapid adaptation to target specific bacteria and their mutant strains, offering a new strategy in the fight against resistant bacterial infections[211,212].
Table 5. Summary of the Main Types of Bacterial Diseases Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues Being Explored to Treat these Diseases
Bacterial Disease/ Bacteria |
Laboratories/Research Groups |
Treatment Path |
Tuberculosis |
Moderna, Pfizer |
mRNA vaccines that induce a robust immune response against the bacteria. |
C. difficile |
Moderna |
mRNA vaccines that protect against recurrent infections in the gastrointestinal tract. |
P. aeruginosa |
Penn Medicine |
mRNA vaccines that encode specific antigens to induce an immune response. |
K. pneumoniae |
MIT, ScienceDaily |
mRNA vaccines that target specific bacterial proteins to fight infection. |
E. Coli |
Pfizer |
mRNA vaccines that protect against pathogenic strains, complementing hygiene practices. |
Streptococcus pneumoniae |
Penn Medicine, AAMC |
mRNA vaccines that induce immunity against multiple serotypes. |
Neisseria gonorrhoeae |
Pfizer |
mRNA vaccines that induce a robust immune response to prevent infections. |
Staphylococcus aureus |
Moderna, Pfizer |
mRNA vaccines that induce an immune response to prevent infections that are difficult to treat. |
Salmonella |
Pfizer, AAMC |
mRNA vaccines to induce a specific immune response against the bacteria. |
Shigella |
Pfizer, AAMC |
mRNA vaccines to induce a specific immune response against the bacteria. |
A promising example is the development of mRNA vaccines against the bacterium Mycobacterium tuberculosis, which causes tuberculosis[213]. Preliminary studies indicate that mRNA vaccines can induce a robust immune response against tuberculosis, one of the world’s deadliest infectious diseases[214]. Another area of research is the creation of mRNA vaccines against Clostridium difficile (C. difficile), a bacteria that causes serious and often recurring infections in the gastrointestinal tract[215]. Early research has shown that mRNA vaccines can protect against C. difficile infection, offering new hope for treating a disease often resistant to conventional treatments[216].
Pseudomonas aeruginosa (P. aeruginosa) is a gram-negative bacterium that causes serious infections, especially in hospitalized and immunocompromised patients[217]. Due to their intrinsic resistance to many antibiotics, the treatment of these infections is challenging[218]. Researchers are developing mRNA vaccines that encode antigens specific to P. aeruginosa, inducing an immune response that can prevent infections and reduce the need for antibiotics[219,220].
Klebsiella pneumoniae (K. pneumoniae) is a gram-negative bacterium that causes respiratory and urinary infections, often resistant to multiple antibiotics[221]. mRNA technology is being exploited to create vaccines that target specific K. pneumoniae proteins, stimulating the immune system to fight these infections effectively[222]. Early studies show that mRNA vaccines could be a promising solution against this resistant bacterium[223].
Certain strains of Escherichia coli (E. coli) cause serious infections, including urinary tract infections and gastroenteritis[224]. Research is underway to develop mRNA vaccines that can protect against pathogenic strains of E. coli, offering a new preventative approach that can complement hygiene practices and reduce the incidence of these infections[225].
Streptococcus pneumoniae is responsible for infections such as pneumonia, meningitis and otitis media[226]. mRNA vaccines are being developed to induce immunity against multiple serotypes of this bacterium, potentially providing broader and more effective protection than current vaccines[227]. This technology could revolutionize the prevention of these infections, especially in vulnerable populations such as children and the elderly[228].
Neisseria gonorrhoeae, the cause of gonorrhea, has shown increasing resistance to available antibiotics, making the disease a global public health concern[229]. The application of mRNA vaccines against N. gonorrhoeae is being investigated, with the aim of inducing a robust immune response that can prevent infection and reduce the spread of antimicrobial resistance[230].
Likewise, mRNA technology is being explored to develop vaccines against bacteria such as Staphylococcus aureus, including strains resistant to several antibiotics[231]. S. aureus is known to cause a variety of infections, from skin infections to severe pneumonia and sepsis, and antibiotic resistance makes treating these infections a significant challenge[232]. mRNA vaccines can induce an immune response that prevents these infections, offering a preventative strategy against a bacteria that is notoriously difficult to treat[211]. It is always important to realize that bacterial resistance is increasing[233], reducing the effectiveness of known antibiotics, something exacerbated by the misuse of these medicines during the Covid-19 pandemic[234], then we experience the urgency of new perspectives to combat bacterial diseases due to the growing number of resistant strains.
Other bacterial diseases that could benefit from mRNA technology include those caused by another gram-negative bacteria, which have an outer membrane that often impedes the effectiveness of traditional antibiotics[235]. The application of mRNA vaccines may offer a new approach to overcoming these barriers by inducing specific immune responses that help penetrate and eliminate these bacteria[236,237]. In parallel, mRNA technology is being explored to treat infections caused by bacteria such as Salmonella[238] and Shigella[239], which are responsible for serious intestinal infections.
3.6 Protozoal Diseases
mRNA technology also demonstrates significant potential in combating diseases caused by protozoa, such as malaria, leishmaniasis and Chagas disease (Table 6). These diseases represent a considerable challenge to public health, especially in tropical and subtropical regions[240,241].
Table 6. This Table Summarizes the Main Types of Diseases Caused by Protozoa that are Being Researched Using mRNA Technology, the Laboratories and Research Groups Involved, and the Therapeutic Avenues that are Being Explored for the Treatment of these Diseases
Protozoan Disease |
Laboratories/Research Groups |
Treatment Path |
Malaria |
Moderna, Pfizer |
mRNA vaccines that induce a robust immune response against specific Plasmodium antigens. |
Leishmaniasis |
Moderna, Pfizer |
mRNA vaccines encoding specific Leishmania antigens to stimulate immune response. |
Chagas disease |
Moderna, Pfizer |
mRNA vaccines encoding immunogenic proteins from Trypanosoma cruzi. |
Sleeping Sickness |
Pfizer, AAMC |
mRNA vaccines encoding specific T. brucei antigens to prevent infection. |
Amebiasis |
Pfizer, ScienceDaily |
mRNA vaccines encoding immunogenic E. histolytica proteins. |
Babesiosis |
Penn Medicine, AAMC |
mRNA vaccines that target specific Babesia antigens to fight infection mRNA vaccines that target specific Babesia antigens to fight infection. |
Cryptosporidiosis |
MIT, ScienceDaily |
mRNA vaccines that encode Cryptosporidium proteins to prevent infections. |
Animal trypanosomiasis |
Penn Medicine, AAMC |
mRNA vaccines that protect cattle against T. brucei infection. |
Toxoplasmosis |
Moderna, Pfizer |
mRNA vaccines that induce an immune response against Toxoplasma gondii. |
Giardiasis |
Moderna, Pfizer |
mRNA vaccines that induce an immune response against Giardia lamblia. |
Malaria, caused by the protozoan Plasmodium spp, is responsible for millions of cases and hundreds of thousands of deaths annually. Recent research indicates that mRNA vaccines can be developed to induce a robust immune response against specific Plasmodium antigens[242,243]. Promising studies have shown that an mRNA vaccine targeting the CSP (circumsporozoite protein) antigen of Plasmodium falciparum was able to induce a strong immune response in preclinical models[244,245].
Leishmaniasis, caused by protozoa of the genus Leishmania, affects millions of people around the world, causing skin, mucous and visceral infections[246]. Recent developments in mRNA technology suggest that it is possible to create vaccines that encode specific Leishmania antigens to stimulate an effective immune response[247,248]. Studies have demonstrated that mRNA vaccines encoding Leishmania major chimeric proteins induced significant protection against infections in murine models and hold promise for humans[249,250].
Chagas disease, caused by the protozoan Trypanosoma cruzi, continues to be a public health problem in Latin America[251]. mRNA vaccines are being explored as a new strategy to prevent T. cruzi infection[240,252]. Preliminary studies indicate that mRNA vaccines encoding immunogenic T. cruzi proteins can induce a protective immune response in experimental models, suggesting a potential new approach to preventing this debilitating disease[253,254].
Sleeping sickness, caused by the protozoan Trypanosoma brucei, is transmitted by the tsetse fly and can be fatal if left untreated[255]. Researchers are investigating mRNA vaccines that encode specific T. brucei antigens, aiming to stimulate a robust immune response that can prevent infection and reduce transmission[256]. Preclinical studies indicate that these vaccines can induce a protective immune response[257].
Amoebiasis, caused by the protozoan Entamoeba histolytica, is an intestinal infection that can lead to severe diarrhea and liver abscesses[258]. mRNA technology is being exploited to create vaccines that encode immunogenic E. histolytica proteins, offering a new approach to preventing this infection[259]. Initial research shows that these vaccines can induce an effective immune response, reducing the disease burden[260].
Babesiosis, caused by protozoa of the genus Babesia, is transmitted by ticks and can result in severe anemia and serious complications in humans and animals[261]. Researchers are developing mRNA vaccines that target specific Babesia antigens, stimulating the immune system to fight the infection[262]. Pre-clinical studies indicate that these vaccines can be effective in inducing a protective immune response[263].
Cryptosporidiosis, caused by the protozoan Cryptosporidium, is an intestinal infection that can be especially dangerous for people with compromised immune systems[264]. mRNA technology is being applied to develop vaccines that encode Cryptosporidium proteins, with the aim of preventing infection and reducing transmission[265]. Initial research suggests that these vaccines can induce a strong immune response[266].
In addition to these diseases, mRNA technology is also being researched for the treatment of toxoplasmosis, caused by Toxoplasma gondii[267], and giardiasis, caused by Giardia lamblia[268]. Research indicates that mRNA vaccines can induce an effective immune response against these pathogens, offering a new avenue for developing vaccines that are currently not available on the market[269,270].
4 CONCLUSION
mRNA technology represents a transformative breakthrough in modern medicine, offering unprecedented opportunities in disease prevention, treatment, and management. This review has demonstrated the vast potential of mRNA applications across a wide range of medical fields, including infectious diseases, chronic illnesses, cancer, and rare genetic disorders. The remarkable efficacy of mRNA vaccines during the COVID-19 pandemic underscored their adaptability, safety, and rapid scalability, which have catalyzed global research into broader therapeutic applications.
Key advancements, such as nucleoside modifications and lipid nanoparticle delivery systems, have addressed the initial challenges of mRNA instability and immunogenicity, enabling its application in precision medicine (Figure 2). Promising developments in using mRNA for gene therapy, cancer immunotherapy, and combating antimicrobial resistance illustrate its versatile utility in addressing complex medical challenges. Furthermore, mRNA’s ability to rapidly adapt to emerging pathogens highlights its critical role in enhancing global preparedness for future pandemics.
However, challenges remain, including cost, storage logistics, public acceptance, and production availability for diverse applications. Continuous research, interdisciplinary collaboration, and global partnerships are essential to overcoming these barriers and maximizing the societal impact of mRNA technology. By fostering innovation and expanding access, mRNA technology has the potential to revolutionize medical science, providing equitable solutions to pressing health challenges worldwide.
Acknowledgements
The author thanks UNIFORMG and the Research Productivity Scholarship Program (PQ) of the Minas Gerais State University (UEMG) for the scholarships and the opportunity to encourage research.
Conflicts of Interest
The author declared no conflict of interest.
Data Availability
Data sharing is not applicable to this review as no datasets were generated or analyzed during the current study.
Copyright and Permissions
Copyright © 2025 The Author(s). Published by Innovation Forever Publishing Group Limited. This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.
Author Contribution
Silva H took part in all stages of writing the article.
Abbreviation List
AAMC, Association of American Medical Colleges
AHP, Acute hepatic porphyria
COPD, Chronic obstructive pulmonary disease
C. difficile, Clostridium difficile
CMV, Cytomegalovirus
DMD, Duchenne muscular dystrophy
HIV, Human immunodeficiency virus
K. pneumoniae, Klebsiella pneumoniae
LNPs, Lipid nanoparticles
LPL, Lipoprotein lipase
mRNA, Messenger RNA
P. aeruginosa, Pseudomonas aeruginosa
RSV, respiratory syncytial virus
SARS-CoV-2, Severe acute respiratory syndrome coronavirus 2
References
[1] Polack FP, Thomas SJ, Kitchin N et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. New Engl J Med, 2020; 383: 2603-2615.[DOI]
[2] Moreira Jr ED, Kitchin N, Xu X et al. Safety and efficacy of a third dose of BNT162b2 Covid-19 vaccine. New Engl J Med, 2022; 386: 1910-1921.[DOI]
[3] Baden LR, El Sahly HM, Essink B et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. New Engl J Med, 2021; 384: 403-416.[DOI]
[4] Silva HM. Sars-Cov-2, evolution and P1 mutation. Acad Lett, 2021; 2021: 1-4.[DOI]
[5] Sahin U, Muik A, Derhovanessian E et al. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature, 2020; 586: 594-599.[DOI]
[6] Carazo S, Skowronski DM, Brisson M et al. Estimated protection of prior SARS-CoV-2 infection against reinfection with the Omicron variant among messenger RNA-vaccinated and nonvaccinated individuals in Quebec, Canada. JAMA Netw open, 2022; 5: e2236670-e2236670.[DOI]
[7] Cheng CW, Wu CY, Wang SW et al. Low-sugar universal mRNA vaccine against coronavirus variants with deletion of glycosites in the S2 or stem of SARS-CoV-2 spike messenger RNA (mRNA). PNAS, 2023; 120: e2314392120.[DOI]
[8] Silva HM. Medicines and Illusions in the fight against COVID-19 in Brazil. Ethics Med Publ Health, 2021; 16: 100622.[DOI]
[9] Islam MS, Sarkar T, Khan SH et al. COVID-19-Related Infodemic and Its Impact on Public Health: A Global Social Media Analysis. Am J Trop Med Hyg, 2020; 103: 1621.[DOI]
[10] Orso D, Federici N, Copetti R et al. Infodemic and the spread of fake news in the COVID-19-era. Eur J Emerg Med, 2020; 27: 327-328.[DOI]
[11] Choe YJ, Yi S, Hwang I et al. Safety and effectiveness of BNT162b2 mRNA Covid-19 vaccine in adolescents. Vaccine, 2022; 40: 691-694.[DOI]
[12] Chen G, Li X, Sun M et al. COVID-19 mRNA vaccines are generally safe in the short term: a vaccine vigilance real-world study says. Front Immunol, 2021; 12: 669010.[DOI]
[13] Krammer F. SARS-CoV-2 vaccines in development. Nature, 2020; 586: 516-527.[DOI]
[14] Dagan N, Barda N, Kepten E et al. BNT162b2 mRNA Covid-19 vaccine in a nationwide mass vaccination setting. New Engl J Med, 2021; 384: 1412-1423.[DOI]
[15] Machado BAS, Hodel KVS, Fonseca LM dos S et al. The importance of RNA-based vaccines in the fight against COVID-19: an overview. Vaccines, 2021; 9: 1345.[DOI]
[16] Hogan MJ, Pardi N. mRNA vaccines in the COVID-19 pandemic and beyond. Annu Rev Med, 2022; 73: 17-39.[DOI]
[17] Pardi N, Hogan MJ, Porter FW et al. mRNA vaccines-a new era in vaccinology. Nat Rev Drug Discov, 2018; 17: 261-279.[DOI]
[18] Shinde K, Bendre S, Kale N et al. The mRNA vaccine heralds a new era in vaccinology. Asian J Pharm Technol, 2022; 12: 257-265.[DOI]
[19] Deng Z, Tian Y, Song J et al. mRNA vaccines: The dawn of a new era of cancer immunotherapy. Front Immunol, 2022; 13: 887125.[DOI]
[20] Jain S, Venkataraman A, Wechsler ME et al. Messenger RNA-based vaccines: Past, present, and future directions in the context of the COVID-19 pandemic. Adv Drug Deliv Rev, 2021; 179: 114000.[DOI]
[21] Li D, Liu C, Li Y et al. Messenger RNA-Based Therapeutics and Vaccines: What’s beyond COVID-19? ACS Pharmacol Transl Sci, 2023; 6: 943-969.[DOI]
[22] Verbeke R, Lentacker I, De Smedt SC et al. Three decades of messenger RNA vaccine development. Nano Today, 2019; 28: 100766.[DOI]
[23] Wolff JA, Malone RW, Williams P et al. Direct gene transfer into mouse muscle in vivo. Science, 1990; 247: 1465-1468.[DOI]
[24] Novo FJ, Gorecki DC, Goldspink G et al. Gene transfer and expression of humanα-galactosidase from mouse muscle in vitro andin vivo. Gene Ther, 1997; 4: 488-492.[DOI]
[25] Bitounis D, Jacquinet E, Rogers MA et al. Strategies to reduce the risks of mRNA drug and vaccine toxicity. Nat Rev Drug Discov, 2024; 23: 281-300.[DOI]
[26] Karikó K, Buckstein M, Ni H et al. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity, 2005; 23: 165-175.[DOI]
[27] Karikó K, Muramatsu H, Welsh FA et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther, 2008; 16: 1833-1840.[DOI]
[28] Weissman D. mRNA transcript therapy. Expert Rev Vaccines, 2015; 14: 265-281.[DOI]
[29] Barrie ES, Smith RM, Sanford JC et al. mRNA transcript diversity creates new opportunities for pharmacological intervention. Mol Pharmacol, 2012; 81: 620-630.[DOI]
[30] Li B, Zhang X, Dong Y. Nanoscale platforms for messenger RNA delivery. Wires Nanomed Nanobiotechnol, 2019; 11: e1530.[DOI]
[31] Wu Z, Li T. Nanoparticle-mediated cytoplasmic delivery of messenger RNA vaccines: challenges and future perspectives. Pharm Res, 2021; 38: 473-478.[DOI]
[32] Hou X, Zaks T, Langer R et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater, 2021; 6: 1078-1094.[DOI]
[33] Tanaka H, Sakurai Y, Akita H. Lipid nanoparticles for mRNA delivery. Drug Deliv Syst, 2022; 37: 237-246.[DOI]
[34] Shrestha NK, Burke PC, Nowacki AS et al. Effectiveness of the 2023-2024 Formulation of the Coronavirus Disease 2019 Messenger RNA Vaccine. Clin Infect Dis, 2024; ciae132.
[35] Fang E, Liu X, Li M et al. Advances in COVID-19 mRNA vaccine development. Signal Transduct Tar, 2022; 7: 94.[DOI]
[36] Szabó GT, Mahiny AJ, Vlatkovic I. COVID-19 mRNA vaccines: Platforms and current developments. Mol Ther, 2022; 30: 1850-1868.[DOI]
[37] Abbasi J. COVID-19 and mRNA vaccines-first large test for a new approach. Jama, 2020; 324: 1125-1127.[DOI]
[38] Shen G, Liu J, Yang H et al. mRNA therapies: Pioneering a new era in rare genetic disease treatment. J Control Release, 2024; 369: 696-721.[DOI]
[39] Córdoba KM, Jericó D, Sampedro A et al. Messenger RNA as a personalized therapy: The moment of truth for rare metabolic diseases. Int Rev Cel Mol Biol, 2022; 372: 55-96.[DOI]
[40] Berraondo P, Martini PGV, Avila MA et al. Messenger RNA therapy for rare genetic metabolic diseases. Gut, 2019; 68: 1323-1330.[DOI]
[41] Sahin U, Derhovanessian E, Miller M et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature, 2017; 547: 222-226.[DOI]
[42] Karikó K, Weissman D. 2023 Nobel Prize in Physiology or Medicine. Curr Sci, 2023; 125: 1168.
[43] Krammer F, Palese P. Profile of Katalin Karikó and Drew Weissman: 2023 Nobel laureates in Physiology or Medicine. PNAS, 2024; 121: e2400423121.[DOI]
[44] Shahcheraghi SH, Ayatollahi J, Aljabali AAA et al. An overview of vaccine development for COVID-19. Ther Deliv, 2021; 12: 235-244.[DOI]
[45] Thorn CR, Sharma D, Combs R et al. The journey of a lifetime-development of Pfizer’s COVID-19 vaccine. Curr Opin Biotechnol, 2022; 78: 102803.[DOI]
[46] Bryce C, Ring P, Ashby S et al. Resilience in the face of uncertainty: early lessons from the COVID-19 pandemic. In: COVID-19. Routledge: London, UK, 2022.
[47] Leão T, Amorim M, Fraga S et al. What doubts, concerns and fears about COVID-19 emerged during the first wave of the pandemic? Patient Educ Couns, 2021; 104: 235-241.[DOI]
[48] Mareiniss DP. The impending storm: COVID-19, pandemics and our overwhelmed emergency departments. Am J Emerg Med, 2020; 38: 1293.[DOI]
[49] Haldane V, De Foo C, Abdalla SM et al. Health systems resilience in managing the COVID-19 pandemic: lessons from 28 countries. Nat Med, 2021; 27: 964-980.[DOI]
[50] Fry CV, Cai X, Zhang Y et al. Consolidation in a crisis: Patterns of international collaboration in early COVID-19 research. PLoS One, 2020; 15: e0236307.[DOI]
[51] Kim K, Cho KT. A review of global collaboration on COVID-19 research during the pandemic in 2020. Sustainability, 2021; 13: 7618.[DOI]
[52] Vijayvargiya P, Garrigos ZE, Almeida NEC et al. Treatment considerations for COVID-19: A critical review of the evidence (or lack thereof). In: Mayo Clinic Proceedings. Elsevier: Amsterdam, Netherlands, 2020.[DOI]
[53] Bong CL, Brasher C, Chikumba E et al. The COVID-19 pandemic: effects on low-and middle-income countries. Anesth Analg, 2020; 131: 86-92.[DOI]
[54] Heitzman J. Impact of COVID-19 pandemic on mental health. Psychiatr Pol, 2020; 54: 187-198.[DOI]
[55] Singh JA, Upshur REG. The granting of emergency use designation to COVID-19 candidate vaccines: implications for COVID-19 vaccine trials. Lancet Infect Dis, 2021; 21: e103-e109.[DOI]
[56] Vasireddy D, Atluri P, Malayala SV et al. Review of COVID-19 vaccines approved in the United States of America for emergency use. J Clin Med Res, 2021; 13: 204.[DOI]
[57] Kesselheim AS, Darrow JJ, Kulldorff M et al. An Overview Of Vaccine Development, Approval, And Regulation, With Implications For COVID-19: Analysis reviews the Food and Drug Administration’s critical vaccine approval role with implications for COVID-19 vaccines. Health Affair, 2021; 40: 25-32.[DOI]
[58] Prüβ BM. Current state of the first COVID-19 vaccines. Vaccines, 2021; 9: 30.[DOI]
[59] Mathieu E, Ritchie H, Ortiz-Ospina E et al. A global database of COVID-19 vaccinations. Nat Hum Behav, 2021; 5: 947-953.[DOI]
[60] Kashte S, Gulbake A, El-Amin III SF et al. COVID-19 vaccines: rapid development, implications, challenges and future prospects. Hum Cell, 2021; 34: 711-733.[DOI]
[61] Plotkin SA. History of vaccine development. Springer Science & Business Media: New York, USA, 2011.
[62] Saleh A, Qamar S, Tekin A et al. Vaccine development throughout history. Cureus, 2021; 13: e16635.[DOI]
[63] Wang H, Li X, Li T et al. The genetic sequence, origin, and diagnosis of SARS-CoV-2. Eur J Clin Microbiol, 2020; 39: 1629-1635.[DOI]
[64] Kramps T, Probst J. Messenger RNA-based vaccines: progress, challenges, applications. Wires Rna, 2013; 4: 737-749.[DOI]
[65] Laczkó D, Hogan MJ, Toulmin SA et al. A single immunization with nucleoside-modified mRNA vaccines elicits strong cellular and humoral immune responses against SARS-CoV-2 in mice. Immunity, 2020; 53: 724-732.[DOI]
[66] Galhardi CP, Freire NP, Fagundes MCM et al. Fake News and vaccine hesitancy in the COVID-19 pandemic in Brazil. Cienc Saude Coletiva, 2022; 27: 1849-1858.[DOI]
[67] Sharma K, Zhang Y, Liu Y. COVID-19 vaccine misinformation campaigns and social media narratives. In: Proceedings of the International AAAI Conference on Web and Social Media. Atlanta, USA, 6-9 June, 2022.[DOI]
[68] Jackson NAC, Kester KE, Casimiro D et al. The promise of mRNA vaccines: a biotech and industrial perspective. npj Vaccines, 2020; 5: 11.[DOI]
[69] Alfagih IM, Aldosari B, AlQuadeib B et al. Nanoparticles as adjuvants and nanodelivery systems for mRNA-based vaccines. Pharmaceutics, 2020; 13: 45.[DOI]
[70] Ramachandran S, Satapathy SR, Dutta T. Delivery strategies for mRNA vaccines. Pharm Med, 2022; 36: 11-20.[DOI]
[71] Zhang J, Xiao T, Cai Y et al. Structure of SARS-CoV-2 spike protein. Curr Opin Virol, 2021; 50: 173-182.[DOI]
[72] Yang Y, Du L. SARS-CoV-2 spike protein: a key target for eliciting persistent neutralizing antibodies. Signal Transduct Tar, 2021; 6: 95.[DOI]
[73] Baum A, Fulton BO, Wloga E et al. Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies. Science, 2020; 369: 1014-1018.[DOI]
[74] Kadali RAK, Janagama R, Yedlapati SH et al. Side effects of messenger RNA vaccines and prior history of COVID-19, a cross-sectional study. Am J Infect Control, 2022; 50: 8-14.[DOI]
[75] Meo SA, Bukhari IA, Akram J et al. COVID-19 vaccines: comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur Rev Med Pharmacol Sci, 2021; 25: 1663-1669.
[76] SeyedAlinaghi S, Karimi A, Pashaei Z et al. Safety and adverse events related to COVID-19 mRNA vaccines; a systematic review. Arch Acad Emerg Med, 2022; 10: e41.
[77] Covid C, Team R, Administration F and D. Allergic reactions including anaphylaxis after receipt of the first dose of Moderna COVID-19 vaccine-United States, December 21, 2020-January 10, 2021. MMWR Morb Mortal Wkly Rep, 2021; 70: 125.[DOI]
[78] Gee J. First month of COVID-19 vaccine safety monitoring-United States, December 14, 2020-January 13, 2021. MMWR Morb Mortal Wkly Rep, 2021; 70: 283-288.[DOI]
[79] Iavarone C, O’hagan DT, Yu D et al. Mechanism of action of mRNA-based vaccines. Expert Rev Vaccines, 2017; 16: 871-881.[DOI]
[80] Awadasseid A, Wu Y, Tanaka Y et al. Current advances in the development of SARS-CoV-2 vaccines. Int J Biol Sci, 2021; 17: 8.[DOI]
[81] Dolgin E. The tangled history of mRNA vaccines. Nature, 2021; 597: 318-324.[DOI]
[82] Pascolo S. Messenger RNA-based vaccines. Expert Opin Biol Ther, 2004; 4: 1285-1294.[DOI]
[83] Garcia-Beltran WF, Denis KJS, Hoelzemer A et al. mRNA-based COVID-19 vaccine boosters induce neutralizing immunity against SARS-CoV-2 Omicron variant. Cell, 2022; 185: 457-466.[DOI]
[84] Plante JA, Liu Y, Liu J et al. Spike mutation D614G alters SARS-CoV-2 fitness. Nature, 2021; 592: 116-121.[DOI]
[85] Faria NR, Claro IM, Candido D et al. Genomic characterisation of an emergent SARS-CoV-2 lineage in Manaus: preliminary findings. Virological, 2021; 372: 815-821.
[86] Chavda VP, Soni S, Vora LK et al. mRNA-based vaccines and therapeutics for COVID-19 and future pandemics. Vaccines, 2022; 10: 2150.[DOI]
[87] Rzymski P, Szuster-Ciesielska A, Dzieciątkowski T et al. mRNA vaccines: The future of prevention of viral infections? J Med Virol, 2023; 95: e28572.[DOI]
[88] Stitz L, Vogel A, Schnee M et al. A thermostable messenger RNA based vaccine against rabies. Plos Neglect Trop D, 2017; 11: e0006108.[DOI]
[89] Blenke EO, Örnskov E, Schöneich C et al. The storage and in-use stability of mRNA vaccines and therapeutics: not a cold case. J Pharm Sci, 2023; 112: 386-403.[DOI]
[90] Fahrni ML, Ismail IAN, Refi DM et al. Management of COVID-19 vaccines cold chain logistics: a scoping review. J Pharm policy Pract, 2022; 15: 16.[DOI]
[91] Buckland B, Sanyal G, Ranheim T et al. Vaccine process technology-A decade of progress. Biotechnol Bioeng, 2024; 121: 2604-2635.[DOI]
[92] Uddin MN, Roni MA. Challenges of storage and stability of mRNA-based COVID-19 vaccines. Vaccines, 2021; 9: 1033.[DOI]
[93] Wouters OJ, Shadlen KC, Salcher-Konrad M et al. Challenges in ensuring global access to COVID-19 vaccines: production, affordability, allocation, and deployment. Lancet, 2021; 397: 1023-1034.[DOI]
[94] Niazi SK. Making COVID-19 mRNA vaccines accessible: challenges resolved. Expert Rev Vaccines, 2022; 21: 1163-1176.[DOI]
[95] Golan MS, Trump BD, Cegan JC et al. The vaccine supply chain: a call for resilience analytics to support COVID-19 vaccine production and distribution. In: COVID-19: systemic risk and resilience. Springer: New York, USA, 2021.
[96] Li D, Liu Q, Yang M et al. Nanomaterials for mRNA-based therapeutics: Challenges and opportunities. Bioeng Transl Med, 2023; 8: e10492.[DOI]
[97] Weng Y, Li C, Yang T et al. The challenge and prospect of mRNA therapeutics landscape. Biotechnol Adv, 2020; 40: 107534.[DOI]
[98] Knezevic I, Liu MA, Peden K et al. Development of mRNA vaccines: scientific and regulatory issues. Vaccines, 2021; 9: 81.[DOI]
[99] Peretti-Watel P, Verger P, Ward JK. To understand mRNA vaccine hesitancy, stop calling the public anti-science. Nat Med, 2024; 30: 923-924.[DOI]
[100] Silva HM. Risks of scientific misinformation through press and pre-print articles. Irish J Med Sci, 2023; 193: 1681-1682.
[101] Silva HM. Iconic Athletes and the Antivaccine Movement: An Improbable Alliance That Reinforces Science Denial. AJPM Focus, 2023; 2: 100066.[DOI]
[102] Silva HM. Deceptive tactics: Misappropriation of scientific literature by ‘Gazeta do Povo’in undermining COVID-19 vaccination efforts. Hum Vacc Immunother, 2024; 20: 2350113.[DOI]
[103] Kaur K, Zangi L. Modified mRNA as a Therapeutic Tool for the Heart. Cardiovasc Drug Ther, 2020; 34: 871-880.[DOI]
[104] Czech MP, Aouadi M, Tesz GJ. RNAi-based therapeutic strategies for metabolic disease. Nat Rev Endocrinol, 2011; 7: 473-484.[DOI]
[105] Zangi L, Lui KO, Von Gise A et al. Modified mRNA directs the fate of heart progenitor cells and induces vascular regeneration after myocardial infarction. Nat Biotechnol, 2013; 31: 898-907.[DOI]
[106] Hadas Y, Katz MG, Bridges CR et al. Modified mRNA as a therapeutic tool to induce cardiac regeneration in ischemic heart disease. Wires Syst Biol Med, 2017; 9: e1367.[DOI]
[107] Wang L, Zeng L, Jiang H et al. Microarray profile of long noncoding RNA and messenger RNA expression in a model of Alzheimer’s disease. Life, 2020; 10: 64.[DOI]
[108] Amakiri N, Kubosumi A, Tran J et al. Amyloid beta and microRNAs in Alzheimer’s disease. Front Neurosci-Switz, 2019; 13: 445024.[DOI]
[109] Boyd AC, Guo S, Huang L et al. New approaches to genetic therapies for cystic fibrosis. J Cyst Fibros, 2020; 19: S54-S59.[DOI]
[110] Flotte TR, Laube BL. Gene therapy in cystic fibrosis. Chest, 2001; 120: 124S-131S.[DOI]
[111] Sel S, Henke W, Dietrich A et al. Treatment of allergic asthma by targeting transcription factors using nucleic-acid based technologies. Curr Pharm Des, 2006; 12: 3293-3304.[DOI]
[112] Mei D, Tan WSD, Tay Y et al. Therapeutic RNA strategies for chronic obstructive pulmonary disease. Trends Pharmacol Sci, 2020; 41: 475-486.[DOI]
[113] Wahlich J, Desai A, Greco F et al. Nanomedicines for the Delivery of Biologics. Pharmaceutics, 2019; 11: 210.[DOI]
[114] Mehta PP, Dhapte-Pawar VS. Novel and evolving therapies for COVID-19 related pulmonary complications. Am J Med Sci, 2021; 361: 557-566.[DOI]
[115] Gorabi AM, Kiaie N, Aslani S et al. Prospects for the potential of RNA interference in the treatment of autoimmune diseases: Small interfering RNAs in the spotlight. J Autoimmun, 2020; 114: 102529.[DOI]
[116] Lipes BD, Keene JD. Autoimmune epitopes in messenger RNA. RNA, 2002; 8: 762-771.[DOI]
[117] Seko Y, Cole S, Kasprzak W et al. The role of cytokine mRNA stability in the pathogenesis of autoimmune disease. Autoimmun Rev, 2006; 5: 299-305.[DOI]
[118] Ragni MV, Chan SY. Innovations in RNA therapy for hemophilia. Blood, 2023; 142: 1613-1621.[DOI]
[119] Chen CY, Tran DM, Cavedon A et al. Treatment of hemophilia A using factor VIII messenger RNA lipid nanoparticles. Mol Ther Acids, 2020; 20: 534-544.[DOI]
[120] Machin N, Ragni MV. An investigational RNAi therapeutic targeting antithrombin for the treatment of hemophilia A and B. J Blood Med, 2018; 9: 135-140.[DOI]
[121] Erez A. Argininosuccinic aciduria: from a monogenic to a complex disorder. Genet Med, 2013; 15: 251-257.[DOI]
[122] Baruteau J, Diez-Fernandez C, Lerner S et al. Argininosuccinic aciduria: recent pathophysiological insights and therapeutic prospects. J Inherit Metab Dis, 2019; 42: 1147-1161.[DOI]
[123] Gurung S, Timmermand OV, Perocheau D et al. mRNA therapy corrects defective glutathione metabolism and restores ureagenesis in preclinical argininosuccinic aciduria. Sci Transl Med, 2024; 16: eadh1334.[DOI]
[124] Gurung S, Timmermand OV, Perocheau D et al. mRNA therapy restores ureagenesis and corrects glutathione metabolism in argininosuccinic aciduria. BioRxiv, 2022; 2022: 512931.[DOI]
[125] Baek R, Coughlan K, Jiang L et al. Characterizing the mechanism of action for mRNA therapeutics for the treatment of propionic acidemia, methylmalonic acidemia, and phenylketonuria. Nat Commun, 2024; 15: 3804.[DOI]
[126] An D, Schneller JL, Frassetto A et al. Systemic messenger RNA therapy as a treatment for methylmalonic acidemia. Cell Rep, 2017; 21: 3548-3558.[DOI]
[127] Martini PGV, Guey LT. A new era for rare genetic diseases: messenger RNA therapy. Hum Gene Ther, 2019; 30: 1180-1189.[DOI]
[128] McNamara JO, Giangrande PH. Toward the full potential of mRNA therapeutics. Mol Ther, 2024; 32: 1600-1601.[DOI]
[129] Hagerman RJ, Berry-Kravis E, Hazlett HC et al. Fragile X syndrome. Nat Rev Dis Primers, 2017; 3: 1-19.[DOI]
[130] Shitik EM, Velmiskina AA, Dolskiy AA et al. Reactivation of FMR1 gene expression is a promising strategy for fragile X syndrome therapy. Gene Ther, 2020; 27: 247-253.[DOI]
[131] Grandi M, Galber C, Gatto C et al. Mitochondrial Dysfunction Causes Cell Death in Patients Affected by Fragile-X-Associated Disorders. Int J Mol Sci, 2024; 25: 3421.[DOI]
[132] Mila M, Alvarez-Mora MI, Madrigal I et al. Fragile X syndrome: An overview and update of the FMR1 gene. Clin Genet, 2018; 93: 197-205.[DOI]
[133] Laurie AD, Kyle CV. A novel frameshift mutation in the lipoprotein lipase gene is rescued by alternative messenger RNA splicing. J Clin Lipidol, 2017; 11: 357-361.[DOI]
[134] Damase TR, Sukhovershin R, Boada C et al. The limitless future of RNA therapeutics. Front Bioeng Biotechnol, 2021; 9: 628137.[DOI]
[135] Cao J, Choi M, Guadagnin E et al. mRNA therapy restores euglycemia and prevents liver tumors in murine model of glycogen storage disease. Nat Commun, 2021; 12: 3090.[DOI]
[136] Chou J, Matern D, Mansfield BC et al. Type I glycogen storage diseases: disorders of the glucose-6-phosphatase complex. Curr Mol Med, 2002; 2: 121-143.[DOI]
[137] Leuzzi R, Fulceri R, Marcolongo P et al. Glucose 6-phosphate transport in fibroblast microsomes from glycogen storage disease type 1b patients: evidence for multiple glucose 6-phosphate transport systems. Biochem J, 2001; 357: 557-562.[DOI]
[138] Greig JA, Chorazeczewski JK, Chowdhary V et al. Lipid nanoparticle-encapsulated mRNA therapy corrects serum total bilirubin level in Crigler-Najjar syndrome mouse model. Mol Ther Meth Clin D, 2023; 29: 32-39.[DOI]
[139] Jansen PLM. Diagnosis and management of Crigler-Najjar syndrome. Eur J Pediatr, 1999; 158: S089-S094.[DOI]
[140] Greig JA, Nordin JML, Draper C et al. Determining the minimally effective dose of a clinical candidate AAV vector in a mouse model of Crigler-Najjar syndrome. Mol Ther Meth Clin D, 2018; 10: 237-244.[DOI]
[141] Robinson E, MacDonald KD, Slaughter K et al. Lipid nanoparticle-delivered chemically modified mRNA restores chloride secretion in cystic fibrosis. Mol Ther, 2018; 26: 2034-2046.[DOI]
[142] Rowe SM, Zuckerman JB, Dorgan D et al. Inhaled mRNA therapy for treatment of cystic fibrosis: Interim results of a randomized, double-blind, placebo-controlled phase 1/2 clinical study. J Cyst Fibros, 2023; 22: 656-664.[DOI]
[143] Wang B, Rudnick S, Cengia B et al. Acute hepatic porphyrias: review and recent progress. Hepatol Commun, 2019; 3: 193-206.[DOI]
[144] Bonkovsky HL, Dixon N, Rudnick S. Pathogenesis and clinical features of the acute hepatic porphyrias (AHPs). Mol Genet Metab, 2019; 128: 213-218.[DOI]
[145] Ricci A, Ventura P. Givosiran for the treatment of acute hepatic porphyria. Expert Rev Clin Pharmacol, 2022; 15: 383-393.[DOI]
[146] Wang B, Ventura P, Takase K et al. Disease burden in patients with acute hepatic porphyria: experience from the phase 3 ENVISION study. Orphanet J Rare Dis, 2022; 17: 327.[DOI]
[147] Furtado D, Cortez-Jugo C, Hung YH et al. mRNA Treatment Rescues Niemann-Pick Disease Type C1 in Patient Fibroblasts. Mol Pharm, 2022; 19: 3987-3999.[DOI]
[148] Vanier MT. Niemann-Pick disease type C. Orphanet J Rare Dis, 2010; 5: 1-18.[DOI]
[149] Paron F, Dardis A, Buratti E. Pre-mRNA splicing defects and RNA binding protein involvement in Niemann Pick type C disease. J Biotechnol, 2020; 318: 20-30.[DOI]
[150] Falzarano MS, Scotton C, Passarelli C et al. Duchenne muscular dystrophy: from diagnosis to therapy. Molecules, 2015; 20: 18168-18184.[DOI]
[151] Duan D, Goemans N, Takeda S et al. Duchenne muscular dystrophy. Nat Rev Dis Primers, 2021; 7: 13.[DOI]
[152] Saifullah, Motohashi N, Tsukahara T et al. Development of therapeutic RNA manipulation for muscular dystrophy. Front Genome Edit, 2022; 4: 863651.[DOI]
[153] Guiraud S, Chen H, Burns DT et al. Advances in genetic therapeutic strategies for Duchenne muscular dystrophy. Exp Physiol, 2015; 100: 1458-1467.[DOI]
[154] Eralp Y. Application of mRNA technology in cancer therapeutics. Vaccines, 2022; 10: 1262.[DOI]
[155] Liu C, Shi Q, Huang X et al. mRNA-based cancer therapeutics. Nat Rev Cancer, 2023; 23: 526-543.[DOI]
[156] Alburquerque-González B, López-Abellán MD, Luengo-Gil G et al. Design of personalized neoantigen RNA vaccines against cancer based on next-generation sequencing data. In: Pharmacogenomics in Drug Discovery and Development. Springer: New York, USA, 2022.
[157] Wang Y, Zhang L, Xu Z et al. mRNA vaccine with antigen-specific checkpoint blockade induces an enhanced immune response against established melanoma. Mol Ther, 2018; 26: 420-434.[DOI]
[158] Sahin U, Oehm P, Derhovanessian E et al. An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature, 2020; 585: 107-112.[DOI]
[159] Lorentzen CL, Haanen JB, Met Ö et al. Clinical advances and ongoing trials of mRNA vaccines for cancer treatment. Lancet Oncol, 2022; 23: e450-e458.[DOI]
[160] García-Pardo M, Gorria T, Malenica I et al. Vaccine therapy in non-small cell lung cancer. Vaccines, 2022; 10: 740.[DOI]
[161] Di Trani CA, Fernandez-Sendin M, Cirella A et al. Advances in mRNA-based drug discovery in cancer immunotherapy. Expert Opin Drug Dis, 2022; 17: 41-53.[DOI]
[162] Barrett DM, Zhao Y, Liu X et al. Treatment of advanced leukemia in mice with mRNA engineered T cells. Hum Gene Ther, 2011; 22: 1575-1586.[DOI]
[163] Xiao K, Lai Y, Yuan W et al. mRNA-based chimeric antigen receptor T cell therapy: Basic principles, recent advances and future directions. Interdiscip Med, 2024; 2: e20230036.[DOI]
[164] Kowalzik F, Schreiner D, Jensen C et al. mRNA-based vaccines. Vaccines, 2021; 9: 390.[DOI]
[165] Sun H, Zhang Y, Wang G et al. mRNA-based therapeutics in cancer treatment. Pharmaceutics, 2023; 15: 622.[DOI]
[166] Ray M, Ruffalo MM, Bar-Joseph Z. Construction of integrated microRNA and mRNA immune cell signatures to predict survival of patients with breast and ovarian cancer. Gene Chromosom Cancer, 2019; 58: 34-42.[DOI]
[167] Met Ö, Balslev E, Flyger H et al. High immunogenic potential of p53 mRNA-transfected dendritic cells in patients with primary breast cancer. Breast Cancer Res Tr, 2011; 125: 395-406.[DOI]
[168] Raab M, Kostova I, Peña-Llopis S et al. Rescue of p53 functions by in vitro-transcribed mRNA impedes the growth of high-grade serous ovarian cancer. Cancer Commun, 2024; 44: 101-126.[DOI]
[169] Wirsching HG, Weller M. Glioblastoma. Malignant Brain Tumors: State-of-the-Art Treat, 2017; 2017: 265-288.
[170] Melnick K, Dastmalchi F, Mitchell D et al. Contemporary RNA therapeutics for glioblastoma. Neuromol Med, 2022; 24: 8-12.[DOI]
[171] Guterres A, Abrahim M, da Costa Neves PC. The role of immune subtyping in glioma mRNA vaccine development. Immunotherapy, 2023; 15: 1057-1072.[DOI]
[172] Lin H, Wang K, Xiong Y et al. Identification of tumor antigens and immune subtypes of glioblastoma for mRNA vaccine development. Front Immunol, 2022; 13: 773264.[DOI]
[173] Uchida S, Kinoh H, Ishii T et al. Systemic delivery of messenger RNA for the treatment of pancreatic cancer using polyplex nanomicelles with a cholesterol moiety. Biomaterials, 2016; 82: 221-228.[DOI]
[174] Rojas LA, Sethna Z, Soares KC et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature, 2023; 618: 144-150.[DOI]
[175] Yu KH, Park J, Mittal A et al. Circulating tumor and invasive cell expression profiling predicts effective therapy in pancreatic cancer. Cancer, 2022; 128: 2958-2966.[DOI]
[176] Liu H, Sun Y, Tian H et al. Characterization of long non-coding RNA and messenger RNA profiles in laryngeal cancer by weighted gene co-expression network analysis. Aging (Albany NY), 2019; 11: 10074.[DOI]
[177] Wang C, Dickie J, Sutavani RV et al. Targeting head and neck cancer by vaccination. Front Immunol, 2018; 9: 830.[DOI]
[178] Chen Y, Jiang N, Chen M et al. Identification of tumor antigens and immune subtypes in head and neck squamous cell carcinoma for mRNA vaccine development. Front Cell Dev Biol, 2022; 10: 1064754.[DOI]
[179] Rastogi I, Muralidhar A, McNeel DG. Vaccines as treatments for prostate cancer. Nat Rev Urol, 2023; 20: 544-559.[DOI]
[180] Zhang A, Ji Q, Sheng X et al. mRNA vaccine in gastrointestinal tumors: Immunomodulatory effects and immunotherapy. Biomed Pharmacother, 2023; 166: 115361.[DOI]
[181] Portillo-Miño JD, Bettin-Gonzalez D, Coral FAM. mRNA vaccines in gastric cancer: How close are we? Vacunas, 2024; 25: 88-96.[DOI]
[182] Rausch S, Schwentner C, Stenzl A et al. mRNA vaccine CV9103 and CV9104 for the treatment of prostate cancer. Hum Vaccin Immunother, 2014; 10: 3146-3152.[DOI]
[183] Qin S, Tang X, Chen Y et al. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Signal Transduct Tar, 2022; 7: 166.[DOI]
[184] Kirby T. mRNA vaccine technology for a multivalent flu vaccine. Lancet Infect Dis, 2023; 23: 157.[DOI]
[185] Deviatkin AA, Simonov RA, Trutneva KA et al. Universal flu mRNA vaccine: promises, prospects, and problems. Vaccines, 2022; 10: 709.[DOI]
[186] Pecetta S, Rappuoli R. mRNA, the beginning of a new influenza vaccine game. PNAS, 2022; 119: e2217533119.[DOI]
[187] Armbruster N, Jasny E, Petsch B. Advances in RNA vaccines for preventive indications: a case study of a vaccine against rabies. Vaccines, 2019; 7: 132.[DOI]
[188] Li J, Liu Q, Liu J et al. An mRNA-based rabies vaccine induces strong protective immune responses in mice and dogs. Virol J, 2022; 19: 184.[DOI]
[189] Alberer M, Gnad-Vogt U, Hong HS et al. Safety and immunogenicity of a mRNA rabies vaccine in healthy adults: an open-label, non-randomised, prospective, first-in-human phase 1 clinical trial. Lancet, 2017; 390: 1511-1520.[DOI]
[190] Richner JM, Himansu S, Dowd KA et al. Modified mRNA vaccines protect against Zika virus infection. Cell, 2017; 168: 1114-1125.[DOI]
[191] Medina-Magües LG, Gergen J, Jasny E et al. mRNA vaccine protects against zika virus. Vaccines, 2021; 9: 1464.[DOI]
[192] Mu Z, Haynes BF, Cain DW. HIV mRNA Vaccines—Progress and Future Paths. Vaccines, 2021; 9: 134.[DOI]
[193] Khalid K, Padda J, Khedr A et al. HIV and messenger RNA (mRNA) vaccine. Cureus, 2021; 13: e16197.[DOI]
[194] Qiu X, Xu S, Lu Y et al. Development of mRNA vaccines against respiratory syncytial virus (RSV). Cytokine Growth F R, 2022; 68: 37-53.[DOI]
[195] Carvalho T. mRNA vaccine effective against RSV respiratory. Nat Med, 2023; 29: 755-756.[DOI]
[196] Fierro C, Brune D, Shaw M et al. Safety and Immunogenicity of a Messenger RNA-Based Cytomegalovirus Vaccine in Healthy Adults: Results From a Phase 1 Randomized Clinical Trial. J Infect Dis, 2024; 230: jiae114.[DOI]
[197] John S, Yuzhakov O, Woods A et al. Multi-antigenic human cytomegalovirus mRNA vaccines that elicit potent humoral and cell-mediated immunity. Vaccine, 2018; 36: 1689-1699.[DOI]
[198] Pascolo S. Synthetic messenger RNA-based vaccines: from scorn to hype. Viruses, 2021; 13: 270.[DOI]
[199] Monath TP, Vasconcelos PFC. Yellow fever. J Clin Virol, 2015; 64: 160-173.[DOI]
[200] Lee J, Woodruff MC, Kim EH et al. Knife’s edge: Balancing immunogenicity and reactogenicity in mRNA vaccines. Exp Mol Med, 2023; 55: 1305-1313.[DOI]
[201] Rigau-Pérez JG, Clark GG, Gubler DJ et al. Dengue and dengue haemorrhagic fever. Lancet, 1998; 352: 971-977.[DOI]
[202] Nasar S, Rashid N, Iftikhar S. Dengue proteins with their role in pathogenesis, and strategies for developing an effective anti‐dengue treatment: A review. J Med Virol, 2020; 92: 941-955.[DOI]
[203] Alameh MG, Weissman D, Pardi N. Messenger RNA-based vaccines against infectious diseases. mRNA Vaccines, 2020; 2020: 111-145.
[204] Bray M, Raymond JL, Geisbert T et al. 3-Deazaneplanocin A induces massively increased interferon-α production in Ebola virus-infected mice. Antivir Res, 2002; 55: 151-159.[DOI]
[205] Caglioti C, Lalle E, Castilletti C et al. Chikungunya virus infection: an overview. New Microbiol, 2013; 36: 211-227.
[206] August A, Attarwala HZ, Himansu S et al. A phase 1 trial of lipid-encapsulated mRNA encoding a monoclonal antibody with neutralizing activity against Chikungunya virus. Nat Med, 2021; 27: 2224-2233.[DOI]
[207] Alkhalil A, Strand S, Mucker E et al. Inhibition of Monkeypox virus replication by RNA interference. Virol J, 2009; 6: 1-10.[DOI]
[208] Morrison R. mRNA Vaccines and their impact on medicine. Microreviews Cell Mol Bio, 2023; 2023: 2.
[209] Hui RWH, Mak LY, Seto WK et al. RNA interference as a novel treatment strategy for chronic hepatitis B infection. Clin Mol Hepatol, 2022; 28: 408.[DOI]
[210] Yang F, Zheng X, Koh S et al. Messenger RNA electroporated hepatitis B virus (HBV) antigen-specific T cell receptor (TCR) redirected T cell therapy is well-tolerated in patients with recurrent HBV-related hepatocellular carcinoma post-liver transplantation: results from a phase I trial. Hepatol Int, 2023; 17: 850-859.[DOI]
[211] Chaudhary N, Weissman D, Whitehead KA. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov, 2021; 20: 817-838.[DOI]
[212] Nitika, Wei J, Hui AM. The development of mRNA vaccines for infectious diseases: recent updates. Infect Drug Resist, 2021; 5271-5285.[DOI]
[213] Shahrear S, Islam ABMMK. Modeling of MT. P495, an mRNA-based vaccine against the phosphate-binding protein PstS1 of Mycobacterium tuberculosis. Mol Divers, 2023; 27: 1613-1632.[DOI]
[214] Vasileva OO, Tereschenko VP, Krapivin BN et al. Immunogenicity of full-length and multi-epitope mRNA vaccines for M. Tuberculosis as demonstrated by the intensity of T-cell response: A comparative study in mice. B Russ State Med U, 2023; 2023: 42-48.[DOI]
[215] Voth DE, Ballard JD. Clostridium difficile toxins: mechanism of action and role in disease. Clin Microbiol Rev, 2005; 18: 247-263.[DOI]
[216] Tan C, Liu T, Chen S et al. Development of multi-epitope mRNA vaccine against Clostridioides difficile using reverse vaccinology and immunoinformatics approaches. Syn Syst Biotechnol, 2024; 9: 667-683.[DOI]
[217] Lyczak JB, Cannon CL, Pier GB. Establishment of Pseudomonas aeruginosa infection: lessons from a versatile opportunist. Microbes Infect, 2000; 2: 1051-1060.[DOI]
[218] Hirsch EB, Tam VH. Impact of multidrug-resistant Pseudomonas aeruginosa infection on patient outcomes. Expert Rev Pharm Out, 2010; 10: 441-451.[DOI]
[219] Wang X, Liu C, Rcheulishvili N et al. Strong immune responses and protection of PcrV and OprF-I mRNA vaccine candidates against Pseudomonas aeruginosa. npj Vaccines, 2023; 8: 76.[DOI]
[220] Ren H, Liu Y, Zhou J et al. Combination of azithromycin and gentamicin for efficient treatment of Pseudomonas aeruginosa infections. J Infect Dis, 2019; 220: 1667-1678.[DOI]
[221] Elemam A, Rahimian J, Mandell W. Infection with panresistant Klebsiella pneumoniae: a report of 2 cases and a brief review of the literature. Clin Infect Dis, 2009; 49: 271-274.[DOI]
[222] Ruhland E, Siemers M, Gerst R et al. The global RNA-RNA interactome of Klebsiella pneumoniae unveils a small RNA regulator of cell division. PNAS, 2024; 121: e2317322121.[DOI]
[223] Zhang X, Shi S, Yao Z et al. Antimicrobial peptide WAM-1: a promising antibacterial and anti-inflammatory drug against carbapenem-resistant Klebsiella pneumoniae. J Antimicrob Chemoth, 2022; 77: 1903-1911.[DOI]
[224] Harrington SM, Dudley EG, Nataro JP. Pathogenesis of enteroaggregative Escherichia coli infection. FEMS Microbiol Lett, 2006; 254: 12-18.[DOI]
[225] Sakai F, Sugita R, Chang JW et al. Transfer-messenger RNA and SmpB mediate bacteriostasis in Escherichia coli cells against tRNA cleavage. Microbiology, 2015; 161: 2019-2028.[DOI]
[226] Mehr S, Wood N. Streptococcus pneumoniae-a review of carriage, infection, serotype replacement and vaccination. Paediatr Respir Rev, 2012; 13: 258-264.[DOI]
[227] Moreira RN, Domingues S, Viegas SC et al. Synergies between RNA degradation and trans-translation in Streptococcus pneumoniae: cross regulation and co-transcription of RNase R and SmpB. BMC Microbiol, 2012; 12: 1-14.[DOI]
[228] Jansson MK, Nguyen DT, Mikkat S et al. Synthetic mRNA delivered to human cells leads to expression of Cpl-1 bacteriophage-endolysin with activity against Streptococcus pneumoniae. Mol Ther-Nucl Acids, 2024; 35: 102145.[DOI]
[229] Unemo M, Shafer WM. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: past, evolution, and future. Clin Microbiol Rev, 2014; 27: 587-613.[DOI]
[230] Williams E, Seib KL, Fairley CK et al. Neisseria gonorrhoeae vaccines: a contemporary overview. Clin Microbiol Rev, 2024; 37: e00094-23.[DOI]
[231] Zhu F, Ma S, Wen H et al. Development of a novel circular mRNA vaccine of six protein combinations against Staphylococcus aureus. J Biomol Struct Dyn, 2023; 41: 10525-10545.[DOI]
[232] Naveed M, Waseem M, Aziz T et al. Identification of bacterial strains and development of anmRNA-based vaccine to combat antibiotic resistance in Staphylococcus aureus via in vitro and in silico approaches. Biomedicines, 2023; 11: 1039.[DOI]
[233] Fair RJ, Tor Y. Antibiotics and bacterial resistance in the 21st century. Perspect Medicin Chem, 2014; 6: PMC-S14459.[DOI]
[234] Silva HM. Antibiotics against viruses: Brazilian doctors adrift. Infect Control Hosp Ep, 2021; 43: 1-5.[DOI]
[235] Naveed M, Mughal MS, Jabeen K et al. Evaluation of the whole proteome to design a novel mRNA-based vaccine against multidrug-resistant Serratia marcescens. Front Microbiol, 2022; 13: 960285.[DOI]
[236] Khalid K, Poh CL. The promising potential of reverse vaccinology-based next-generation vaccine development over conventional vaccines against antibiotic-resistant bacteria. Vaccines, 2023; 11: 1264.[DOI]
[237] Miller SI. Antibiotic resistance and regulation of the gram-negative bacterial outer membrane barrier by host innate immune molecules. mBio, 2016; 7: 10-1128.[DOI]
[238] Imdad MJ, Khan MN, Alam HS et al. Design and in silico analysis of mRNA vaccine construct against Salmonella. J Biomol Struct Dyn, 2023; 41: 7248-7264.[DOI]
[239] Jawalagatti V, Kirthika P, Lee JH. Oral mRNA vaccines against infectious diseases-A bacterial perspective. Front Immunol, 2022; 13: 884862.[DOI]
[240] Versteeg L, Almutairi MM, Hotez PJ et al. Enlisting the mRNA vaccine platform to combat parasitic infections. Vaccines, 2019; 7: 122.[DOI]
[241] You H, Jones MK, Gordon CA et al. The mRNA vaccine technology era and the future control of parasitic infections. Clin Microbiol Rev, 2023; 36: e00241-21.[DOI]
[242] Tsoumani ME, Voyiatzaki C, Efstathiou A. Malaria vaccines: from the past towards the mRNA vaccine era. Vaccines, 2023; 11: 1452.[DOI]
[243] Matarazzo L, Bettencourt PJG. mRNA vaccines: a new opportunity for malaria, tuberculosis and HIV. Front Immunol, 2023; 14: 1172691.[DOI]
[244] Hayashi CTH, Cao Y, Clark LC et al. mRNA-LNP expressing PfCSP and Pfs25 vaccine candidates targeting infection and transmission of Plasmodium falciparum. npj Vaccines, 2022; 7: 155.[DOI]
[245] Borkens Y. Malaria & mRNA Vaccines: A Possible Salvation from One of the Most Relevant Infectious Diseases of the Global South. Acta Parasitol, 2023; 68: 916-928.[DOI]
[246] Kobets T, Grekov I, Lipoldova M. Leishmaniasis: prevention, parasite detection and treatment. Curr Med Chem, 2012; 19: 1443-1474.[DOI]
[247] Duthie MS, Machado BAS, Badaró R et al. Leishmaniasis vaccines: applications of RNA technology and targeted clinical trial designs. Pathogens, 2022; 11: 1259.[DOI]
[248] Alvar J, Croft SL, Kaye P et al. Case study for a vaccine against leishmaniasis. Vaccine, 2013; 31: B244-B249.[DOI]
[249] Vale DL, Freitas CS, Martins VT et al. Efficacy of an Immunotherapy Combining Immunogenic Chimeric Protein Plus Adjuvant and Amphotericin B against Murine Visceral Leishmaniasis. Biology, 2023; 12: 851.[DOI]
[250] Lage DP, Ribeiro PAF, Dias DS et al. Liposomal formulation of ChimeraT, a multiple T-cell epitope-containing recombinant protein, is a candidate vaccine for human visceral leishmaniasis. Vaccines, 2020; 8: 289.[DOI]
[251] Moncayo Á, Silveira AC. Current epidemiological trends of Chagas disease in Latin America and future challenges: Epidemiology, surveillance, and health policies. Am Trypanos Chagas Dis, 2017; 2017: 59-88.[DOI]
[252] Maldonado E, Morales-Pison S, Urbina F et al. Vaccine design against chagas disease focused on the use of nucleic acids. Vaccines, 2022; 10: 587.[DOI]
[253] Poveda C, Leão AC, Mancino C et al. Heterologous mRNA-protein vaccination with Tc24 induces a robust cellular immune response against Trypanosoma cruzi, characterized by an increased level of polyfunctional CD8+ T-cells. Curr Res Immunol, 2023; 4: 100066.[DOI]
[254] Rios LE, Vázquez-Chagoyán JC, Pacheco AO et al. Immunity and vaccine development efforts against Trypanosoma cruzi. Acta Trop, 2019; 200: 105168.[DOI]
[255] Malvy D, Chappuis F. Sleeping sickness. Clin Microbiol Infect, 2011; 17: 986-995.[DOI]
[256] Hulpia F, Mabille D, Campagnaro GD et al. Combining tubercidin and cordycepin scaffolds results in highly active candidates to treat late-stage sleeping sickness. Nat Commun, 2019; 10: 5564.[DOI]
[257] Mulindwa J, Leiss K, Ibberson D et al. Transcriptomes of Trypanosoma brucei rhodesiense from sleeping sickness patients, rodents and culture: Effects of strain, growth conditions and RNA preparation methods. Plos Neglect Trop D, 2018; 12: e0006280.[DOI]
[258] Stauffer W, Ravdin JI. Entamoeba histolytica: an update. Curr Opin Infect Dis, 2003; 16: 479-485.[DOI]
[259] Singh RS, Walia AK, Kanwar JR et al. Amoebiasis vaccine development: A snapshot on E. histolytica with emphasis on perspectives of Gal/GalNAc lectin. Int J Biol Macromol, 2016; 91: 258-268.[DOI]
[260] Huston CD, Petri Jr WA. Vaccines Against Entamoeba histolytica. In: New Generation Vaccines. CRC Press: Boca Raton, USA, 2016.
[261] Vannier E, Krause PJ. 105 - Babesiosis. In: Ryan ET, Hill DR, Solomon T et al. (ed). Hunter's Tropical Medicine and Emerging Infectious Diseases (Tenth Edition). Elsevier, the Netherlands. 2020: 799-802.[DOI]
[262] Santos JHM, Siddle HV, Raza A et al. Exploring the landscape of Babesia bovis vaccines: progress, challenges, and opportunities. Parasite Vector, 2023; 16: 274.[DOI]
[263] Hakimi H, Asada M, Kawazu S. Recent advances in molecular genetic tools for babesia. Vet Sci, 2021; 8: 222.[DOI]
[264] Ramirez NE, Ward LA, Sreevatsan S. A review of the biology and epidemiology of cryptosporidiosis in humans and animals. Microbes Infect, 2004; 6: 773-785.[DOI]
[265] Dayao DA, Jaskiewcz J, Lee S et al. Development of two mouse models for vaccine evaluation against Cryptosporidiosis. Infect Immun, 2022; 90: e00127-22.[DOI]
[266] Castellanos-Gonzalez A, Sadiqova A, Ortega-Mendez J et al. RNA-based therapy for Cryptosporidium parvum infection: proof-of-concept studies. Infect Immun, 2022; 90: e00196-22.[DOI]
[267] Zhang Y, Li S, Chu H et al. A novel mRNA vaccine, TGGT1_278620 mRNA-LNP, prolongs the survival time in BALB/c mice with acute toxoplasmosis. Microbiol Spectr, 2024; 12: e02866-23.[DOI]
[268] Lee P, Abdul-Wahid A, Faubert G. Vaccination Against Giardia. Springer: Vienna, Austria, 2011.
[269] Kolev NG, Tschudi C, Ullu E. RNA interference in protozoan parasites: achievements and challenges. Eukaryot Cell, 2011; 10: 1156-1163.[DOI]
[270] Ospina-Villa JD, López-Camarillo C, Castañón-Sánchez CA et al. Advances on aptamers against protozoan parasites. Genes, 2018; 9: 584.[DOI]

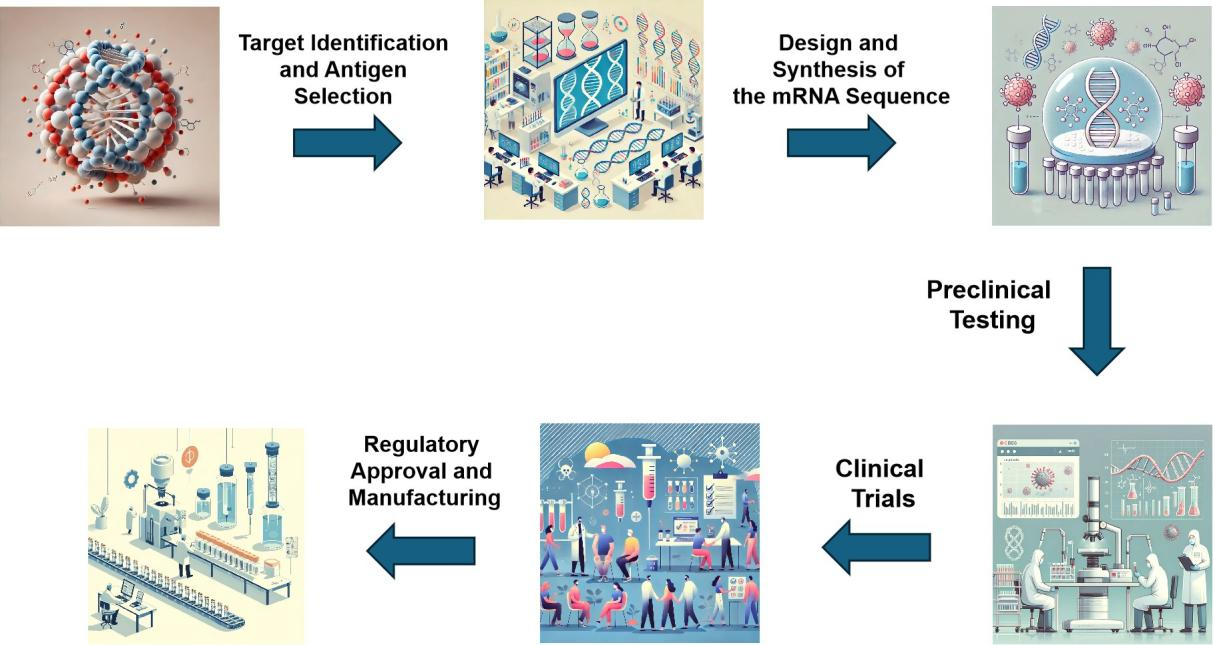




 Copyright ©
Copyright ©