Association Between COVID-19 and Osteoporosis: A Bidirectional Mendelian Randomization Study
Shihang Chen1, Song Xue1,2,3*
1National Clinical Research Center for Metabolic Diseases, Metabolic Syndrome Research Center, Key Laboratory of Diabetes Immunology, Ministry of Education, and Department of Metabolism and Endocrinology, The Second Xiangya Hospital of Central South University, Changsha, Hunan Province, China
2Affiliated Hospital of Jiangxi University of Traditional Chinese Medicine / Jiangxi Provincial Key Research Laboratory of Traditional Chinese Medicine-Key Laboratory of Chronic Renal Failure, Nanchang, Jiangxi Province, China
3College of Resource Processing and Bioengineering, Central South University, Changsha, Hunan Province, China
*Correspondence to: Song Xue, Department of Nephrology, Affiliated Hospital of Jiangxi University of Traditional Chinese Medicine, Nanchang, 330006, Jiangxi Province, China; Email: 2022@csu.edu.cn
DOI: 10.53964/cme.2024008
Abstract
Methods: Analysis of the causal relationship between the two through a two-way Mendelian pathway. Finding disease-related Single nucleotide Polymorphism as working variables through the Genome-wide association study database. The primary analysis for the Mendelian randomization (MR) study was the inverse variance weighting (IVW) method. The pleiotropy was estimated through the intercept from MR-Egger regression, and heterogeneity was assessed through Cochran’s Q test in IVW approach.
Results: In the forward MR analysis, neither exposure to COVID-19 infection, hospitalization or severe infection was significantly associated with osteoporosis or drug-induced osteoporosis outcome (P>0.05); in the reverse MR analysis, exposures to osteoporosis was not significantly associated with COVID-19 outcomes (infection, hospitalization, severe case) (P>0.05).
Keywords: COVID-19, osteoporosis, Mendelian randomization
1 INTRODUCTION
The current COVID-19 pandemic has had enormous health[1] and economic consequences[1] worldwide due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection[2]. Glucocorticoid therapy is widely used to treat patients with COVID-19[3,4] However, glucocorticoid therapy accelerates the loss of calcium from bone and increases the prevalence of osteoporosis[5,6]. On the other hand, SARS-CoV-2 may have direct and indirect effects on osteoclasts and osteoblasts and contribute to osteoporosis[7,8]. Whether there is a causal relationship between osteoporosis and infection with COVID-19 is therefore difficult to predict.
Vitamin D (VitD) is a key hormone for bone health and vitamin D supplementation may reduce the incidence of osteoporosis[9], and studies have shown a significant positive association between vitamin D deficiency and COVID-19 infection and mortality[10], with vitamin D supplementation reducing COVID-19 infection and mortality[11]. Patients with osteoporosis combined with COVID-19 infection also have poorer health outcomes compared to those without osteoporosis[12]. We are therefore interested in whether there is a potential bidirectional relationship between osteoporosis and COVID-19 and the efficacy of vitamin D supplementation improve COVID-19 infection outcomes via the bone density pathway or not.
The Mendelian randomization (MR) framework tests for a potential causal relationship between exposure and outcome by using genetic variation as an instrumental variable[13]. We analyzed the potential causal relationship between three COVID-19 prevalence states (hospitalized cases, severe cases, infected cases) and osteoporosis or drug-induced osteopenia. Bidirectional MR studies were constructed by comparing genome-wide gene extracts of shared genes reported for each trait[14,15].
2 MATERIALS AND METHODS
2.1 Study Design and Assumption
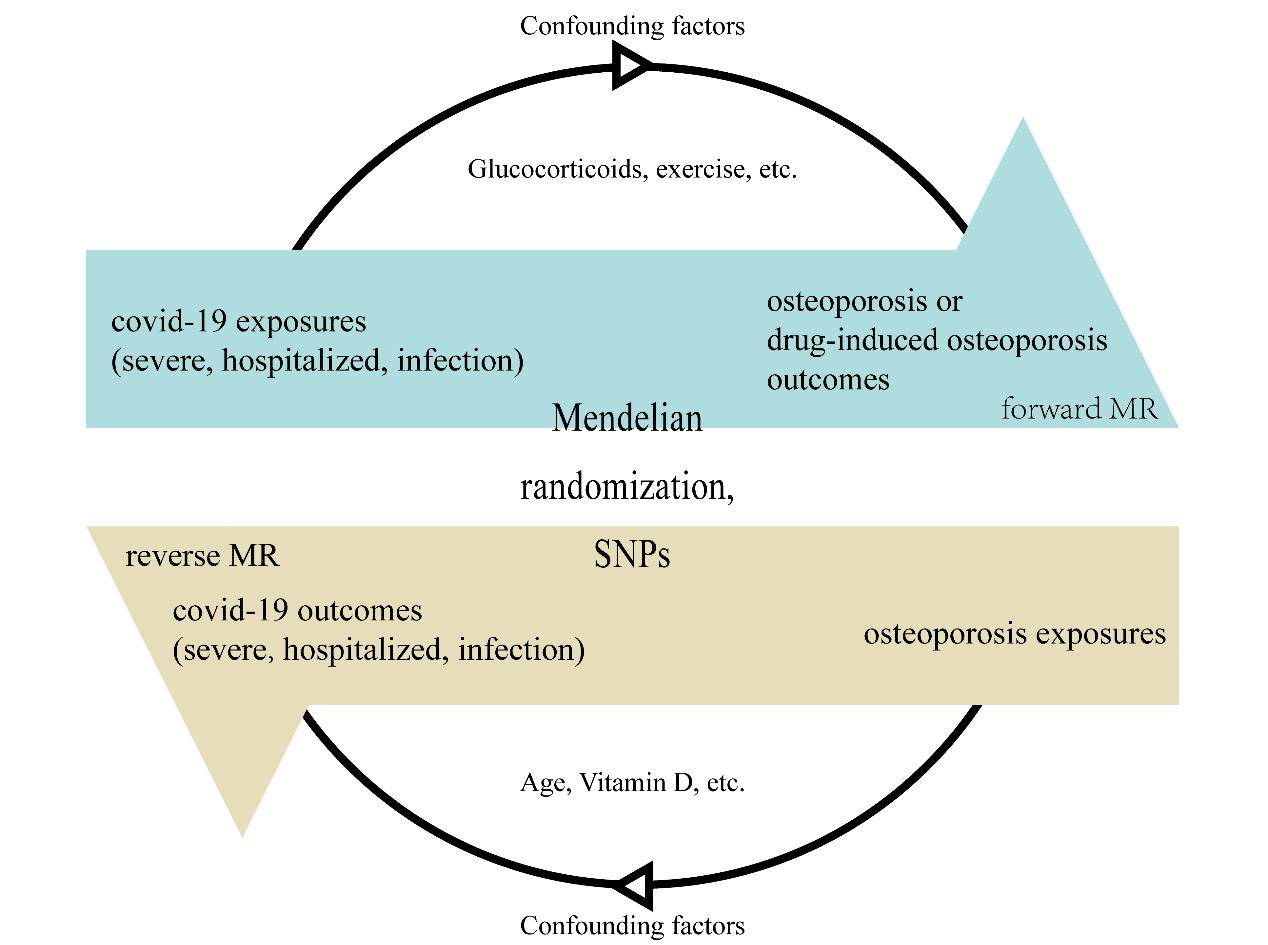
Genome-wide association study (GWAS) data were used to conduct bidirectional MR analyses examining the bidirectional association between COVID-19 and osteoporosis. Forward MR analyses used COVID-19 as exposure and osteoporosis as outcome, whereas reverse MR analyses used osteoporosis as exposure and COVID-19 as outcome. Figure 1 shows a brief description and the core assumption of this bidirectional MR design.
|
Figure 1. A brief description and the core assumption of this bidirectional MR design. Using SNPs as working variables to exclude confounding factors (age, vitamin D, exercise, etc.) and explore whether COVID-19 and osteoporosis are causally related to each other.
2.2 Instrumental Variables Selection Principles
Single nucleotide polymorphism (SNPs) were selected as instrumental variables based on the following principles: (1) SNPs with genome-wide significance (P<1.0×10-8); (2) SNPs were independent, by calculating the linkage disequilibrium based on the European 1,000 Genome reference panel(r2<0.01). In reverse MR analysis, to determine whether SNPs are associated with potential risk factors, we searched for all SNPs in PhenoScanner (http://www.phenoscanner.medschl.cam.ac.uk/)[16-18]. SNPs potentially related to factors associated with COVID-19 infection or severity, such as blood cell-related indices[19], body fat[20], ulcerative colitis[21,22], etc., were excluded. Weak instrument variables were evaluated by calculating the F-statistic[23], with an F-statistic of 10 considered sufficiently robust to mitigate weak instrument bias[24].
2.3 Data Sources and Instrumental Variables Selection for Forward MR
In the forward MR analysis, instrumental variables were derived from the top 10k SNPs of COVID19-hg GWAS meta-analyses round 7 (https://www.covid19hg.org/results/r7/). For the SNPs with missing RSID, we retrieve the RSID through ‘https://www.ncbi.nlm.nih.gov/snp/’ using EA, OA, POF and other indicators. Instrumental variables obtained under different exposure scenarios according to selection principles: 7 SNPs for severe COVID-19; 10 SNPs for hospitalized COVID-19; 6 SNPs for COVID-19 infection. The outcome data were obtained from the following two GWAS datasets: the osteoporosis patient dataset (https://gwas.mrcieu.ac.uk/datasets/finn-b-M13_OSTEOPOROSIS/) and the drug-induced osteoporosis patient dataset (https://gwas.mrcieu.ac.uk/datasets/finn-b-DRUGADVERS_OSTEOPO/). Table S1 summarizes the basic information and demographic characteristics of each dataset., Table S2 summarizes those SNPs.
2.4 Data Sources and Instrumental Variables Selection for Reverse MR
In the reverse MR analysis, instrumental variables were derived from the dataset of non-cancer illness code self-reported osteoporosis (ukb-a-87, https://gwas.mrcieu.ac.uk/datasets/ukb-a-87/). 12 SNPs were used as instrumental variables according to the selection principles. The outcome data were obtained from the COVID19-hg GWAS meta-analyses round 7 (https://www.covid19hg.org/results/r7/). Table S1 summarizes the basic information and demographic characteristics of each dataset.
For the binary outcomes, the odd ratios (OR) and 95% credibility interval (CI) were applied to estimate the degree of causality. Causal estimates for binary outcomes, p-values, β and their standard errors are also given. All p-values are two-tailed. All analyses were performed by applying the TwoSampleMR, MendelianRandomization and data.table packages in R (version 4.2.2, www.r-project.org/).
3 RESULTS
3.1 Forward MR Analysis
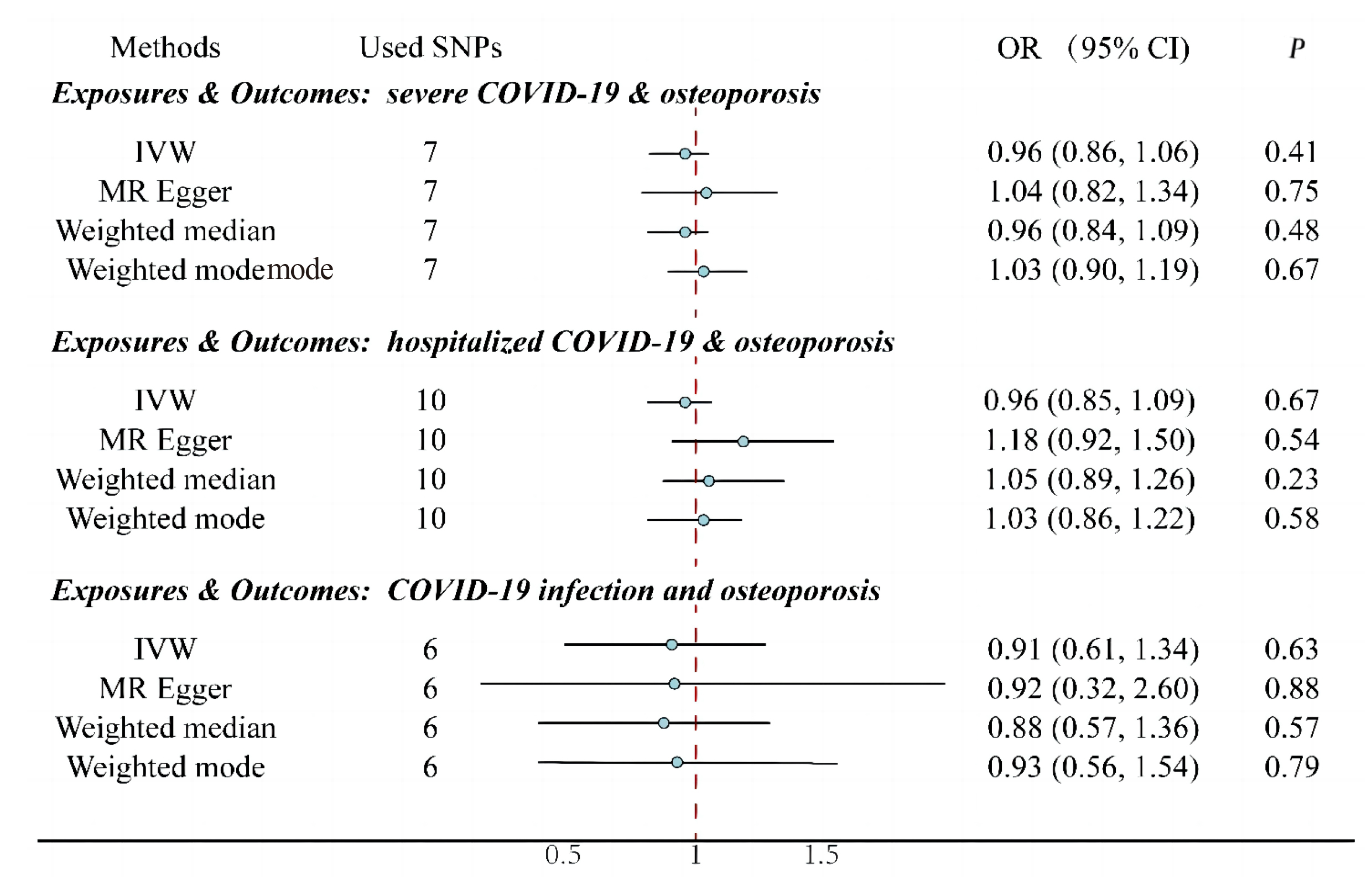
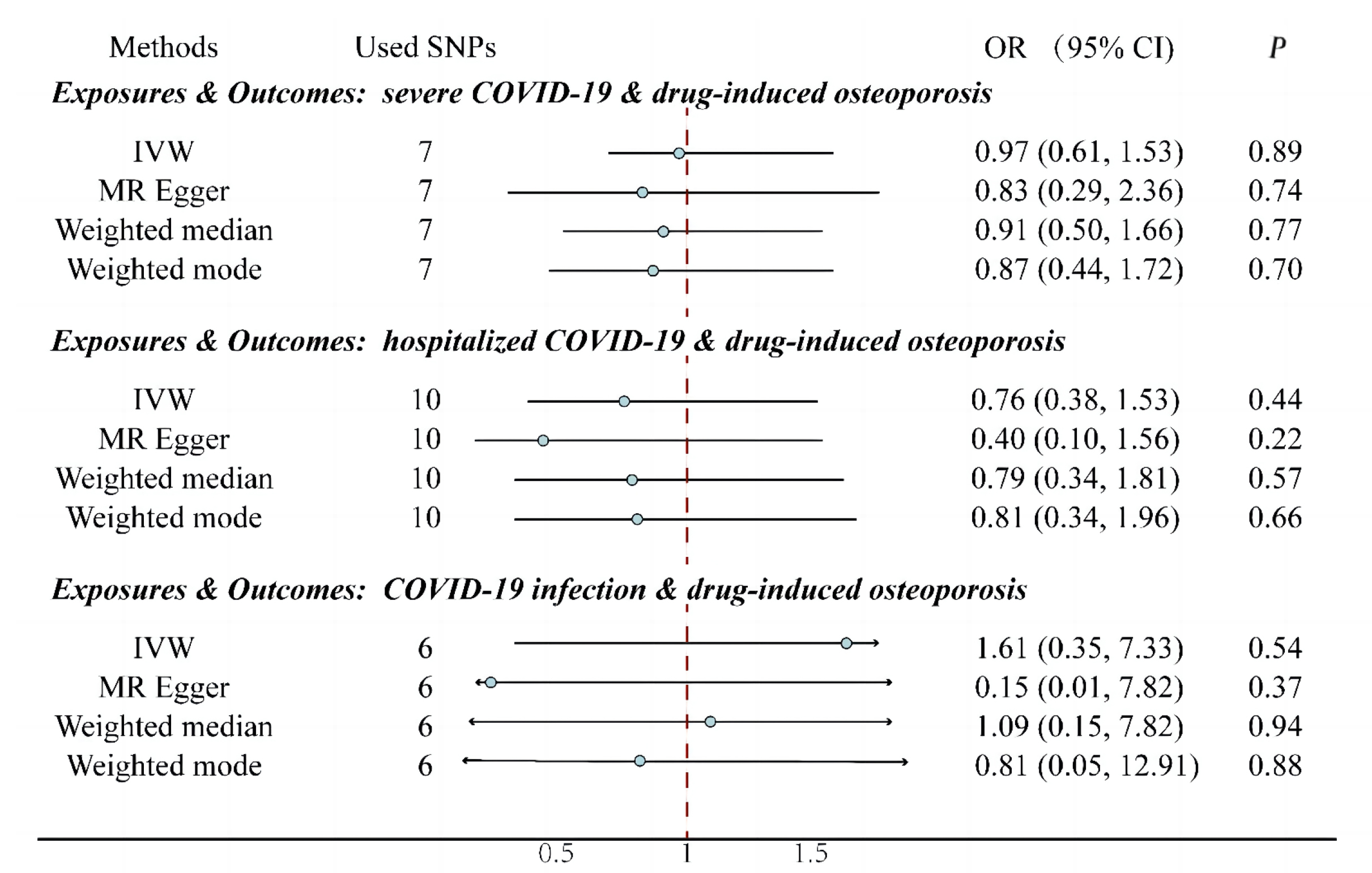
There was no significant relationship between COVID-19 patients and osteoporosis or drug-induced osteoporosis by genetic prediction, as shown in Figure 2 and Figure 3. Neither expose to severe COVID-19 (OR: 0.96; 95% CI: 0.86-1.06; P=0.41) nor hospitalized COVID-19 (OR: 0.96; 95% CI: 0.85-1.09; P=0.67) nor COVID-19 infection (OR: 0.91; 95% CI: 0.61-1.34; P=0.63) had a genetically determined responsibility for the outcome of osteoporosis (Figure 2). There was also no genetically determined liability between drug-induced osteoporosis outcomes and exposure to COVID-19 infection (OR: 1.61; 95% CI: 0.35-7.33; P=0.54), COVID-19 hospitalization (OR: 0.76; 95% CI: 0.38-1.53; P=0.44), and severe COVID-19 (OR: 0.97; 95% CI: 0.61-1.53; P=0.89) (Figure 3). All p-values were greater than 0.05 in heterogeneity analysis (Table 1) and pleiotropic effect test (Table 2). Therefore, it was concluded that there was no heterogeneity or pleiotropic effect.
|
Figure 2. Effect of exposure to COVID-19 (infection, hospitalization, and severe disease) on the outcomes of osteoporosis. IVW: Inverse Variance Weighting; Used SNPs: Number of SNPs used as working variables when performing MR. SNPs inclusion criteria: P<1×10-8, r2<0.01. There was no significant relationship between COVID-19 patients and osteoporosis by MR.
|
Figure 3. Effect of exposure to COVID-19 (infection, hospitalization, and severe disease) on the outcomes of drug-induced osteoporosis. IVW: Inverse Variance Weighting; Used SNPs: Number of SNPs used as working variables when performing MR. SNPs inclusion criteria: P<1×10-8, r2<0.01. There was no significant relationship between COVID-19 patients and drug-induced osteoporosis by MR.
Table 1. MR Heterogeneity Test
Exposures |
Outcomes |
Methods |
Q |
Q_df |
Q_pval |
Forward MR |
|||||
Severe COVID-19 |
Osteoporosis |
MR Egger |
7.02 |
5 |
0.22 |
IVW |
7.88 |
6 |
0.25 |
||
Drug-induced osteoporosis |
MR Egger |
5.28 |
5 |
0.38 |
|
IVW |
5.39 |
6 |
0.5 |
||
Hospitalized COVID-19 |
Osteoporosis |
MR Egger |
5.61 |
8 |
0.69 |
IVW |
9.08 |
9 |
0.43 |
||
Drug-induced osteoporosis |
MR Egger |
10.31 |
8 |
0.24 |
|
IVW |
11.78 |
9 |
0.23 |
||
COVID-19 infection |
Osteoporosis |
MR Egger |
7.91 |
4 |
0.09 |
IVW |
7.91 |
5 |
0.16 |
||
Drug-induced osteoporosis |
MR Egger |
2.54 |
4 |
0.64 |
|
IVW |
4.50 |
5 |
0.48 |
||
Reverse MR |
|||||
Osteoporosis |
Severe COVID-19 |
MR Egger |
13.93 |
11 |
0.24 |
IVW |
13.94 |
12 |
0.3 |
||
Hospitalized COVID-19 |
MR Egger |
10.49 |
11 |
0.49 |
|
IVW |
12.61 |
12 |
0.40 |
||
COVID-19 infection |
MR Egger |
11.48 |
11 |
0.40 |
|
IVW |
13.30 |
12 |
0.35 |
||
Notes: Q: Cochrane’s Q; Q_df: The freedom of Cochrane’s Q; Q_pval: P for Cochrane’s Q were derived from Cochrane’s Q test and P<0.05 indicates a possible heterogeneity. There was no heterogeneity between COVID-19 to osteoporosis.
Table 2. MR Pleiotropic Effect Test
Exposures |
Outcomes |
Methods |
Intercept |
SE |
P |
Forward MR |
|||||
Severe COVID-19 |
Osteoporosis |
MR Egger |
-0.03 |
0.03 |
0.47 |
Drug-induced osteoporosis |
MR Egger |
0.05 |
0.14 |
0.76 |
|
Hospitalized COVID-19 |
Osteoporosis |
MR Egger |
-0.04 |
0.02 |
0.10 |
Drug-induced osteoporosis |
MR Egger |
0.14 |
0.13 |
0.32 |
|
COVID-19 infection |
Osteoporosis |
MR Egger |
-0.00 |
0.04 |
0.98 |
Drug-induced osteoporosis |
MR Egger |
-0.22 |
0.16 |
0.23 |
|
Reverse MR |
|||||
Osteoporosis |
Severe COVID-19 |
MR Egger |
-0.00 |
0.03 |
0.97 |
Hospitalized COVID-19 |
MR Egger |
-0.03 |
0.02 |
0.17 |
|
COVID-19 infection |
MR Egger |
-0.01 |
0.01 |
0.21 |
|
Notes: Intercept: The farther the intercept distance 0 is, the greater the pleiotropic effect. P-values less than 0.05 are considered to have pleiotropic effect. There was no pleiotropic effect between COVID-19 to osteoporosis.
We analyzed other osteoporosis GWAS datasets from ‘ieu open gwas project’ (https://gwas.mrcieu.ac.uk/), but the results all suggested no significant genetic link between COVID-19 exposures and osteoporosis outcomes (Table S3). We also performed a secondary analysis by selecting SNPs with a significance level of P<1×10-7 and r2<0.1, and the results were still not statistically different (Table S4).
3.2 Reverse MR Analysis
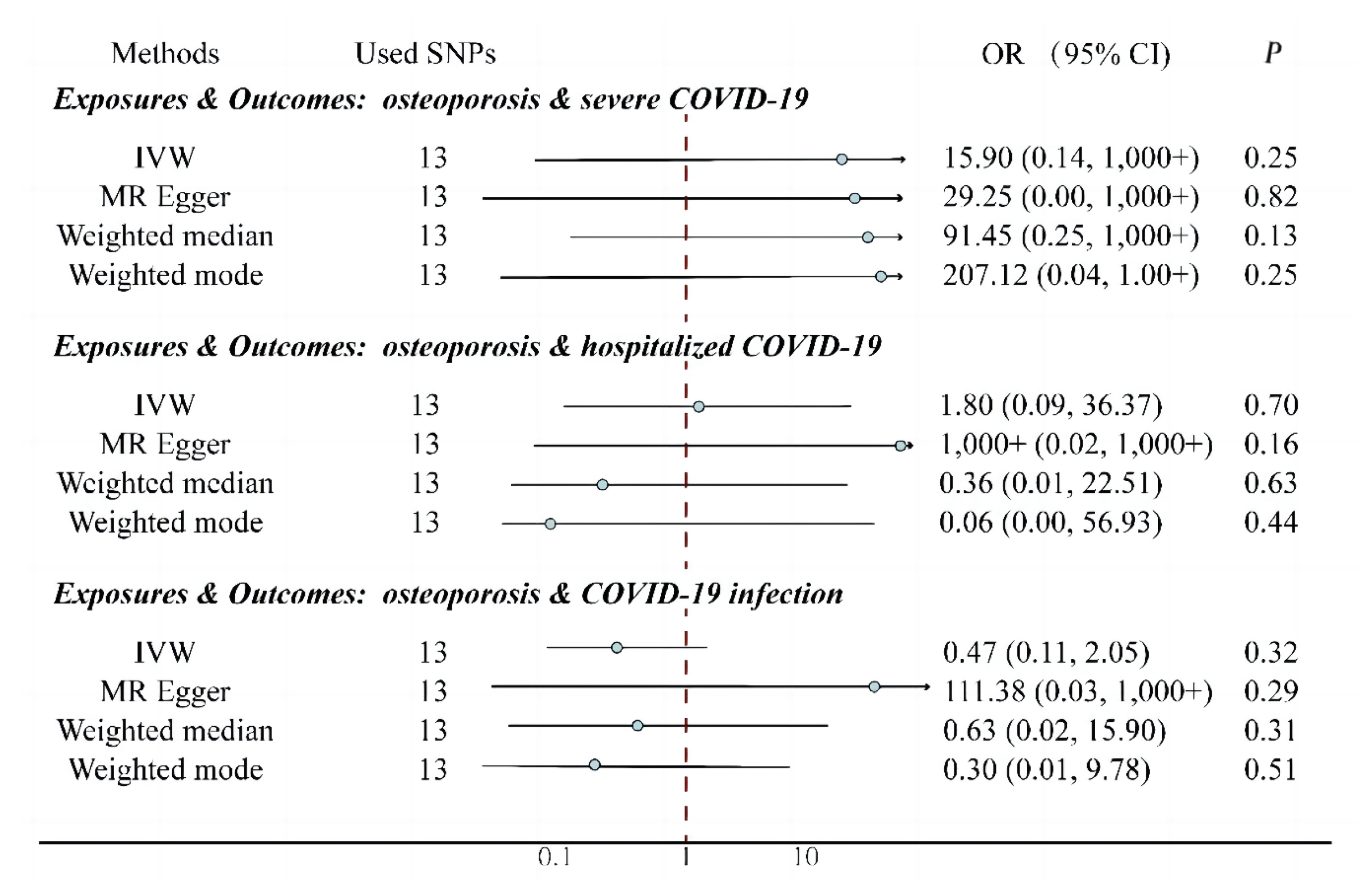
There was no significant relationship between osteoporosis exposition and COVID-19 outcomes by genetic prediction, as shown in figure 4. Patients with osteoporosis compared to non-osteoporotic patients had no genetically determined responsibility outcome of severe COVID-19, hospitalized COVID-19, COVID-19 infection (Figure 4). By pleiotropic effect test (Table 2) as well as heterogeneity analysis (Table 1), all P-values were greater than 0.05. It was concluded that there was no heterogeneity as well as pleiotropic effect.
|
Figure 4. Effect of exposure to osteoporosis on the outcomes of COVID-19 (infection, hospitalization, and severe disease). IVW: Inverse Variance Weighting; Used SNPs: Number of SNPs used as working variables when performing MR. SNPs inclusion criteria: P<1×10-8, r2<0.01. There was no significant relationship between osteoporosis exposures and COVID-19 outcomes.
We excluded SNPs associated with both osteoporosis and other diseases due to the large 95% CI of the results (Figure 4), but the 95% CI range after exclusion was still large and no significant relationship between osteoporosis exposition and COVID-19 outcomes was found by genetic prediction. The excluded SNPs and the reasons are shown in (Table S5). The excluded results are shown in (Table S6). There was no heterogeneity as well as pleiotropic effect.
4 DISCUSSION
A multicenter study showed that patients with COVID-19 requiring intensive care had significantly lower bone mineral density (BMD) than patients treated in a non-intensive care setting[31]. Some patients report persistent bone pain or burning sensation after COVID-19 infection[32]. In addition, animal studies have shown that COVID-19 infected C57/b6[7] or golden Syrian hamsters[8] activate osteoclast-mediated bone loss. BMD in patients with osteoporosis was decreased after COVID-19 infection[12]. Based on the above findings we thought that there should be a significant increase in the global prevalence of osteoporosis after the COVID-19 pandemic in recent years, yet we see no such reports. Using Mendelian regression analysis, we also found no significant association between COVID-19 infection (severe, hospitalized or infection alone) and osteoporosis outcomes.
As to why the trait appeared in animal experiments, we believe that it was assumed that a large number of COVID-19 pathogens were present for a long time in animal experiments, whereas in humans COVID-19 pathogens are present in much smaller relative numbers and for much less time. This resulted in a different outcome.
Based on relevant research[33-35], thought that the possible causes of osteoporosis after COVID-19 may come from these confounding factors: (1) the use of glucocorticoids for treat COVID-19; (2) long term stay at indoors and reduce the sun exposure time after infection; (3) decreased cardiorespiratory fitness and outdoor exercise; (4) inflammatory storm 4. inflammatory storms; (5) a significant decline in the quality of osteoporosis treatment during the COVID-19 pandemic. Therefore, we believe that for COVID-19 patients who are also at high risk for osteoporosis, the use of glucocorticoids should be minimized, and alternative medications should be considered[36]. Additionally, indoor exercise is crucial for preventing osteoporosis in these patients. We should standardize osteoporosis treatment to better face future public health emergencies[37].
Some clinical studies have found that patients with lower BMD have significantly higher rates of severe clinical morbidity compared to those with higher BMD, and that BMD can be used as a predictor of COVID-19 clinical outcomes[38-41]. However, our study showed that exposure to osteoporosis was not related to the COVID-19 infection and the symptoms. The possibility of such outcomes on clinical study may arise from the interference of confounding factors: Osteoporosis is more common in older people[40], who are more likely to develop COVID-19[42] and may have more severe COVID-19[43]; people with osteoporosis often have more comorbidities[40] and may be in worse health than non-osteoporotic patients. Therefore, the allocation of medical resources during a pandemic should not be based on bone density or prioritize osteoporotic patients for additional resources. Instead, medical resource allocation should consider other relevant indicators, such as age[44], gender[44], GDP[45], etc.
Serum 1,25-dihydroxyvitamin D (1,25(OH)(2)D) was significantly decreased in osteoporotic patients (P<0.001) than controls[46], and COVID-19 outcomes are worse in patients with low serum levels of 1,25(OH)(2)D[10,47]. VitD supplementation may reduce the risk of influenza and COVID-19 infection and death[11]. However, our findings suggest that COVID-19 infection is not associated with osteoporosis. We therefore conclude that vitamin supplementation improves COVID-19 outcomes should not act by affecting the BMD pathway.
4.1 Limitation
Our study only covers the GWAS dataset for the European population, and it is not possible to conclude whether it is applicable to the Asian and African populations; No MR analysis was performed on whether exposure to secondary infection with COVID-19 had an effect on osteoporosis outcomes; In this study, summary-level data is used to evaluate the causal relationship, not individual-level data; Not Collaboration with experts in the field.
5 CONCLUSION
In this bidirectional MR study, non-genetic association was found between osteoporosis and COVID-19. During the COVID-19 pandemic, there were many mildly ill or asymptomatic patients, and our research suggests that COVID-19 infection alone does not increase the likelihood of developing osteoporosis later in life. There is also no association between simple osteoporosis and COVID-19 infection and secondary attacks.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (NO.82070807, NO.91749118, NO.81770775, NO.81730022); National Natural Science Foundation of China Regional Science Fund Project (NO.82060857); Jiangxi Provincial Department of Education Technology Research Project (NO.GJJ2200936); Leading Talents Program of Hunan Province (NO.2022RC3078); Natural Science Foundation of Hunan Province, China (NO.2021JJ30976); and National Key Research and Development Program (NO.2019YFA0801900, NO.2018YFC2000100).
Conflicts of Interest
The authors declared no conflict of interest.
Author Contribution
Chen S was responsible for designing the study, interpreting the data, and writing the original draft. Xue S provided supervision and project administration, and contributed to the writing, review, and editing processes. Both of the authors made substantial contributions to the manuscript and approved the final version for submission.
Abbreviation List
BMD, Bone mineral density
CI, Credibility interval
GWAS, Genome-wide association study
IVW, Inverse variance weighting
MR, Mendelian randomization
OR, Odd ratios
SARS-CoV-2, Severe acute respiratory syndrome coronavirus 2
SNPs, Single nucleotide polymorphism
VitD, Vitamin D
References
[1] Chakraborty I, Maity P. COVID-19 outbreak: Migration, effects on society, global environment and prevention. Sci Total Environ, 2020; 728: 138882.[DOI]
[2] Velavan TP, Meyer CG. The COVID-19 epidemic. Trop Med Int Health, 2020; 25: 278.[DOI]
[3] Zha L, Li S, Pan L et al. Corticosteroid treatment of patients with coronavirus disease 2019 (COVID-19). Med J Australia, 2020; 212: 416-420.[DOI]
[4] Wang J, Yang W, Chen P et al. The proportion and effect of corticosteroid therapy in patients with COVID-19 infection: A systematic review and meta-analysis. PLoS One, 2021; 16: e0249481.[DOI]
[5] Lane NE. Epidemiology, etiology, and diagnosis of osteoporosis. Am J Obstet Gynecol, 2006; 194: S3-S11.[DOI]
[6] Conde FA, Aronson WJ. Risk factors for male osteoporosis. Urol Oncol-Semin Ori, 2003; 21: 380-383.[DOI]
[7] Awosanya OD, Dalloul CE, Blosser RJ et al. Osteoclast-mediated bone loss observed in a COVID-19 mouse model. Bone, 2022; 154: 116227.[DOI]
[8] Qiao W, Lau HE, Xie H et al. SARS-CoV-2 infection induces inflammatory bone loss in golden Syrian hamsters. Nat Commun, 2022; 13: 2539.[DOI]
[9] Capozzi A, Scambia G, Lello S. Calcium, vitamin D, vitamin K2, and magnesium supplementation and skeletal health. Maturitas, 2020; 140: 55-63.[DOI]
[10] Ali N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J Infect Public Heal, 2020; 13: 1373-1380.[DOI]
[11] Grant WB, Lahore H, McDonnell SL et al. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients, 2020; 12: 988.[DOI]
[12] Elmedany SH, Badr OI, Abu-Zaid MH et al. Bone mineral density changes in osteoporotic and osteopenic patients after COVID-19 infection. Egypt Rheumatol Reh, 2022; 49: 1-8.[DOI]
[13] Allman PH, Aban IB, Tiwari HK et al. An introduction to Mendelian randomization with applications in neurology. Mult Scler Relat Dis, 2018; 24: 72-78.[DOI]
[14] Sanderson E, Glymour MM, Holmes MV et al. Mendelian randomization. Nat Rev Method Prime, 2022; 2: 6.[DOI]
[15] Lawlor DA, Harbord RM, Sterne JA et al. Mendelian randomization: Using genes as instruments for making causal inferences in epidemiology. Stat Med, 2008; 27: 1133-1163.[DOI]
[16] Liang X, Cai J, Fan Y. Causal association between tea intake and risk for gout: a Mendelian randomization study. Front Genet, 2023; 14: 1220931.[DOI]
[17] Kamat MA, Blackshaw JA, Young R et al. PhenoScanner V2: an expanded tool for searching human genotype-phenotype associations. Bioinformatics, 2019; 35: 4851-4853.[DOI]
[18] Staley JR, Blackshaw J, Kamat MA et al. PhenoScanner: a database of human genotype-phenotype associations. Bioinformatics, 2016; 32: 3207-3209.[DOI]
[19] Lin Y, Yang Y, Xiang N et al. Characterization and trajectories of hematological parameters prior to severe COVID-19 based on a large-scale prospective health checkup cohort in western China: a longitudinal study of 13-year follow-up. Bmc Med, 2024; 22: 105.[DOI]
[20] Yoshiji S, Tanaka D, Minamino H et al. Causal associations between body fat accumulation and COVID-19 severity: A Mendelian randomization study. Front Endocrinol, 2022; 13: 899625.[DOI]
[21] Mazza S, Sorce A, Peyvandi F et al. A fatal case of COVID-19 pneumonia occurring in a patient with severe acute ulcerative colitis. Gut, 2020; 69: 1148-1149.[DOI]
[22] Aydın MF, Taşdemir H. Ulcerative colitis in a COVID-19 patient: A case report. Turk J Gastroenterol, 2021; 32: 543.[DOI]
[23] Andrews I, Stock JH, Sun L. Weak instruments in instrumental variables regression: Theory and practice. Annu Rev Econ, 2019; 11: 727-753.[DOI]
[24] Staiger DO, Stock JH. Instrumental variables regression with weak instruments. National Bureau of Economic Research Cambridge, Mass, USA, 1994.[DOI]
[25] Cheung CL, Ho SC, Krishnamoorthy S et al. COVID-19 and platelet traits: A bidirectional Mendelian randomization study. J Med Virol, 2022; 94: 4735-4743.[DOI]
[26] Li X, Cheng S, Cheng J et al. Habitual Coffee Consumption Increases Risk of Primary Open-Angle Glaucoma: A Mendelian Randomization Study. Ophthalmology, 2022; 129: 1014-1021.[DOI]
[27] Peng H, Wang S, Wang M et al. Nonalcoholic fatty liver disease and cardiovascular diseases: A Mendelian randomization study. Metabolism, 2022; 133: 155220.[DOI]
[28] Zhou T, Sun D, Li X et al. Educational attainment and drinking behaviors: Mendelian randomization study in UK Biobank. Mol Psychiatry, 2021; 26: 4355-4366.[DOI]
[29] Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol, 2017; 32: 377-389.[DOI]
[30] Wu F, Huang Y, Hu J et al. Mendelian randomization study of inflammatory bowel disease and bone mineral density. BMC Med, 2020; 18: 312.[DOI]
[31] Kottlors J, Große Hokamp N, Fervers P et al. Early extrapulmonary prognostic features in chest computed tomography in COVID-19 pneumonia: Bone mineral density is a relevant predictor for the clinical outcome - A multicenter feasibility study. Bone, 2021; 144: 115790.[DOI]
[32] Davis HE, Assaf GS, McCorkell L et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine, 2021; 38: 101019.[DOI]
[33] Cvijetić S, Keser I, Boschiero D et al. Osteosarcopenic Adiposity and Nutritional Status in Older Nursing Home Residents during the COVID-19 Pandemic. Nutrients, 2023; 15: 227.[DOI]
[34] Ozen S, Cakmak E. Prevalence of chronic low back pain and associated risk factors in healthcare workers during the Covid-19 pandemic. COJ Nurse Healthcare, 2021; 7:810-8.
[35] Peeters JJM, van den Berg P, van den Bergh JP et al. Osteoporosis care during the COVID-19 pandemic in the Netherlands: A national survey. Arch Osteoporos, 2021; 16: 1-8.
[36] Yu EW, Tsourdi E, Clarke BL et al. Osteoporosis management in the era of COVID-19. J Bone Miner Res, 2020; 35: 1009-1013.[DOI]
[37] Lippi G, Henry BM, Bovo C et al. Health risks and potential remedies during prolonged lockdowns for coronavirus disease 2019 (COVID-19). Diagnosis, 2020; 7: 85-90.[DOI]
[38] Cromer SJ, Yu EW. Challenges and opportunities for osteoporosis care during the COVID-19 pandemic. J Clin Endocr Metab, 2021; 106: e4795-e4808.[DOI]
[39] Hasan LK, Deadwiler B, Haratian A et al. Effects of COVID-19 on the musculoskeletal system: clinician’s guide. Orthop Res Rev, 2021; 13: 141-150.[DOI]
[40] Tahtabasi M, Kilicaslan N, Akin Y et al. The prognostic value of vertebral bone density on chest CT in hospitalized COVID-19 patients. J Clin Densitom, 2021; 24: 506-515.[DOI]
[41] Kottlors J, Hokamp NG, Fervers P et al. Early extrapulmonary prognostic features in chest computed tomography in COVID-19 pneumonia: Bone mineral density is a relevant predictor for the clinical outcome-A multicenter feasibility study. Bone, 2021; 144: 115790.[DOI]
[42] Noor FM, Islam MM. Prevalence and associated risk factors of mortality among COVID-19 patients: a meta-analysis. J Commun Health, 2020; 45: 1270-1282.[DOI]
[43] Tosato M, Carfì A, Martis I et al. Prevalence and predictors of persistence of COVID-19 symptoms in older adults: a single-center study. J Am Med Dir Assoc, 2021; 22: 1840-1844.[DOI]
[44] Barek MA, Aziz MA, Islam MS. Impact of age, sex, comorbidities and clinical symptoms on the severity of COVID-19 cases: A meta-analysis with 55 studies and 10014 cases. Heliyon, 2020; 6: e05684.[DOI]
[45] Basak P, Abir T, Al Mamun A et al. A global study on the correlates of gross domestic product (GDP) and COVID-19 vaccine distribution. Vaccines, 2022; 10: 266.[DOI]
[46] Gallagher JC, Riggs BL, Eisman J et al. Intestinal calcium absorption and serum vitamin D metabolites in normal subjects and osteoporotic patients: effect of age and dietary calcium. J Clin Invest, 1979; 64: 729-736.[DOI]
[47] Ye K, Tang F, Liao X et al. Does serum vitamin D level affect COVID-19 infection and its severity?-A case-control study. J Am Coll Nutr, 2021; 40: 724-731.[DOI]
Copyright © 2024 The Author(s). This open-access article is licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, sharing, adaptation, distribution, and reproduction in any medium, provided the original work is properly cited.





 Copyright ©
Copyright ©